

Chinese Journal of Organic Chemistry >
Preparation of Axially Grafted Temperature-Responsive Chiral Salen MnIII and Application in Asymmetric Epoxidation of Olefins in Water
Received date: 2022-06-16
Revised date: 2022-08-01
Online published: 2022-08-25
Supported by
Hubei Provincial Education Department Youth and Middle-Aged Talents Project(Q20212704); National Natural Science Foundation of China(22108065); National Natural Science Foundation of China(21774029)
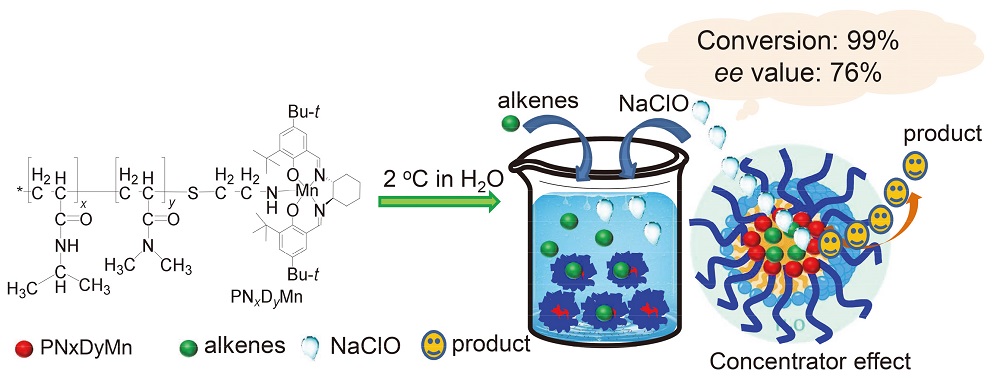
A series of temperature-responsive polymers (poly N-isopropylacrylamide-co-N,N-dimethylacrylamide, PNxDy) were prepared by using N-isopropylacrylamide (NIPAAm) and N,N-dimethylacrylamide (DMAM) as reaction monomers, azobisisobutyronitrile as chain initiator, and mercaptoethylamine hydrochlorides as chain transfer agents. Then, a novel thermoresponsive surfactant-type chiral salen MnIII catalyst PNxDyMn was developed by axially grafting “smart” polymer (PNxDy) onto the metal center of a neat chiral salen MnIII complex. The temperature-sensitivity, structure and morphology of the polymers were studied through a series of characterizations, and it was found that these temperature-responsive chiral polymers can efficiently catalyze the asymmetric epoxidation of olefins in pure water. Only 0.5 mol% PN75D5Mn was sufficient to exhibit extremely high activity (conversion>99%) with up to 76% enantioselectivity and turnover frequency (TOF) (396/h) for a substrate of indene in water. The conversion rate of the substrate styrene was as high as 99% in 5 min, and the TOF value reached 2376/h. Characterization and experiments proved that PNxDyMn can self-assemble into nanoreactors for the asymmetric epoxidation of olefin in water, leading to an acceleration of reaction and causing selective effects. After reaction, the PNxDyMn exhibited hydrophobic properties upon heating above its lower critical solution temperature (LCST), and precipitated from the aqueous system for recovery. It can be recycled and reused for seven times without significant loss of activity, and no organic solvents were required for the reaction and separation process.
Biao Han , Yaoyao Zhang , Shuhan Chen , Mengge Zhao , Nan Li , Weishuang Li , Lei Zhu . Preparation of Axially Grafted Temperature-Responsive Chiral Salen MnIII and Application in Asymmetric Epoxidation of Olefins in Water[J]. Chinese Journal of Organic Chemistry, 2023 , 43(1) : 244 -253 . DOI: 10.6023/cjoc202206026
| [1] | Nguyen, L. T.; Baudequin, T.; Cui, Z. F.; Ye, H. Biotechnol. Bioeng. 2022, 119, 2345. |
| [2] | Don, T. M.; Lu, K. Y.; Lin, L. J.; Hsu, C. H.; Wu, J. Y.; Mi, F. L. Mol. Pharmaceut. 2017, 14, 4648. |
| [3] | Zhang, Y. Y.; Han, B.; Zhou, L. J.; Wang, M. Y.; Li, B. J.; Wang, L. S.; Zhu, L. Acta Polym. Sin. 2021, 52, 723. (in Chinese) |
| [3] | (张瑶瑶, 韩彪, 周丽洁, 王明宇, 李博解, 汪连生, 朱磊, 高分子学报, 2021, 52, 723.) |
| [4] | Lu, D. N.; Liu, Z. X.; Zhang, M. L.; Wang, X. G.; Liu, Z. Biochem. Eng. J. 2006, 27, 336. |
