

Chinese Journal of Organic Chemistry >
Design and Synthesis of a Zn2+ Fluorescent Probe Based on Aggregation Induced Luminescence Properties
Received date: 2022-07-03
Revised date: 2022-08-01
Online published: 2022-09-02
Supported by
Shaanxi Provincial Technology Innovation Guidance Special Fund(2022QFY09-09); Shaanxi Province Key Research Program(2020GY-246); Natural Science Foundation of Shaanxi Province(S2018-JC-QN-0631); Special Scientific Research Project of Shaanxi Provincial Department of Education(17JK0017); Special Scientific Research Project of Shaanxi Provincial Department of Education(20JK0473); Key Laboratory of Se-Enriched Products Development and Quality Control, Ministry of Agriculture(Se-2020C04); National Undergraduate Training Program for Innovation and Entrepreneurship(S202111397012); Shaanxi Provincial Innovation Experiment Program for University Students(S202111397039)
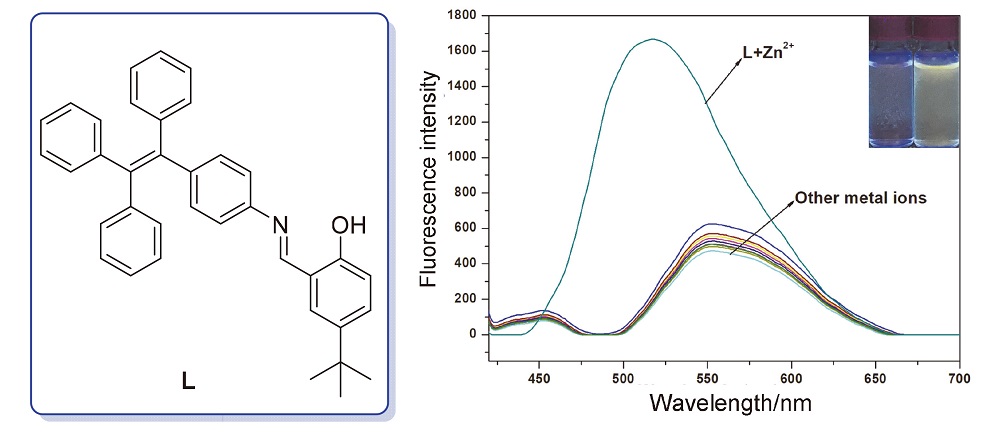
In this paper, a novel aggregation-induced emission (AIE) effect Zn2+ fluorescent probe L was constructed with aggregation-induced emission (AIE), tetraphenylethylene as the fluorescent group and 5-tert-butyl-2-hydroxybenzene as the recognition group. The structure of probe L was characterized by NMR and ESI-MS, and its fluorescence properties was studied by UV-vis and fluorescence spectroscopy. In ethanol/phosphate (PBS) (V∶V=7∶3, pH=7.4) solution, the probe L exhibited fluorescence-enhanced high selectivity and sensitivity detection of Zn2+, the detection limit was 34.1 nmol•L–1. In the range of 0~3.0×10–5 mol•L–1, the probe showed a good linear relationship to Zn2+. The obvious color change can be observed with the naked eye under UV light and natural light, which can realize the visual detection of Zn2+. The response mechanism of the probe to Zn2+ was studied by Job’s plot, ESI-MS and density functional theory (DFT) theoretical calculation. The novel probe reported in this paper can serve as a convenient tool for the analytical determination of Zn2+.
