

Chinese Journal of Organic Chemistry >
Advances in Transition-Metal-Catalyzed Keto Carbonyl-Directed C—H Bond Functionalization Reactions
Received date: 2022-05-20
Revised date: 2022-08-09
Online published: 2022-08-25
Supported by
National Natural Science Foundation of China(22025109); National Natural Science Foundation of China(21772202); Beijing National Laboratory for Molecular Sciences(BNLMS-CXXM-201901); K. C. Wong Education Foundation
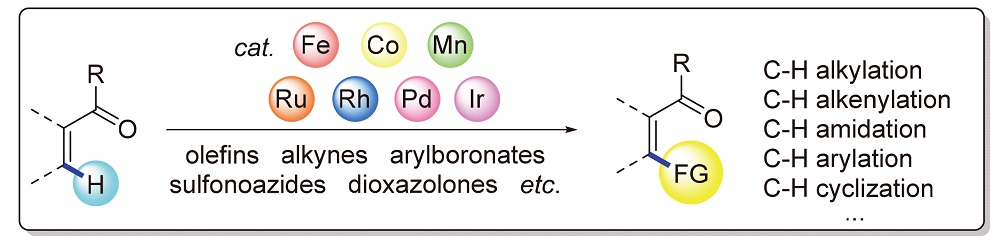
In the past two decades, transition-metal-catalyzed keto carbonyl-directed C—H bond activation has evloved as a powerful and convenient tool for the construction of C—C and C—X (X=N, F, O) bonds at the unconventional reaction sites of ketones. Among them, keto carbonyl-directed C—H bond activation reactions catalyzed by noble metals, involving ruthenium, rhodium, palladium and iridium, have been widely explored, whilst inexpensive 3d metals, such as manganese, iron and cobalt, have gradually emerged as hotspot catalysts in keto carbonyl-directed C—H activation reactions recently. In this review, advances on transition-metal-catalyzed keto carbonyl-directed C—H bond functionalization reactions from 2014 to 2021 are summarized, which are devided by reaction categories such as alkylation, alkenylation, amidation, arylation, cyclization, and so on.
Silin Chen , Yunhui Yang , Chao Chen , Congyang Wang . Advances in Transition-Metal-Catalyzed Keto Carbonyl-Directed C—H Bond Functionalization Reactions[J]. Chinese Journal of Organic Chemistry, 2023 , 43(1) : 1 -16 . DOI: 10.6023/cjoc202205033
| [1] | (a) Zheng, Q.-Z.; Jiao, N. Tetrahedron Lett. 2014, 55, 1121. |
| [1] | (b) Huang, Z.; Lim, H. N.; Mo, F.; Young, M. C.; Dong, G. Chem. Soc. Rev. 2015, 44, 7764. |
| [1] | (c) Sambiagio, C.; Schonbauer, D.; Blieck, R.; Dao-Huy, T.; Pototschnig, G.; Schaaf, P.; Wiesinger, T.; Zia, M. F.; Wencel- Delord, J.; Besset, T.; Maes, B. U. W.; Schnurch, M. Chem. Soc. Rev. 2018, 47, 6603. |
| [2] | Murai, S.; Kakiuchi, F.; Sekine, S.; Tanaka, Y.; Kamatani, A.; Sonoda, M.; Chatani, N. Nature 1993, 366, 529. |
| [3] | Wang, Z.; Wang, C. Green Synth. Catal. 2021, 2, 66. |
| [4] | Crisenza, G. E.; McCreanor, N. G.; Bower, J. F. J. Am. Chem. Soc. 2014, 136, 10258. |
| [5] | Tsuchikama, K.; Kasagawa, M.; Hashimoto, Y.-K.; Endo, K.; Shibata, T. J. Organomet. Chem. 2008, 693, 3939. |
| [6] | Kimura, N.; Kochi, T.; Kakiuchi, F. J. Am. Chem. Soc. 2017, 139. 14849. |
| [7] | Kimura, N.; Kochi, T.; Kakiuchi, F. Asian J. Org. Chem. 2019, 8, 1115. |
| [8] | Kommagalla, Y.; Srinivas, K.; Ramana, C. V. Chem.-Eur. J. 2014, 20, 7884. |
