

Chinese Journal of Organic Chemistry >
Ring Opening Amination of gem-Difluorocyclopropanes with Cyclobutanone Hydrazones
Received date: 2022-08-24
Revised date: 2022-11-02
Online published: 2022-12-12
Supported by
National Natural Science Foundation of China(22201145); Scientific Research Foundation of High-level Talents of Nantong University(03083031); Universities Natural Science Research Project of Jiangsu Province(21KJB150015); Large Instruments Open Foundation of Nantong University(KFJN2215)
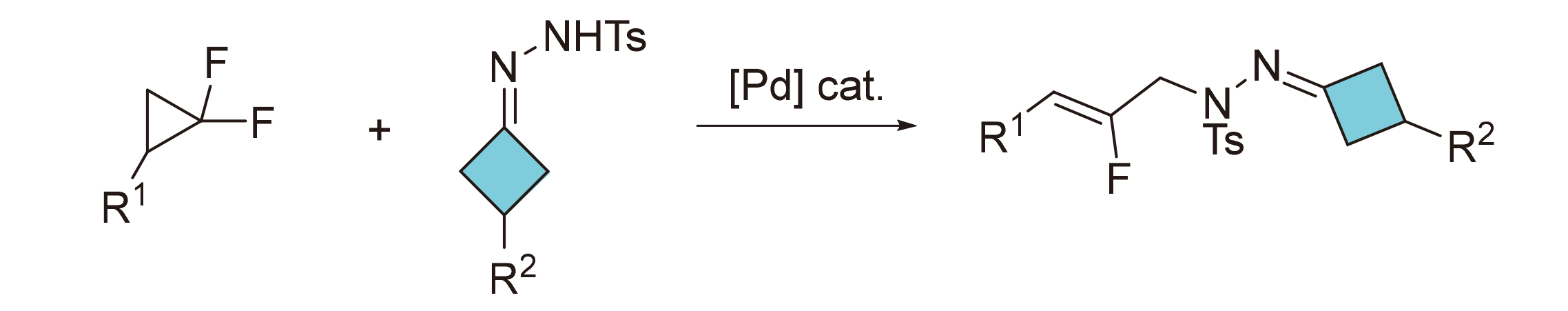
N-Fluoroallyl hydrazones with four-ring structure were effectively synthesized using cyclobutanone hydrazones and gem-difluorocyclopropanes as raw materials through palladium catalyzed ring opening of difluorocyclopropanes and nucleophilic capture process of nitrogen nucleophile. This protocol shows broad substrate scope and good functional group tolerance, which can be applied for gram-scale preparation.
Zhipeng Liang , Hao Ye , Haibin Zhang , Guomin Jiang , Xinxing Wu . Ring Opening Amination of gem-Difluorocyclopropanes with Cyclobutanone Hydrazones[J]. Chinese Journal of Organic Chemistry, 2023 , 43(4) : 1483 -1491 . DOI: 10.6023/cjoc202208031
| [1] | (a) Hancock, E. N.; Wiest, J. M.; Brown, M. K. Nat. Prod. Rep. 2019, 36, 1383. |
| [1] | (b) Li, J.; Gao, K.; Bian, M.; Ding, H. Org. Chem. Front. 2020, 7, 136. |
| [1] | (c) Lukin, K.; Kishore, V.; Gordon, T. Org. Process Res. Dev. 2013, 17, 666. |
| [1] | (d) Beck, J. C.; Lacker, C. R.; Chapman, L. M.; Reisman, S. E. Chem. Sci. 2019, 10, 2315. |
| [1] | (e) Cui, Y.; Xia, D. Chin. J. Org. Chem. 2021, 41, 907. (in Chinese) |
| [1] | (崔英翠, 夏德斌, 有机化学, 2021, 41, 907.) |
| [2] | (a) Biletskyi, B.; Colonna, P.; Masson, K.; Parrain, J.-L.; Commeiras, L.; Chouraqui, G. Chem. Soc. Rev. 2021, 50, 7513. |
| [2] | (b) Murakami, M.; Ishida, N. Chem. Rev. 2021, 121, 264. |
| [3] | For selected examples: (a) Wang, Y.; Hu, M.; Ding, L.; Wang, Y.; Wang, X.-N.; Chang, J. Org. Lett. 2022, 24, 5056. |
| [3] | (b) Li, H.; Hsung, R. P.; DeKorver, K. A.; Wei, Y. Org. Lett. 2010, 12, 37806. |
| [3] | (c) K?lmel, D. K.; Ratnayake, A. S.; Flanagan, M. E.; Tsai, M.-H.; Duan, C.; Song, C. Org. Lett. 2020, 22, 2908. |
| [3] | (d) Luzung, M. R.; Mauleón, P.; Toste, F. D. J. Am. Chem. Soc. 2007, 129, 12402. |
