

Chinese Journal of Organic Chemistry >
Design and Synthesis of N-Tetrazole and N-Oxadiazole Heterocyclic Derivatives of Sinomenine
Received date: 2022-11-08
Revised date: 2023-01-25
Online published: 2023-02-15
Supported by
Starting Grants of Anhui Jianzhu University(2020QDZ03); Foundation of Anhui Jianzhu University(JZ202219); Foundation of Anhui Jianzhu University(JZ202249)
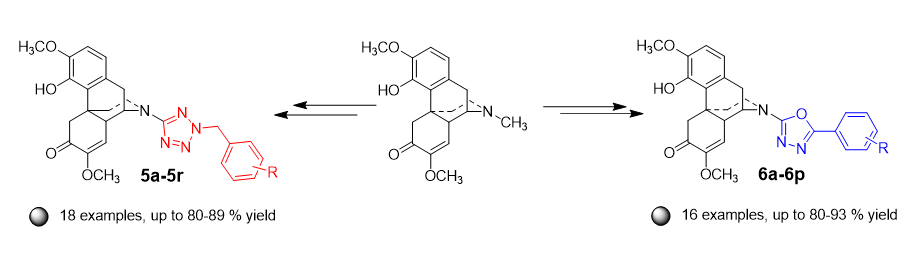
Two new synthesis methods were established, N-tetrazole and N-1,3,4-oxadiazole substituted heterocyclic derivatives of sinomenine were constructed on the N atom of D-ring, and 34 new sinomenine heterocyclic derivatives were synthesized. N-CN sinomenine at 17-position was generated by substituted reaction between cyanogen bromide and sinomenine, and then N-tetrazole sinomenine was achieved by 1,3-dipolar cycloaddition reaction with NaN3. The reaction of tetrazole with benzyl chloride produced N-tetrazole heterocyclic derivatives of sinomenine. N-1,3,4-Oxadiazole heterocyclic derivatives of sinomenine were obtained by acylation reaction and the Huisigen rearrangement reaction of tetrazole with acyl chloride reagents. These methods with mild condition gave good to excellent yields. This method provides new methods for structural modification of natural products, the synthesized compounds expand the library of sinomenine derivatives, and this work provides a basis study for drug molecular design and potential activity study.
Key words: sinomenine; tetrazole; oxadiazole; derivative
Xuedan Wu , Ruixiang Xu , Xiaolong Fang , Kehua Zhang , Jie Jin . Design and Synthesis of N-Tetrazole and N-Oxadiazole Heterocyclic Derivatives of Sinomenine[J]. Chinese Journal of Organic Chemistry, 2023 , 43(7) : 2506 -2518 . DOI: 10.6023/cjoc202211009
| [1] | (a) Wang, Y.; Fang, Y. F.; Huang, W. H.; Zhou, X.; Wang, M. H.; Zhong, B.; Peng, D. Z. J. Ethnopharmacol. 2005, 98, 37. |
| [1] | (b) Cheng, Y.; Zhang, J. B.; Hou, W. P.; Wang, D. H.; Li, F. R.; Zhang, Y. Q.; Yuan, F. H. Int. Immunopharmacol. 2009, 9, 894. |
| [1] | (c) Zhou, Y.; Liu, H. Y.; Song, J.; Cao, L.; Tang, L. M.; Qi, C. J. Mol. Med. Rep. 2018, 18, 3691. |
| [1] | (d) Dong, B. Med. Sci. Monit. 2018, 24, 3631. |
| [2] | Chen, S. P.; Zhou, Y. Q.; Sun, J.; Cao, F.; Braun, C.; Luo, F.; Ye, D. W.; Tian, Y. K. Mol. Pain 2018, 14, 1. |
| [3] | Wang, Y.; Ma, D. L.; Jie, Y.; Wu, Y. Y.; Pan, Z. Q. Immunotherapy 2012, 4, 581. |
| [4] | (a) Liu, Z.; Zheng, J. F.; Yang, L.Q.; Yi, L.; Hu, B. Acta Physiol. Sin. 2007, 59, 285 (in Chinese). |
| [4] | (刘珍, 郑济芳, 杨露青, 易岚, 胡弼, 生理学报, 2007, 59, 285.) |
| [4] | (b) Zhu, W. L.; Zhang, Y. N.; Huang, Y. J.; Lu, L. Int. Rev. Neurobiol. 2017, 135, 279. |
