

Chinese Journal of Organic Chemistry >
Three New Secondary Metabolites from the Mangrove-Derived Fungus Daldinia eschscholtzii HJ004
Received date: 2022-05-23
Revised date: 2022-07-11
Online published: 2022-08-25
Supported by
Key Research and Development Program of Hainan Province(ZDYF2021SHFZ270); National Natural Science Foundation of China(32160108); National Natural Science Foundation of China(41866005); Natural Science Foundation of Hainan Province(220RC593); Key Science and Technology Program of Hainan Province(ZDKJ202008); Specific Research Fund of the Innovation Platform for Academicians of Hainan Province(YSPTZX202030)
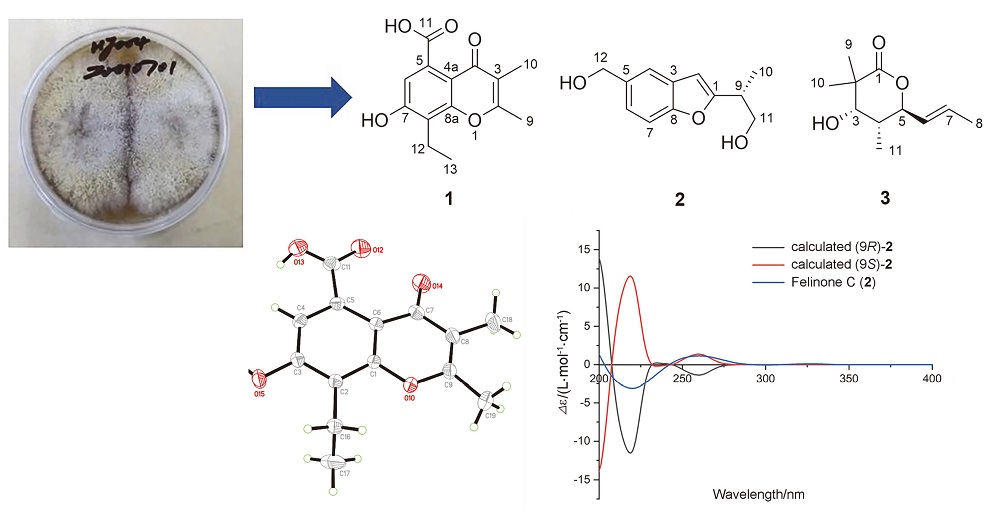
Three new compounds, together with eight known compounds, were isolated from the mangrove-derived fungus Daldinia eschscholtzii HJ004. The structure of 8-ethyl-7-hydroxy-5-carboxyl-2,3-dimethylchromone (1) was extensively elucidated and characterized by HRESIMS, 1D and 2D NMR, and single-crystal X-ray crystallography. The absolute configurations of felinone C (2) and helicascolide G (3) were determined by using electronic circular dichroism (ECD) method or comparing circular dichroism (CD) data with the literature. The radical scavenging capabilities of the 11 compounds obtained were tested by total antioxidant capacity assay kit with a rapid ABTS method. Among them 4 compounds exhibited stronger antioxidant activity with IC50 values range from 5.57 μmol/L to 195.03 μmol/L, than the positive control trolox (IC50=292.12 μmol/L). These results indicate that chromones have the potential values of developing antioxidants.
