

Chinese Journal of Organic Chemistry >
Intermolecular Halogen and Hydrogen Bonding-Controlled Self-Assembly of Network Structures
Received date: 2022-07-04
Revised date: 2022-09-04
Online published: 2022-10-10
Supported by
Key Scientific Research Projects of Colleges and Universities of Henan Province(21A150045); Start-up Foundation for Scientific Research of Newly Recruited PhD of Shangqiu Normal University(700200); Start-up Foundation for Scientific Research of Newly Recruited PhD of Shangqiu Normal University(700205)
Two kinds of tetra-dentate halogen bonding donors were synthesized by introducing tetrafluoroiodide benzene into tetraphenylene methane and tetraphenyl porphyrin modules. 3,3',5,5'-Tetramethyl-4,4'-bipyridine (TMBP) was synthesized as halogen bonding acceptor. A type of supramolecular network structure was constructed by self-assembly of tetraphenylmethane-centered halogen donor and TMBP in solid phase based on intermolecular I…N halogen bonds and H…N hydrogen bonds. The crystal structure shows that one tetrahedral halogen bonding donor binds four TMBP molecules through two sets of I…N halogen bonds and two sets of H…N hydrogen bonds, correspondingly, one TMBP molecule binds two tetrahedral molecules through one set of I…N halogen bonds and one set of H…N hydrogen bonds to form a monolayer network structure distributed with square structures with grid width of 2.37 nm. And further stacking is controlled by other hydrogen and halogen bonds between layers. The crystal data of tetradentate halogen bonding donor molecules of porphyrins show that donor molecules assemble themselves in plane controlled by more complex weak intermolecular C-I…π and H…F interaction, and further stacking is controlled by π-π stacking between layers.
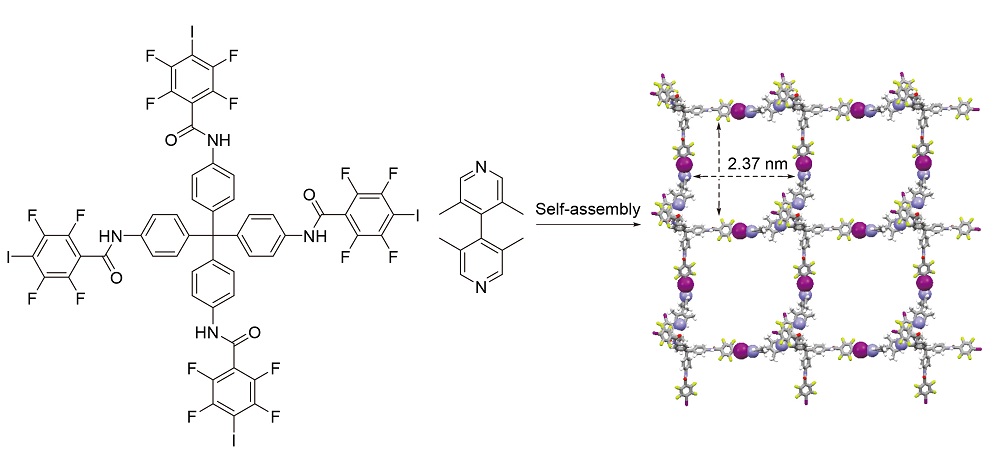
Fen Li , Chuanzhi Liu , Zhiyuan Hu , Panpan Luo , Rongzheng Cui , Yanke Huang , Xinming Liu , Lantao Liu , Wei Wu . Intermolecular Halogen and Hydrogen Bonding-Controlled Self-Assembly of Network Structures[J]. Chinese Journal of Organic Chemistry, 2023 , 43(2) : 705 -711 . DOI: 10.6023/cjoc202207007
| [1] | (a) Calhorda, M. J. Chem. Commun. 2000, 801. |
| [1] | (b) Steiner, T. Angew. Chem., Int. Ed. 2002, 41, 48. |
| [1] | (c) Cavallo, G.; Metrangolo, P.; Milani, R.; Pilati, T.; Priimagi, A.; Resnati, G.; Terraneo, G. Chem. Rev. 2016, 116, 2478. |
| [1] | (d) Ding, X.-H.; Chang, Y.-Z.; Ou, C.-Q.; Lin, J.-Y.; Xie, L.-H.; Huang, W. Nat. Sci. Rev. 2020, 7, 1906. |
| [2] | (a) Metrangolo, P.; Meyer, F.; Pilati, T.; Proserpio, D. M.; Resnati, G. Chem.-Eur. J. 2007, 13, 5765. |
| [2] | (b) Gong, G.-F.; Lv, S.-H.; Han, J.-X.; Xie, F.; Li, Q.; Xia, N.; Zeng, W.; Chen, Y.; Wang, L.; Wang, J.-K.; Chen, S.-G. Angew. Chem., Int. Ed. 2021, 60, 14831. |