| [5] | (a) Qian, L.; Xu, Z. L.; Zhang, W.; Wilson, B.; Hong, J. S.; Flood, P. M. J. Neuroinflammation 2007, 4, 1. |
| [5] | (b) Yang, Y. Q.; Wang, H. D.; Li, L. W.; Li, X.; Wang, Q.; Ding, H.; Wang, X. L.; Ye, Z. N.; Wu, L. Y.; Zhang, M. L.; Pan, H. Front. Neurosci. 2016, 10, 580. |
| [6] | (a) Jiang, S. L.; Gao, Y. B.; Hou, W.; Liu, R.; Qi, X.; Xu, X.; Li, J.; Bao, H. G.; Hua, B. J. Oncol. Lett. 2016, 12, 1380. |
| [6] | (b) Song, L. Q.; Liu, D.; Zhao, Y.; He, J. J.; Kang, H. F.; Dai, Z. J.; Wang, X. J.; Zhang, S. Q.; Zan, Y. Biochem. Biophys. Res. Commun. 2015, 464, 705. |
| [6] | (c) Yang, H. B.; Yin, P. H.; Shi, Z.; Ma, Y. C.; Zhao, C. G.; Zheng, J. P.; Chen, T. Oncol. Lett. 2016, 11, 411. |
| [7] | (a) Luo, D. M.S. Thesis, Changsha University of Science & Technology, Changsha, 2013 (in Chinese). |
| [7] | (罗丹, 硕士论文,长沙理工大学,长沙, 2013.) |
| [7] | (b) Yang, F.; Guan, Z. J.; Cai, X. Y.; Wu, Q. Y.; Meng, Q. G. Acad. J. Second Mil. Med. Univ. 2015, 36, 413 (in Chinese). |
| [7] | (杨帆, 管忠俊, 柴晓云, 吴秋业, 孟庆国, 第二军医大学学报, 2015, 36, 413.) |
| [8] | Xie, G. Y.; Zhang, Q.; Huang, Y. Y.; Zheng, H.; Ding, S. J. South-Cent. Univ. Natl. (Nat. Sci. Ed.) 2020, 39, 221 (in Chinese). |
| [8] | (谢光勇, 张倩, 黄业迎, 郑浩, 丁爽, 中南民族大学学报, 2020, 39, 221.) |
| [9] | Kazemnejadi, M.; Mahmoudi, B.; Sharafi, Z.; Nasseri, M. A.; Allahresani, A.; Esmaeilpour, M. Appl. Organomet. Chem. 2020, 34, 1. |
| [10] | (a) Demko, Z. P.; Sharpless, K. B. J. Org. Chem. 2001, 66, 7945. |
| [10] | (b) Himo, F.; Demko, Z. P.; Noodleman, L.; Sharpless, K. B. J. Am. Chem. Soc. 2003, 125, 9983. |
| [10] | (c) Jin, T.; Kitahara, F.; Kamijo, S.; Yamamoto, Y. Tetrahedron Lett. 2008, 49, 2824. |
| [10] | (d) Das, B.; Reddy, C.; Kumar, D. Synlett 2010, 391. |
| [10] | (e) Harusawa, S.; Yoneyama, H.; Usami, Y.; Komeda, S. Synthesis 2013, 45, 1051. |
| [10] | (f) Myznikov, L.; Vorona, S.; Artamonova, T.; Zevatskii, Y. Synthesis 2014, 46, 0781. |
| [10] | (g) Coca, A.; Turek, E.; Feinn, L. Synth. Commun. 2015, 45, 218. |
| [10] | (h) Coca, A.; Feinn, L.; Dudley, J. Synth. Commun. 2015, 45, 1023. |
| [10] | (i) Myznikov, L. V.; Vorona, S. V.; Artamonova, T. V. Russ. J. Gen. Chem. 2017, 87, 731. |
| [10] | (j) Witkowski, T. G.; Sebastiao, E.; Gabidullin, B.; Hu, A.; Zhang, F.; Murugesu, M. ACS Appl. Energy Mater. 2018, 1, 589. |
| [11] | (a) Seldes, A. M.; Accorso, N. D.; Souto, M. F.; Alho, M. M.; Arabehety, C. G. J. Mass Spectrom. 2001, 36, 1069. |
| [11] | (b) Reichart, B.; Kappe, C. O. Tetrahedron Lett. 2012, 53, 952. |
| [11] | (c) Jia, Y. H.; Yang, K. X.; Chen, S. L. J. Phys. Chem. A 2017, 122, 8. |
| [11] | (d) Obydennov, D. L.; SimbIR ATRtseva, A. E.; Piksin, S. E.; Sosnovskikh, V. Y. ACS Omega 2020, 5, 33406. |
/
| 〈 |
|
〉 |