Key words: Daldinia eschscholtzii; chromone; lactone; antioxidant activity
Wang Bin , Zeng Weinv , Li Gaoyu , Xiao Mei , Wei Fangfang , Luo Youping , Niu Zhigang , Huang Guolei , Zheng Caijuan . Three New Secondary Metabolites from the Mangrove-Derived Fungus Daldinia eschscholtzii HJ004[J]. Chinese Journal of Organic Chemistry, 2023 , 43(1) : 332 -337 . DOI: 10.6023/cjoc202205041
| [1] | Chen, S. H.; Cai, R. L.; Liu, Z. M.; Cui, H.; She, Z. G. Nat. Prod. Rep. 2022, 39, 560. |
| [2] | Deshmukh, S. K.; Gupta, M. K.; Prakash, V.; Reddy, M. S. J. Fungi 2018, 4, 101. |
| [3] | Li, S. J.; Jiao, F. W.; Li, W.; Zhang, X.; Yan, W.; Jiao, R. H. J. Nat. Prod. 2020, 83, 2976. |
| [4] | Carroll, A. R.; Copp, B. R.; Davis, R. A.; Keyzers, R. A.; Prinsep, M. R. Nat. Prod. Rep. 2021, 38, 362. |
| [5] | Guo, H. X.; Huang, C. Y.; Yan, Z. Y.; Chen, T.; Hong, K.; Long, Y. H. Chin. J. Nat. Med. 2020, 18, 855. |
| [6] | Zhang, Y. L.; Zhang, J.; Jiang, N.; Lu, Y. H.; Wang, L.; Xu, S. H.; Wang, W.; Zhang, G. F.; Xu, Q.; Ge, H. M.; Ma, J.; Song, Y. C.; Tan, R. X. J. Am. Chem. Soc. 2011, 133, 5931. |
| [7] | Zhang, A. H.; Jiang, N.; Wang, X. Q.; Tan, R. X. Sci. Rep. 2019, 9, 1. |
| [8] | Du, L.; King, J. B.; Cichewicz, R. H. J. Nat. Prod. 2014, 77, 2454. |
| [9] | Lin, L. P.; Tan, R. X. Chin. J. Chem. 2018, 36, 749. |
| [10] | Qin, X. D.; Dong, Z. J.; Liu, J. K.; Yang, L. M.; Wang, R. R.; Zheng, Y. T.; Lu, Y.; Wu, Y. S.; Zheng, Q. T. Helv. Chim. Acta 2006, 89, 127. |
| [11] | Mei, R. Q.; Huang, G. L.; Wang, B.; Bai, M.; Luo, Y. P.; Chen, G. Y.; Zheng, C. J. Chin. J. Org. Chem. 2019, 39, 1479. (in Chinese) |
| [11] | (梅荣清, 黄国雷, 王斌, 白猛, 罗由萍, 陈光英, 郑彩娟, 有机化学, 2019, 39, 1479.) |
| [12] | Bai, M.; Zheng, C. J.; Huang, G. L.; Mei, R. Q.; Wang, B.; Luo, Y. P.; Zheng, C.; Niu, Z. G.; Chen, G. Y. J. Nat. Prod. 2019, 82, 1155. |
| [13] | Liao, H. X.; Shao, T. M.; Mei, R. Q.; Huang, G. L.; Zhou, X. M.; Zheng, C. J.; Wang, C. Y. Mar. Drugs 2019, 17, 710. |
| [14] | Liao, H. X.; Zheng, C. J.; Huang, G. L.; Mei, R. Q.; Nong, X. H.; Shao, T. M.; Chen, G. Y.; Wang, C. Y. J. Nat. Prod. 2019, 82, 2211. |
| [15] | Du, F. Y.; Li, X. M.; Zhang, P.; Li, C. S.; Wang, B. G. Mar. Drugs 2014, 12, 2816. |
| [16] | Poch, G. K.; Gloer, J. B. J. Nat. Prod. 1989, 52, 257. |
| [17] | Rao, C. R.; Venkateswarlu, V. Recl. Trav. Chim. Pays-Bas 1956, 75, 1321. |
| [18] | Dai, J. Q.; Krohn, K.; Floerke, U.; Draeger, S.; Schulz, B.; Kiss-Szikszai, A.; Antus, S.; Kurtan, T.; Van Ree, T. Eur. J. Org. Chem. 2006, 2006, 3498. |
| [19] | Yang, W. C.; Chen, Y.; Cai, R. L.; Zou, G.; Wang, B.; She, Z. G. Chem. Biodiversity 2020, 17, e2000192. |
| [20] | Kato, H.; Li, W.; Koike, M.; Wang, Y. H.; Koike, K. Phytochemistry 2010, 71, 1925. |
| [21] | Cao, F.; Yang, Q.; Shao, C. L.; Kong, C. J.; Zheng, J. J.; Liu, Y. F.; Wang, C. Y. Mar. Drugs 2015, 13, 4171. |
| [22] | Zhang, P.; Meng, L. H.; Attila, M.; Li, X. M.; Tibor, K.; Wang, B. G. RSC Adv. 2015, 5, 39870. |
| [23] | Chen, Y.; Meng, G. L.; Bai, W. L.; Ma, Y.; Xie, L. P.; Altaf, N.; Qian, Y. N.; Han, Y.; Ji, Y. Clin. Exp. Pharmacol. Physiol. 2017, 44, 266. |
/
| 〈 |
|
〉 |