

Chinese Journal of Organic Chemistry >
Synthesis and Herbicidal Activity of Novel Pyrimido[5,4-e]-[1,2,4]triazolo[1,5-c]pyrimidine Derivatives
Received date: 2023-01-08
Revised date: 2023-03-01
Online published: 2023-03-31
Supported by
National Natural Science Foundation of China(21562026); National Natural Science Foundation of China(21762025); Young Talents Program of Jiangxi Normal University.
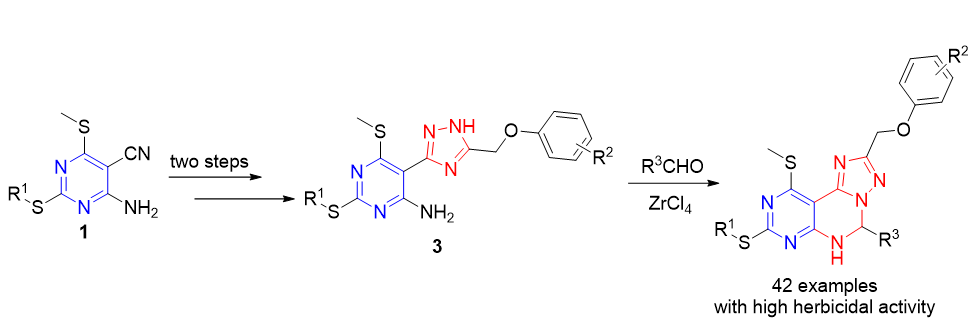
To discover N-containing fused heterocyclic compounds with novel structure and excellent herbicidal activity, forty-two pyrimido[5,4-e][1,2,4]triazolo[1,5-c]pyrimidine derivatives (Ι-1~Ι-42) were synthesied using 2-(alkylthio)-5- carbonitrile-6-(methylthio)pyrimidine-4-aminos as starting materials. Their structures were clearly confirmed by melting point, 1H NMR, 13C NMR, infrared absorption spectroscopy (IR), and high-resolution mass spectra (HRMS). The preliminary bioassay indicated that many target compounds exhibited remarkable herbicidal activities against the monocotyledons (Triticum aestivum L., Echinochloa crusgalli, and Sorghum bicolor) and dicotyledons (Raphanus sativus, Brassica campestris, and Cucumis sativus) at the concentration of 100 mg/L. Especially, compounds Ι-13, Ι-14, Ι-23, Ι-25 and Ι-30~Ι-42 showed excellent herbicidal activities against the roots and stalks of the six test plant at 100 mg/L with 100% inhibition rates.
Hai Lin , Huixiang Nie , Anlin Zhao , Tao Wang , Jin Luo . Synthesis and Herbicidal Activity of Novel Pyrimido[5,4-e]-[1,2,4]triazolo[1,5-c]pyrimidine Derivatives[J]. Chinese Journal of Organic Chemistry, 2023 , 43(7) : 2462 -2475 . DOI: 10.6023/cjoc202301008
| [1] | Navjeet, K.; Neha, A.; Yamini, V.; Pooja, G.; Pranshu, B.; Nirmala, J. K. Curr. Org. Chem. 2021, 25, 1270. |
| [2] | Eftekhari-Sis, B.; Zirak, M.; Akbari, A. Chem. Rev. 2013, 113, 2958. |
| [3] | Yadagiri, D.; Rivas, M.; Gevorgyan, V. J. Org. Chem. 2020, 85, 11030. |
| [4] | Kerru, N.; Gummidi, L.; Maddila, S.; Gangu, K. K.; Jonnalagadda, S. B. Molecules 2020, 25, 1909. |
| [5] | Gao, B.; Yang, B.; Feng, X. D.; Li, C. Nat. Prod. Rep. 2022, 39, 139. |
| [6] | Pedrood, K.; Montazer, M. N.; Larijani, B.; Mahdavi, M. Synthesis 2021, 53, 2342. |
| [7] | Sbei, N. Listratova, A. V.; Titov, A. A.; Voskressensky, L. G. Synthesis 2019, 51, 2455. |
| [8] | Lv, Z. C.; Wang, H. M.; Quan, Z. C.; Gao, Y.; Lei, A. W. Chem. Commun. 2019, 55, 12332. |
| [9] | Liu, Z. K.; Zhao, Q. Q.; Gao, Y.; Hou, Y. X.; Hu, X. Q. Adv. Synth. Catal. 2021, 363, 411. |
| [10] | Chen, X.; Yang, H. Y.; Hu?lsey, M. J.; Yan, N. ACS Sustainable Chem. Eng. 2017, 5, 11096. |
| [11] | Dastan, A.; Kulkarni, A.; T?r?k, B. Green Chem. 2012, 14, 17. |
| [12] | Romdhane, A.; Said, A. B.; Cherif, M.; Jannet, H. B. Med. Chem. Res. 2016, 25, 1358. |
| [13] | Hassan, A. Y.; Abdel-Aziem, A.; Hussain, A. O. J. Heterocycl. Chem. 2020, 57, 542. |
| [14] | Hidalgo, F. J.; Zamora, R. J. Agric. Food Chem. 2022, 70, 79. |
| [15] | Saranya, P. V.; Neetha, M.; Radhika, S.; Anilkumar, G. J. Heterocycl. Chem. 2021, 58, 673. |
| [16] | Wu, P.; Zhang, Y.; Cheng, Y. J. Org. Chem. 2022, 87, 2779. |
| [17] | Li, M.; Wang, Y.; Xu, Y. H. Chin. J. Org. Chem. 2021, 41, 3073 (in Chinese). |
| [17] | (李曼, 汪颖, 徐允河, 有机化学, 2021, 41, 3073.) |
| [18] | Giudice, M. R. D.; Borioni, A.; Mustazza, C.; Gatta, F. J. Heterocycl. Chem. 1994, 31, 1503. |
| [19] | Mohadeszadeh, M.; Rahimizadeh, M.; Eshghi, H. Shiri, A.; Gholizadeh, M.; Shams, A. Helv. Chim. Acta 2015, 98, 474. |
| [20] | Abdelhamid, A. O.; El-Sayed, I. E.; Hussein, M. Z.; Mangoud, M. M. Molecules 2016, 21, 1072. |
| [21] | Sandmann, G. Pestic. Biochem. Phys. 2001, 70, 86. |
| [22] | Zhu, J. J.; He, L. H.; Luo, J.; Xiong, J.; Wang, T. Phosphorus, Sulfur Silicon Relat. Elem. 2021, 196, 948. |
| [23] | Yang, L. Y.; Sun, Y.; Lu, Z. F.; Liang, J. R.; Wang, T.; Luo, J. J. Heterocycl. Chem. 2022, 59, 704. |
| [24] | Yang, L. Y.; Sun, Y.; He, L. H.; Fan., Y. J.; Wang, T.; Luo, J. J. Mol. Struct. 2022, 1259, 132722. |
| [25] | Sun, Q.; Wu, R. Z.; Cai, S. T.; Lin, Y.; Sellers, L.; Sakamoto, K.; He, B.; Peterson, B. R. J. Med. Chem. 2011, 54, 1126. |
| [26] | Zhao, A. L.; Liu, S.; Zhu, Y. M.; Wang, T.; Luo, J. Chin. J. Org. Chem. 2017, 37, 1877 (in Chinese). |
| [26] | (赵安林, 刘姝, 朱咏梅, 王涛, 罗劲, 有机化学, 2017, 37, 1877.) |
/
| 〈 |
|
〉 |