

Chinese Journal of Organic Chemistry >
Recent Progress in Selective C-F Bond Activation of Trifluoromethyl Alkenes
Received date: 2022-10-17
Revised date: 2022-11-03
Online published: 2022-11-04
Selective C-F bond activation of the trifluoromethyl group is an important approach for the synthesis of gem-difluoro compounds. Given the importance of trifluoromethyl alkenes in synthetic chemistry as well as their diversity in selectivity, the research on selective C-F bond activation of these compounds has received considerable interest. To this end, The research progress is summarized, including nucleophilic addition/defluorination (SN2' type), acid-promoted defluorinative Friedel-Crafts arylation, (formal) ipso-defluorinative functionalization, and transition-metal-catalyzed migratory insertion/ defluorination.
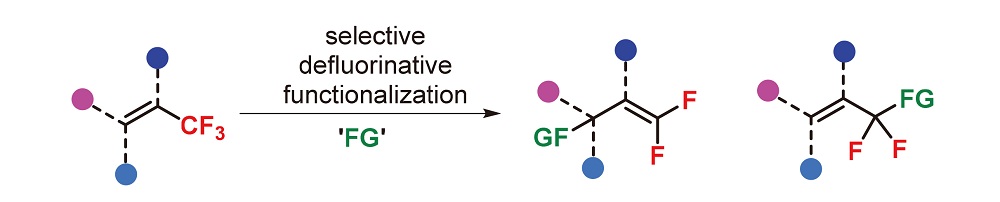
Key words: trifluoromethyl alkene; C-F bond activation; synthetic method
Shuyong Song , Senmiao Xu . Recent Progress in Selective C-F Bond Activation of Trifluoromethyl Alkenes[J]. Chinese Journal of Organic Chemistry, 2023 , 43(2) : 411 -425 . DOI: 10.6023/cjoc202210016
| [1] | O'Hagan, D. Chem. Soc. Rev. 2008, 37, 308. |
| [2] | Jaroschik, F. Chem. Eur. J. 2018, 24, 14572. |
| [3] | (a) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320. |
| [3] | (b) Dhara, M. G.; Banerjee, S. Prog. Polym. Sci. 2010, 35, 1022. |
| [3] | (c) Wozniak, A. I.; Yegorov, A. S.; Ivanov, V. S.; Igumnov, S. M.; Tcarkova, K. V. J. Fluorine Chem. 2015, 180, 45. |
| [4] | (a) Charpentier, J.; Fruh, N.; Togni, A. Chem. Rev. 2015, 115, 650. |
| [4] | (b) Ni, C.; Hu, M.; Hu, J. Chem. Rev. 2015, 115, 765. |
| [4] | (c) Liang, T.; Neumann, C. N.; Ritter, T. Angew. Chem. Int. Ed. 2013, 52, 8214. |
| [5] | (a) Amii, H.; Uneyama, K. Chem. Rev. 2009, 109, 2119. |
| [5] | (b) Chelucci, G. Chem. Rev. 2012, 112, 1344. |
| [6] | Luo, Y. R. Comprehensive Handbook of Chemical Bond Energies, CRC Press, Boca Raton, FL, 2007, pp. 211-217. |
| [7] | Uneyama, K. Organofluorine chemistry, John wiley & sons, 2008. |
| [8] | (a) Xiao, T.; Li, L.; Zhou, L. J. Org. Chem. 2016, 81, 7908. |
| [8] | (b) Lang, S. B.; Wiles, R. J.; Kelly, C. B.; Molander, G. A. Angew. Chem. Int. Ed. 2017, 56, 15073. |
| [8] | (c) Wu, L. H.; Cheng, J. K.; Shen, L.; Shen, Z. L.; Loh, T. P. Adv. Synth. Catal. 2018, 360, 3894. |
| [9] | (a) Zhang, X. X.; Cao, S. Tetrahedron Lett. 2017, 58, 375. |
| [9] | (b) Pan, Y.; Qiu, J.; Silverman, R. B. J. Med. Chem. 2003, 46, 5292. |
| [10] | (a) Ichikawa, J. J. Fluorine Chem. 2000, 105, 257. |