| [2] | (c) Zheng, Q.-N.; Liu, X.-H.; Chen, T.; Yan, H.-J.; Cook, T.; Wang, D.; Stang, P. J.; Wan, L.-J. J. Am. Chem. Soc. 2015, 137, 6128. |
| [2] | (d) Pfrunder, M. C.; Brock, A. J.; Brown, J. J.; Grosjean, A.; Ward, J.; McMurtrie, J. C.; Clegg, J. K. Chem. Commun. 2018, 54, 3974 |
| [3] | (a) Jungbauer, S. H.; Bulfield, D.; Kniep, F.; Lehmann, C. W.; Herdtweck, E.; Huber, S. M. J. Am. Chem. Soc. 2014, 136, 16740. |
| [3] | (b) Dumele, O.; Schreib, B.; Warzok, U.; Trapp, N.; Schalley, C. A.; Diederich, F. Angew. Chem., Int. Ed. 2016, 55, 1. |
| [4] | Jentzsch, A. V.; Matile, S. Top. Curr. Chem. 2015, 358, 205. |
| [5] | (a) Biswal, B. P.; Valligatla, S.; Wang, M-C.; Banerjee, T.; Saad, N. A.; Mariserla, B. M. K.; Chandrasekhar, N.; Becker, D.; Addicoat, M.; Senkovska, I.; Berger, R.; Rao, D. N.; Kaskel, S.; Feng, X.-L. Angew. Chem., Int. Ed. 2019, 58, 6896. |
| [5] | (b) Meng, Y.; Luo, Y.; Shi, J.-L.; Ding, H.-M.; Lang, X.-J.; Chen, W.; Zheng, A.-M.; Sun, J.-L.; Wang, C. Angew. Chem., Int. Ed. 2020, 59, 3624. |
| [5] | (c) Yang, B.; Yu, S.-B.; Zhang, P.-Q.; Wang, Z.-K.; Qi, Q.-Y.; Wang, X.-Q.; Xu, X.-H.; Yang, H.-B.; Wu, Z.-Q.; Liu, Y.; Ma, D.; Li, Z.-T. Angew. Chem., Int. Ed. 2021, 60, 26268. |
| [5] | (d) Yang, B.; Zhang, X.-D.; Li, J.; Tian, J.; Wu, Y.-P.; Yu, F.-X.; Wang, R.; Wang, H.; Zhang, D.-W.; Liu, Y.; Zhou, L.; Li, Z.-T. CCS Chem. 2019, 1, 156. |
| [5] | (e) Yu, S.-B.; Lin, F.; Tian, J.; Yu, J.; Zhang, D.-W.; Li, Z.-T. Chem. Soc. Rev. 2022, 51, 434. |
| [6] | (a) Muniappan, S.; Lipstmana, S.; Goldberg, I. Chem. Commun. 2008, 1777. |
| [6] | (b) Syssa-Magalé, J. L.; Boubekeur, K.; Leroy, J.; Chamoreau, L. M.; Favec, C.; Sch?llhorn, B. CrystEngComm 2014, 16, 10380. |
| [6] | (c) Spilfogel, T. S.; Titi, H. M.; Fri??i?, T. Cryst. Growth Des. 2021, 21, 1810. |
| [7] | Gunawardana, C. A.; Dakovi?b, M.; Aaker?y, C. B. Chem. Commun. 2018, 54, 607. |
| [8] | Dumele, O.; Trapp, N.; Diederich, F. Angew. Chem., Int. Ed. 2015, 54, 12339. |
| [9] | (a) Liu, C.-Z.; Koppireddi, S.; Wang, H.; Zhang, D.-W.; Li, Z.-T. Angew. Chem., Int. Ed. 2019, 58, 226. |
| [9] | (b) Liu, C.-Z.; Koppireddi, S.; Wang, H.; Zhang, D.-W.; Li, Z.-T. Chin. Chem. Lett. 2019, 30, 953. |
| [9] | (c) Koppireddi, S.; Liu, C.-Z.; Wang, H.; Zhang, D.-W.; Li, Z.-T. CrystEngComm 2019, 21, 2626. |
| [10] | Li, C.; Ren, S.-F.; Hou, J.-L.; Yi, H.-P.; Zhu, S.-Z.; Jiang, X.-K.; Li, Z.-T. Angew. Chem., Int. Ed. 2005, 44, 5725. |
| [11] | Li, Z.; Twieg, R. J. Chem.-Eur. J. 2015, 21, 15534. |
| [12] | Dang, Q. M.; Gilmore, S. T.; Lalwani, K.; Conk, R. J.; Simpson, J. H.; Leopold, M. C. Langmuir 2022, 38, 4747. |
| [13] | Rang, A.; Engeser, M.; Maier, N. M.; Martin Nieger, M.; Lindner, W.; Schalley, C. A. Chem.-Eur. J. 2008, 14, 3855. |
/
| 〈 |
|
〉 |