| [9] | Srinivas, K.; Dangat, Y.; Kommagalla, Y.; Vanka, K.; Ramana, C. V. Chem.-Eur. J. 2017, 23, 7570. |
| [10] | Bettadapur, K. R.; Lanke, V.; Prabhu, K. R. Org. Lett. 2015, 17. 4658. |
| [11] | Sherikar, M. S.; Kapanaiah, R.; Lanke, V.; Prabhu, K. R. Chem. Commun. 2018, 54, 12113. |
| [12] | Han, S. H.; Kim, S.; De, U.; Mishra, N. K.; Park, J.; Sharma, S.; Kwak, J. H.; Han, S.; Kim, H. S.; Kim, I. S. J. Org. Chem. 2016, 81, 12416. |
| [13] | Borah, A. J.; Shi, Z. Chem. Commun. 2017, 53, 3945. |
| [14] | Li, J.-F.; Zhao, R.-F.; Zhou, F.-Q.; She, M.-Y.; Zhang, J.; Yin, B.; Zhang, S.-Y.; Li, J.-L. Org. Chem. Front. 2019, 6, 2607. |
| [15] | Chen, X.; Zheng, G.; Li, Y.; Song, G.; Li, X. Org. Lett. 2017, 19, 6184. |
| [16] | Lee, S. H.; Kundu, A.; Han, S. H.; Mishra, N. K.; Kim, K. S.; Choi, M. H.; Pandey, A. K.; Park, J. S.; Kim, H. S.; Kim, I. S. ACS Omega 2018, 3, 2661. |
| [17] | Shang, R.; Ilies, L.; Nakamura, E. J. Am. Chem. Soc. 2016, 138, 10132. |
| [18] | Santhoshkumar, R.; Mannathan, S.; Cheng, C. H. Org. Lett. 2014, 16, 4208. |
| [19] | Li, G.; Wan, L.; Zhang, G.; Leow, D.; Spangler, J.; Yu, J. Q. J. Am. Chem. Soc. 2015, 137, 4391. |
| [20] | Hu, Y.; Zhou, B.; Chen, H.; Wang, C. Angew. Chem., Int. Ed. 2018, 57, 12071. |
| [21] | Wang, C.; Zhang, Q. Green Synth. Catal. 2022, 3, 287. |
| [22] | Lanke, V.; Bettadapur, K. R.; Prabhu, K. R. Org. Lett. 2016, 18, 5496. |
| [23] | Bakthadoss, M.; Kumar, P. V.; Reddy, T. S. Eur. J. Org. Chem. 2017, 2017, 4439. |
| [24] | Sk, M. R.; Bera, S. S.; Maji, M. S. Adv. Synth. Catal. 2019, 361, 585. |
| [25] | Sk, M. R.; Maji, M. S. Org. Chem. Front. 2020, 7, 19. |
| [26] | Elumalai, K.; Leong, W. K. Tetrahedron Lett. 2018, 59, 113. |
| [27] | (a) Li, C.; Wang, S. M.; Qin, H. L. Org. Lett. 2018, 20, 4699. |
| [27] | (b) Bettadapur, K. R.; Sherikar, M. S.; Lanke, V.; Prabhu, K. R. Asian J. Org. Chem. 2018, 7, 1338. |
| [28] | Dana, S.; Giri, C. K.; Baidya, M. Org. Lett. 2021, 23, 6855. |
| [29] | Hu, F.; Szostak, M. Chem. Commun. 2016, 52, 9715. |
| [30] | Kim, J.; Chang, S. Angew. Chem., Int. Ed. 2014, 53, 2203. |
| [31] | Kong, X.; Xu, B. Org. Lett. 2018, 20, 4495. |
| [32] | Bera, S. S.; Sk, M. R.; Maji, M. S. Chem.-Eur. J. 2019, 25, 1806. |
| [33] | Kim, Y.; Park, J.; Chang, S. Org. Lett. 2016, 18, 1892. |
| [34] | Song, Z.; Antonchick, A. P. Org. Biomol. Chem. 2016, 14, 4804. |
| [35] | Xu, L.; Tan, L.; Ma, D. J. Org. Chem. 2016, 81, 10476. |
| [36] | Hande, A. E.; Prabhu, K. R. J. Org. Chem. 2017, 82, 13405. |
| [37] | Lanke, V.; Prabhu, K. R. Chem. Commun. 2017, 53, 5117. |