| [10] | (b) Shen, Q.; Huang, Y.-G.; Liu, C.; Xiao, J.-G.; Chen, Q.-Y.; Guo, Y. J. Fluorine Chem. 2015, 179, 14. |
| [11] | Wang, Z.; Sun, Y.; Shen, L. Y.; Yang, W. C.; Meng, F.; Li, P. Org. Chem. Front. 2022, 9, 853. |
| [12] | (a) Bégué, J. P.; Bonnet-Delpon, D.; Rock, M. H. Tetrahedron Lett. 1995, 36, 5003. |
| [12] | (b) Funabiki, K.; Sawa, K.; Shibata, K.; Matsui, M. Synlett 2002, 1134. |
| [12] | (c) Bégué, J. P.; Bonnet-Delpon, D.; Rock, M. H. J. Chem. Soc. Perkin Trans. 1 1996, 12, 1409. |
| [12] | (d) Kendrick, D. A.; Kolb, M. J. Fluorine Chem. 1989, 45, 265. |
| [12] | (e) Coates, G.; Tan, H. Y.; Kalff, C.; White, A. J.; Crimmin, M. R. Angew. Chem. Int. Ed. 2019, 58, 12514. |
| [13] | Wang, X. L.; Wang, C. Y.; Bolm, C. Org. Lett. 2022, 24, 7461. |
| [14] | Xing, W. L.; Wang, J. X.; Fu, M. C.; Fu, Y. Chin. J. Chem. 2022, 40, 323. |
| [15] | Fujita, T.; Ikeda, M.; Hattori, M.; Sakoda, K.; Ichikawa, J. Synthesis 2014, 46, 1493. |
| [16] | Fuchibe, K.; Takahashi, M.; Ichikawa, J. Angew. Chem. Int. Ed. 2012, 51, 12059. |
| [17] | Yang, J. R.; Mao, A.; Yue, Z. T.; Zhu, W. X.; Luo, X. W.; Zhu, C. W.; Xiao, Y. J.; Zhang, J. L. Chem. Commun. 2015, 51, 8326. |
| [18] | (a) Uneyama, K. Organofluorine Chemistry, Blackwell, Oxford, 2006. |
| [18] | (b) Smart, B. E. In Organofluorine Chemistry: Principles and Commercial Applications, Eds.: Banks, R. E.; Smart, B. E.; Tatlow, J. C., Plenum Press, New York, 1994, p. 57. |
| [19] | Fuchibe, K.; Hatta, H.; Oh, K.; Oki, R.; Ichikawa, J. Angew. Chem. Int. Ed. 2017, 56, 5890. |
| [20] | Zeng, H.; Zhu, C. L.; Jiang, H. L. Org. Lett. 2019, 21, 1130. |
| [21] | (a) Zhang, L. J.; Zhang, W.; Liu, J.; Hu, J. B. J. Org. Chem. 2009, 74, 2850. |
| [21] | (b) Haufe, G.; Suzuki, S.; Yasui, H.; Terada, C.; Kitayama, T.; Shiro, M.; Shibata, N. Angew. Chem. Int. Ed. 2012, 51, 12275. |
| [21] | (c) Champagne, P. A.; Pomarole, J.; The?rien, M. E?.; Benhassine, Y.; Beaulieu, S.; Legault, C. Y.; Paquin, J. F. Org. Lett. 2013, 15, 2210. |
| [21] | (d) Choi, C.; Nuhant, P.; Mousseau, J. J.; Yang, X.; Gerstenberger, B. S.; Williams, J. M.; Wright, S. W. Org. Lett. 2016, 18, 5748. |
| [22] | Zeng, H.; Cai, Y. Y.; Jiang, H. F.; Zhu, C. L. Org. Lett. 2021, 23, 66. |
| [23] | Phillips, N. A.; Coates, G. J.; White, A. J.; Crimmin, M. R. Chem. Eur. J. 2020, 26, 5365. |
| [24] | (a) Xie, L.-G.; Wang, Z.-X. Angew. Chem. Int. Ed. 2011, 50, 4901. |
| [24] | (b) Maity, P.; Shacklady-McAtee, D. M.; Yap, G. P.; Sirianni, E. R.; Watson, M. P. J. Am. Chem. Soc. 2013, 135, 280. |
| [24] | (c) Moragas, T.; Gaydou, M.; Martin, R. Angew. Chem. Int. Ed. 2016, 55, 5053. |