| [3] | (e) Okamoto, K.; Shimbayashi, T.; Tamura, E.; Ohe, K. Org. Lett. 2015, 17, 5843. |
| [3] | (f) Oderinde, M. S.; Ramirez, A.; Murali Dhar, T. G.; L. A. M.; Jorge, C.; Aulakh, D.; Sandhu, B.; Pawluczyk, J.; Sarjeant, A. A.; Meanwell, N. A.; Mathur, A.; Kempson, J. J. Org. Chem. 2021, 86, 1730. |
| [3] | (g) Conner, M. L.; Kevin Brown, M. ; J. Org. Chem. 2016, 81, 8050. |
| [3] | (h) Zhu, H.-T.; Ke, S.; Zhou, N.-N.; Fan, M.-J.; Yang,, D.-S. Org. Lett. 2016, 18, 4554. |
| [3] | (i) Sakai, K.; Kochi, T.; Kakiuchi, F. Org. Lett. 2013, 15, 1024. |
| [4] | For selected examples: (a) Oishi, S.; Kamitani, H.; Kodama, E.; Matsuoka, M.; Fujii, N. Org. Biomol. Chem. 2009, 7, 2872. |
| [4] | (b) Fujita, T.; Fuchibe, K.; Ichikawa, J. Angew. Chem., Int. Ed. 2019, 58, 390. |
| [4] | (c) Osada, S.; Sano, S.; Ueyama, M.; Chuman, Y.; Kodama, H.; Sakaguchi, K. Bioorg. Med. Chem. 2010, 18, 605. |
| [4] | (d) Zhang, X.; Cao, S. Tetrahedron Lett. 2017, 58, 375. |
| [4] | (e) Bartlett, P. A.; Otake, A. J. Org. Chem. 1995, 60, 3107. |
| [5] | For selected examples: (a) Dolbier, W. R.; Battiste, M. A. Chem. Rev. 2003, 103, 1071. |
| [5] | (b) Wenz, J.; Rettenmeier, C. A.; Wadepohl, H.; Gade, L. H. Chem. Commun. 2016, 52, 202. |
| [5] | (c) Song, X.; Xu, C.; Wang, M. Tetrahedron Lett. 2017, 58, 1806. |
| [5] | (d) Song, X.; Xu, C.; Du, D.; Zhao, Z.; Zhu, D.; Wang, M. Org. Lett. 2017, 19, 6542. |
| [5] | (e) Fu, Z.; Zhu, J.; Guo, S.; Lin, A. Chem. Commun. 2021, 57, 1262. |
| [5] | (f) Jiang, Z.-T.; Huang, J.; Zeng, Y.; Hu, F.; Xia, Y. Angew. Chem., Int. Ed. 2021, 60, 10626 |
| [6] | For selected examples: (a) Ahmed, E.-A. M. A.; Suliman, A. M. Y.; Gong, T.-J.; Fu, Y. Org. Lett. 2019, 21, 5645. |
| [6] | (b) Ahmed, E.-A. M. A.; Suliman, A. M. Y.; Gong, T.-J.; Fu, Y. Org. Lett. 2020, 22, 1414. |
| [7] | Xu, J.; Ahmed, E.-A.; Xiao, B.; Lu, Q.-Q.; Wang, Y.-L.; Yu, C.-G.; Fu, Y. Angew. Chem., Int. Ed. 2015, 54, 8231. |
| [8] | Lv, L.; Li, C.-J. Angew. Chem., Int. Ed. 2021, 60, 13098. |
| [9] | Xiong, B.; Chen, X.; Liu, J.; Zhang, X.; Xia, Y.; Lian, Z. ACS Catal. 2021, 11, 11960. |
| [10] | (a) Ning, X.; Chen, Y.; Hu, F.; Xia, Y. Org. Lett. 2021, 23, 8348. |
| [10] | (b) Ye, H.; Ren, T.; Wu, X. Chin. J. Org. Chem. 2021, 41, 4338. (in Chinese) |
| [10] | (叶浩, 任婷婷, 吴新星, 有机化学, 2021, 41, 4338.) |
/
| 〈 |
|
〉 |