| [38] | Chen, S.; Feng, B.; Zheng, X.; Yin, J.; Yang, S.; You, J. Org. Lett. 2017, 19, 2502. |
| [39] | Shi, X.; Xu, W.; Wang, R.; Zeng, X.; Qiu, H.; Wang, M. J. Org. Chem. 2020, 85, 3911. |
| [40] | Wang, F.; Jin, L.; Kong, L.; Li, X. Org. Lett. 2017, 19, 1812. |
| [41] | Shi, P.; Wang, L.; Chen, K.; Wang, J.; Zhu, J. Org. Lett. 2017, 19, 2418. |
| [42] | Yamamoto, T.; Yamakawa, T. RSC Adv. 2015, 5, 105829. |
| [43] | Ogiwara, Y.; Miyake, M.; Kochi, T.; Kakiuchi, F. Organometallics 2017, 36, 159. |
| [44] | Suzuki, I.; Kondo, H.; Kochi, T.; Kakiuchi, F. J. Org. Chem. 2019, 84, 12975. |
| [45] | Zhang, B.; Wang, H. W.; Kang, Y. S.; Zhang, P.; Xu, H. J.; Lu, Y.; Sun, W. Y. Org. Lett. 2017, 19. |
| [46] | Zhang, C.; Rao, Y. Org. Lett. 2015, 17, 4456. |
| [47] | Bruneau, C.; Gramage-Doria, R. Adv. Synth. Catal. 2016, 358, 3847. |
| [48] | Paymode, D. J.; Ramana, C. V. J. Org. Chem. 2015, 80, 11551. |
| [49] | Yang, Y.; Gao, P.; Zhao, Y.; Shi, Z. Angew. Chem., Int. Ed. 2017, 56, 3966. |
| [50] | Rago, A. J.; Dong, G. Green Synth. Catal. 2021, 2, 216. |
| [51] | Shibata, T.; Ryu, N.; Takano, H. Adv. Synth. Catal. 2015, 357, 1131. |
| [52] | Shinde, V. S.; Mane, M. V.; Cavallo, L.; Rueping, M. Chem.-Eur. J. 2020, 26, 8308. |
| [53] | Zhou, S.; Wang, J.; Wang, L.; Song, C.; Chen, K.; Zhu, J. Angew. Chem., Int. Ed. 2016, 55, 9384. |
| [54] | Zhao, Y.; Li, S.; Zheng, X.; Tang, J.; She, Z.; Gao, G.; You, J. Angew. Chem., Int. Ed. 2017, 56, 4286. |
| [55] | Yu, Y.; Wu, Q.; Liu, D.; Hu, L.; Yu, L.; Tan, Z.; Zhu, G. J. Org. Chem. 2019, 84, 7449. |
| [56] | Zhou, B.; Hu, Y.; Liu, T.; Wang, C. Nat. Commun. 2017, 8, 1169. |
| [57] | Liu, T.; Hu, Y.; Yang, Y.; Wang, C. CCS Chem. 2020, 2, 749. |
| [58] | Huo, J.; Yang, Y.; Wang, C. Org. Lett. 2021, 23, 3384. |
| [59] | Dethe, D. H.; C, B. N.; Bhat, A. A. J. Org. Chem. 2020, 85, 7565. |
| [60] | Yanagawa, M.; Harada, S.; Hirose, S.; Nemoto, T. Adv. Synth. Catal. 2021, 363, 2189. |
| [61] | Sk, M. R.; Bera, S. S.; Maji, M. S. Org. Lett. 2018, 20, 134. |
| [62] | Ali, S.; Huo, J.; Wang, C. Org. Lett. 2019, 21, 6961. |
| [63] | Zhang, K.; Khan, R.; Chen, J.; Zhang, X.; Gao, Y.; Zhou, Y.; Li, K.; Tian, Y.; Fan, B. Org. Lett. 2020, 22, 3339. |
| [64] | Lee, P. Y.; Liang, P.; Yu, W. Y. Org. Lett. 2017, 19, 2082. |
| [65] | Zhang, J.; Wu, M.; Fan, J.; Xu, Q.; Xie, M. Chem. Commun. 2019, 55, 8102. |
| [66] | Wu, Q.; Mao, Y. J.; Zhou, K.; Wang, S.; Chen, L.; Xu, Z. Y.; Lou, S. J.; Xu, D. Q. Chem. Commun. 2021, 57, 4544. |
| [67] | Tan, X.; Massignan, L.; Hou, X.; Frey, J.; Oliveira, J. C. A.; Hussain, M. N.; Ackermann, L. Angew. Chem., Int. Ed. 2021, 60, 13264. |
/
| 〈 |
|
〉 |