| [24] | (d) Wang, D. Y.; Yang, Z. K.; Wang, C.; Zhang, A.; Uchiyama, M. Angew. Chem. Int. Ed. 2018, 57, 3641. |
| [24] | (e) Yi, Y. Q. Q.; Yang, W. C.; Zhai, D. D.; Zhang, X. Y.; Li, S.-Q.; Guan, B.-T. Chem. Commun. 2016, 52, 10894. |
| [25] | Tang, L. L.; Liu, Z. Y.; She, W. Z.; Feng, C. Chem. Sci. 2019, 10, 8701. |
| [26] | Zhu, C.; Sun, M. M.; Chen, K.; Liu, H. D.; Feng, C. Angew. Chem. Int. Ed. 2021, 60, 20237. |
| [27] | Yan, S. S.; Wu, D. S.; Ye, J. H.; Gong, L.; Zeng, X.; Ran, C. K.; Gui, Y. Y.; Li, J.; Yu, D. G. ACS Catal. 2019, 9, 6987. |
| [28] | Ichikawa, J.; Nadano, R.; Ito, N. Chem. Commun. 2006, 42, 4425. |
| [29] | Miura, T.; Ito, Y.; Murakami, M. Chem. Lett. 2008, 37, 1006. |
| [30] | Huang, Y.; Hayashi, T. J. Am. Chem. Soc. 2016, 138, 12340. |
| [31] | Jang, Y. J.; Rose, D.; Mirabi, B.; Lautens, M. Angew. Chem. Int. Ed. 2018, 57, 16147. |
| [32] | Ichitsuka, T.; Fujita, T.; Arita, T.; Ichikawa, J. Angew. Chem. Int. Ed. 2014, 53, 7564. |
| [33] | Ichitsuka, T.; Fujita, T.; Ichikawa, J. ACS Catal. 2015, 5, 5947. |
| [34] | Zhang, X. M.; Liu, Y. X.; Chen, G.; Pei, G.; Bi, S. W. Organometallics 2017, 36, 3739. |
| [35] | Zhu, C.; Liu, Z. Y.; Tang, L. N.; Zhang, H.; Zhang, Y. F.; Walsh, P.; Feng, C. Nat. Commun. 2020, 11, 4860. |
| [36] | Qiu, J.; Wang, C. C.; Zhou, L.; Lou, Y. X.; Yang, K.; Song, Q. L. Org. Lett. 2022, 24, 2446. |
| [37] | Dai, W. P.; Lin, Y. Y.; Wan, Y.; Cao, S. Org. Chem. Front. 2018, 5, 55. |
| [38] | Wang, M. Y.; Pu, X. H.; Zhao, Y. F.; Wang, P. P.; Li, Z. X.; Zhu, C. D.; Shi, Z. Z. J. Am. Chem. Soc. 2018, 140, 9061. |
| [39] | Corbera?n, R.; Mszar, N. W.; Hoveyda, A. H. Angew. Chem. Int. Ed. 2011, 50, 7079. |
| [40] | Liu, Y.; Zhou, Y. H.; Zhao, Y. L.; Qu, J. P. Org. Lett. 2017, 19, 946. |
| [41] | Akiyama, S.; Nomura, S.; Kubota, K.; Ito, H. Org. Lett. 2020, 85, 4172 |
| [42] | Gao, P.; Yuan, C. C.; Zhao, Y.; Shi, Z. Z. Chem. 2018, 4, 2201. |
| [43] | Kojima, R.; Akiyama, S.; Ito, H. Angew. Chem. Int. Ed. 2018, 57, 7196. |
| [44] | Paioti, P. H.; Del Pozo, J.; Mikus, M. S.; Lee, J.; Koh, M. J.; Romiti, F.; Hoveyda, A. H. J. Am. Chem. Soc. 2019, 141, 19917. |
| [45] | Gao, P.; Wang, G. Q.; Xi, L. L.; Wang, M. Y.; Li, S. H.; Shi, Z. Z. Chin. J. Chem. 2019, 37, 1009. |
| [46] | Fan, Z. W.; Ye, M. X.; Wang, Y. H.; Qiu, J.; Li, W. Y.; Ma, X. X.; Yang, K. Song, Q. L. ACS Cent. Sci. 2022, 8, 1134. |
| [47] | Gao, P.; Gao,L. Z.; Xi, L. L.; Zhang, Z. D.; Li, S. H.; Shi, Z. Z. Org. Chem. Front. 2020, 7, 2618. |
| [48] | Oyama, N.; Akiyama, S.; Kubota, K.; Imamoto, T.; Ito, H. Eur. J. Org. Chem. 2022, 31, e202200664. |
/
| 〈 |
|
〉 |