| [5] | Pelton, R. J. Colloid Interf. Sci. 2010, 348, 673. |
| [6] | Cui, S. X.; Pang, X. C.; Zhang, S.; Yu, Y.; Ma, H. W.; Zhang, X. Langmuir 2012, 28, 5151. |
| [7] | Mackenzie, K. J.; Francis, M. B. J. Am. Chem. Soc. 2013, 135, 293. |
| [8] | Scarpa, J. S.; Mueller, D. D.; Klotz, I. M. J. Am. Chem. Soc. 1967, 89, 6024. |
| [9] | Li, Y.; Zhang, L.; Shi, Y.; Huang, J.; Yang, Y.; Ming, D. Polymers 2020, 12, 2565. |
| [10] | Hirayama, S.; Oohora, K.; Uchihashi, T.; Hayashi, T. J. Am. Chem. Soc. 2020, 142, 1822. |
| [11] | Zhang, Y. Y.; Tan, R.; Gao, M. Q.; Hao, P. B.; Yin, D. H. Green Chem. 2017, 19, 1182. |
| [12] | Keogh, R.; Blackman, L. D.; Foster, J. C.; Varlas, S.; O’Reilly, R. K. Macromol. Rapid Commun. 2020, 41, 1900599. |
| [13] | Ree, B. J.; Satoh, Y.; Jin, K. S.; Isono, T.; Kim, W. J.; Kakuchi, T.; Satoh, T.; Ree, M. NPG Asia Mater. 2017, 9, 453. |
| [14] | Baleiz?o, C.; Garcia, H. Chem. Rev. 2006, 106, 3987. |
| [15] | Lifchits, O.; Reisinger, C. M.; List, B. J. Am. Chem. Soc. 2010, 132, 10227. |
| [16] | Liao, S. H.; List, B. Angew. Chem., Int. Ed. 2010, 49, 628. |
| [17] | Song, F.; Wang, C.; Lin, W. Chem. Commun. 2011, 47, 8256. |
| [18] | Jia, Y. H.; Alothman, A. A.; Liang, R.; Li, X. Y.; Ouyang, W. Y.; Wang, X. D.; Wu, Y.; Asman, S. M.; Li, Z. R.; Gao, M.; Sun, Y. Materials 2020, 13, 4860. |
| [19] | Wurst, J. M.; Liu, G. D.; Tan, D. S. J. Am. Chem. Soc. 2011, 133, 7916. |
| [20] | Zhang, W.; Loebach, J. L.; Wilson, S. R.; Jacobsen, E. N. J. Am. Chem. Soc. 1990, 112, 2801. |
| [21] | Larrow, J. F.; Jacobsen, E. N. J. Org. Chem. 1994, 59, 1939. |
| [22] | Francesco, B.; Chiara, G.; Andrea, P.; Tomaselli, G. A.; Toscano, R. M.; Sfrazzetto, G. T. Int. J. Mol. Sci. 2016, 17, 1112. |
| [23] | Huang, J.; Xie, J. J.; Cai, J. L. Appl. Organomet. Chem. 2019, 33, 4982. |
| [24] | Baleizao, C.; Garcia, H. Chem. Rev. 2006, 106, 3987. |
| [25] | Zhao, J.; Zhang, Y.; Han, F.; Zhao, S. Carbohydr. Res. 2009, 344, 61. |
| [26] | Drozd, V. A.; Ottenbacher, R. V.; Bryliakov, K. P. Molecules 2022, 27, 2538. |
| [27] | Su, Y. T.; Yu, F.; Liu, G. X.; Huang, Z. Chin. J. Chem. 2022, 40, 2263. |
| [28] | Lin, B. J.; Zhang, X. M.; Zhou, C. Y.; Che, C. M. Org. Chem. Front. 2021, 8, 1216. |
| [29] | Zimmerman, J.; Anastas, P.; Erythropel, H.; Leitner, W. Science 2020, 367, 397. |
| [30] | Zhang, Y. Y.; Tan, R.; Zhao, G. W.; Luo, X. F.; Xing, C.; Yin, D. H. J. Catal. 2016, 335, 62. |
| [31] | Yu, K.; Gu, Z.; Ji, R.; Lou, L. L.; Ding, F.; Zhang, C.; Liu, S. J. Catal. 2007, 252, 312. |
| [32] | Zihnil, A. I. M.; Eun, B. K.; Nuraeni, L.; Insik, I.; Sung, Y. P. RSC Adv. 2017, 7, 11149. |
| [33] | Tan, R.; Yin, D.; Yu, N.; Zhao, H.; Yin, D. H. J. Catal. 2009, 263, 284. |
| [34] | Lou, L. L.; Yu, K.; Ding, F.; Peng, X. J.; Dong, M. M.; Zhang, C.; Liu, S. X. J. Catal. 2007, 249, 102. |
| [35] | Kureshy, R. I.; Khan, N. H.; Abdi, S. H. R.; Singh, S.; Ahmad, I.; Jasra, R. V.; Vyas, A. P. J. Catal. 2004, 224, 229. |
| [36] | Bell, D.; Davies, M. R.; Geen, G. R.; Mann, I. S. Synthesis 1995, 707. |
/
| 〈 |
|
〉 |