

Chinese Journal of Organic Chemistry >
Silver-Catalyzed Enantioselective Intermolecular γ-Amination of β,γ-Unsaturated Amide
Received date: 2022-11-16
Revised date: 2023-03-05
Online published: 2023-04-10
Supported by
The National Natural Science Foundation of China(22071147)
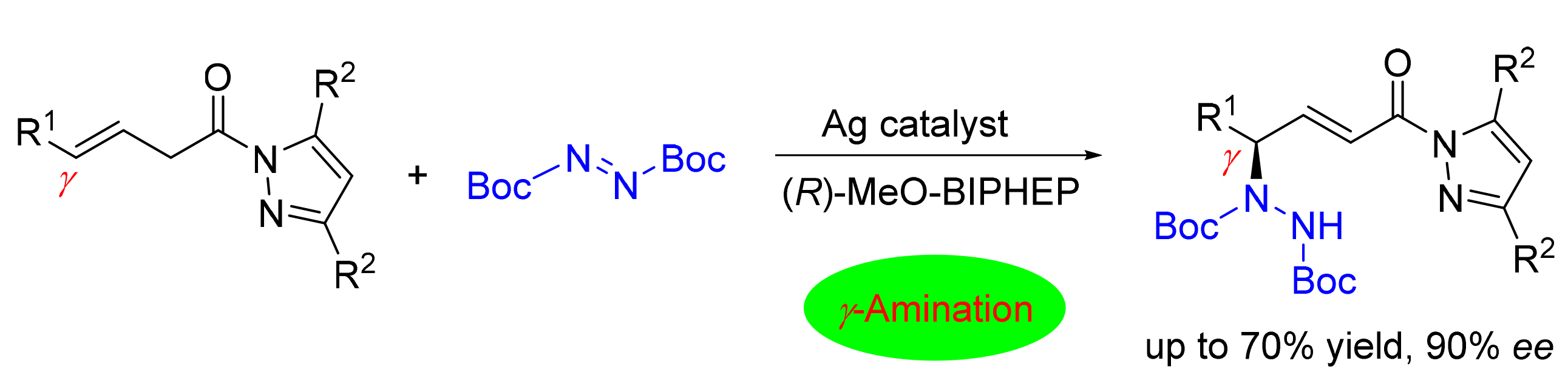
Unnatural γ-amino acid structures are widely present in natural products and drug molecules. Therefore, the synthesis of unnatural amino acids through direct asymmetric amination reaction has important research and application values. An efficient γ-amination reaction using azodicarboxylate as nitrogen source is reported. The asymmetric reaction of azodicarboxylate with β,γ-unsaturated amides was realized by using monovalent silver as Lewis acid catalyst and biphenyl axial chiral bisphosphine compound as ligand. The reaction has good substrate applicability, and a series of chiral γ-amino acid derivatives were obtained in moderate yields with an enantioselectivity of 90% ee.
Jun Ding , Xiaokun Shi , Yu Hao , Heyuan Bai , Shuyu Zhang . Silver-Catalyzed Enantioselective Intermolecular γ-Amination of β,γ-Unsaturated Amide[J]. Chinese Journal of Organic Chemistry, 2023 , 43(8) : 2946 -2952 . DOI: 10.6023/cjoc202211019
| [1] | (a) Müller, P.; Fruit, C. Chem. Rev. 2003, 103, 2905. |
| [1] | (b) Hili, R.; Yudin, A. K. Nat. Chem. Biol. 2006, 2, 284. |
| [1] | (c) Br?se, S. ChemBioChem 2008, 9, 1509. |
| [1] | (d) Valeur, E.; Bradley, M. Chem. Soc. Rev. 2009, 38, 606. |
| [1] | (e) Cho, S. H.; Kim, J. Y.; Kwak, J.; Chang, S. Chem. Soc. Rev. 2011, 40, 5068. |
| [1] | (f) Chen, Y.; Zhu, J.; Zhao, S. Chin. J. Org. Chem. 2019, 39, 1923. (in Chinese) |
| [1] | ( 陈樱, 祝家楠, 赵圣印, 有机化学, 2019, 39, 1923.) |
| [2] | (a) Chebib, M.; Johnston, G. A. R. Clin. Exp. Pharmacol. Physiol. 1999, 26, 937. |
| [2] | (b) Ordó?ez, M.; Cativiela, C. Tetrahedron: Asymmetry 2007, 18, 3. |
| [2] | (c) Sigel, E.; Steinmann, M. E. J. Biol. Chem. 2012, 287, 40224. |
| [3] | Lapin, I. CNS Drug Rev. 2001, 7, 471. |
| [4] | (a) Chizh, B. A.; G?hring, M.; Tr?ster, A.; Quartey, G. K.; Schmelz, M.; Koppert, W. Br. J. Anaesth. 2007, 98, 246. |
| [4] | (b) Ha, K.-Y.; Carragee, E.; Cheng, I.; Kwon, S.-E.; Kim, Y.-H. J. Korean Med. Sci. 2011, 26, 404. |
| [5] | (a) Bertelsen, S.; Marigo, M.; Brandes, S.; Dinér, P.; J?rgensen, K. A. J. Am. Chem. Soc. 2006, 128, 12973. |
| [5] | (b) Wang, J.; Chen, J.; Kee, C. W.; Tan, C.-H. Angew. Chem., Int. Ed. 2012, 51, 2382. |
| [5] | (c) Takeda, T.; Terada, M. J. Am. Chem. Soc. 2013, 135, 15306. |
| [5] | (d) Yang, X.; Toste, F. D. J. Am. Chem. Soc. 2015, 137, 3205. |
| [5] | (e) Chen, X.; Liu, X.; Mohr, J. T. Org. Lett. 2016, 18, 716. |
| [5] | (f) Shang, M.; Wang, X.; Koo, S. M.; Youn, J.; Chan, J. Z.; Yao, W.; Hastings, B. T.; Wasa, M. J. Am. Chem. Soc. 2017, 139, 95. |
| [6] | (a) Bai, H.-Y.; Ma, Z.-G.; Yi, M.; Lin, J.-B.; Zhang, S.-Y. ACS Catal. 2017, 7, 2042. |
| [6] | (b) Fu, X.; Bai, H.-Y.; Zhu, G.-D.; Huang, Y.; Zhang, S.-Y. Org. Lett. 2018, 20, 3469. |
| [6] | (c) Fu, X.; Hao, Y.; Bai, H.-Y.; Duan, A.; Zhang, S.-Y. Org. Lett. 2021, 23, 25. |
| [6] | (d) Qiu, X.-Y.; Li, Z.-H.; Zhou, J.; Lian, P.-F.; Dong, L.-K.; Ding, T.-M.; Bai, H.-Y.; Zhang, S.-Y. ACS Catal. 2022, 12, 7511. |
| [7] | Zhang, H.-J.; Shi, C.-Y.; Zhong, F.; Yin, L. J. Am. Chem. Soc. 2017, 139, 2196. |
/
| 〈 |
|
〉 |