Jidong Zhang , Wanlin Yan , Wenqiang Hu , Dian Guo , Dalong Zhang , Xiaoxin Quan , Xianpan Bu , Siyu Chen . Design and Synthesis of a Zn2+ Fluorescent Probe Based on Aggregation Induced Luminescence Properties[J]. Chinese Journal of Organic Chemistry, 2023 , 43(1) : 326 -331 . DOI: 10.6023/cjoc202207004
| [1] | Maret, W. Adv. Nutr. 2013, 4, 82. |
| [2] | Xu, H.; Zhu, C.; Chen, Y.; Bai, Y.; Han, Z.; Yao, S.; Jiao, Y.; Yuan, H.; He, W.; Guo, Z. Chem. Sci. 2020, 11, 11037. |
| [3] | Plum, L. M.; Rink, L.; Haase H. Int. J. Environ. Res. Public Health 2010, 7, 1342. |
| [4] | Merriman, C.; Huang, Q.; Rutter, G. A.; Fu, D. J. Biol. Chem. 2016, 29, 26950. |
| [5] | Carter, K. P.; Young, A. M.; Palmer, A. E. Chem. Rev. 2014, 114, 4564. |
| [6] | Zhang, J.; Yan, Z.; Wang, S.; She, M.; Zhang, Z.; Cai, W.; Liu, P.; Li, J. Dyes Pigm. 2018, 150, 112. |
| [7] | Zhang, J.; Zhan, Y.; Qi, Y.; Wang, R.; Meng, L. Chin. J. Org. Chem. 2020, 40, 1847. (in Chinese) |
| [7] | (张继东, 詹妍, 齐怡, 王瑞鹏, 孟莉, 有机化学, 2020, 40, 1847.) |
| [8] | Wu, D.; Sedgwick, A. C.; Gunnlaugsson, T.; Akkaya, E. U.; Yoon, J.; James, T. D. Chem. Soc. Rev. 2017, 46, 710. |
| [9] | Nie, J.; Li, N.; Ni, Z.; Zhao, Y.; Zhang, L. Tetrahedron Lett. 2017, 58, 1980. |
| [10] | Pan, S.; Tang, H.; Song, Z.; Li, J.; Guo, Y. Chin. J. Chem. 2017, 35, 1263. |
| [11] | Cao, J., Zhao, C.; Wang, X.; Zhang, Y.; Zhu, W. Chem. Commun. 2012, 48, 9897. |
| [12] | Rai, A.; Singh, A. K.; Sonkar, A. K.; Tripathi, K.; Mishra, L. Spectrochim. Acta, Part A 2019, 216, 1. |
| [13] | Burdette, S. C.; Walkup, G. K.; Spingler, B.; Tsien, R. Y.; Lippard, S. J. J. Am. Chem. Soc. 2001, 123, 7831. |
| [14] | Luo, J.; Xie, Z.; Lam, J. W.; Cheng, L.; Chen, H.; Qiu, C.; Tang, B. Z. Chem. Commun. 2001, 18, 1740. |
| [15] | Tang, A.; Yin, Y.; Chen, Z.; Fan, C.; Liu, G.; Pu, S. Tetrahedron 2019, 75, 130489. |
| [16] | He, X.; Wang, X.; Zhang, L.; Fang, G.; Liu, J.; Wang, S. Sens. Actuators, B Chem. 2018, 271, 289. |
| [17] | Sun, H.; Jiang, Y.; Nie, J.; Wei, J.; Miao, B.; Zhao, Y.; Ni, Z. Mater. Chem. Front. 2021, 5, 347. |
| [18] | Jiang, S.; Chen, S.; Wang, Z.; Guo, H.; Yang, F. Sens. Actuators, B Chem. 2020, 308, 127734. |
| [19] | Li, Z.; Huo, Y.; Yang, X.; Ji, S. Chin. J. Org. Chem. 2016, 36, 2317. (in Chinese) |
| [19] | (李宗植, 霍延平, 阳香华, 籍少敏, 有机化学, 2016, 36, 2317.) |
| [20] | Zhou, Z.; Yan, X.; Saha, M. L.; Zhang, M.; Wang, M.; Li, X.; Stang, P. J. J. Am. Chem. Soc. 2016, 138, 13131. |
| [21] | Yan, X.; Wang, H.; Hauke, C. E.; Cook, T. R.; Wang, M.; Saha, M. L.; Stang, P. J. J. Am. Chem. Soc. 2015, 137, 15276. |
| [22] | Hong, Y.; Chen, S.; Leung, C. W. T.; Lam, J. W. Y.; Liu, J.; Tseng, N. W.; Tang, B. Z. ACS Appl. Mater. Interfaces 2011, 3, 3411. |
| [23] | Yang, Y.; Gao, C. Y.; Zhang, N.; Dong, D. Sens. Actuators, B Chem. 2016, 222, 741. |
| [24] | Mu, C.; Zhang, Z.; Hou, Y.; Liu, H.; Ma, L.; Li, X.; Zhang, M. Angew. Chem., Int. Ed. 2021, 133, 12401. |
| [25] | Wang, L.; Li, Y.; You, X.; Xu, K.; Feng, Q.; Wang, J., Hou, H.; Liu, Y.; Li, K. J. Mater. Chem. C 2017, 5, 65. |
| [26] | Jia, J.; Zhao H. Org. Electron. 2019, 73, 55. |
| [27] | Sedgwick, A. C.; Wu, L.; Han, H. H.; Bull, S. D.; He, X. P.; James, T. D.; Yoon, J.; Sessler, J. L.; Tang, B.Z.; Tian, H. Chem. Soc. Rev. 2018, 47, 8842. |
/
| 〈 |
|
〉 |