

Chinese Journal of Organic Chemistry >
Photocatalytic Activation of C(sp3)—H Bonds to Form C—S Bonds Catalyzed by (Oxybis(4,1-phenylene))bis(phenylmethanone)
Received date: 2022-10-18
Revised date: 2023-01-09
Online published: 2023-04-23
Supported by
The National Natural Science Foundation(21376222); The Natural Science Foundation of Zhejiang Province(LY18B060011)
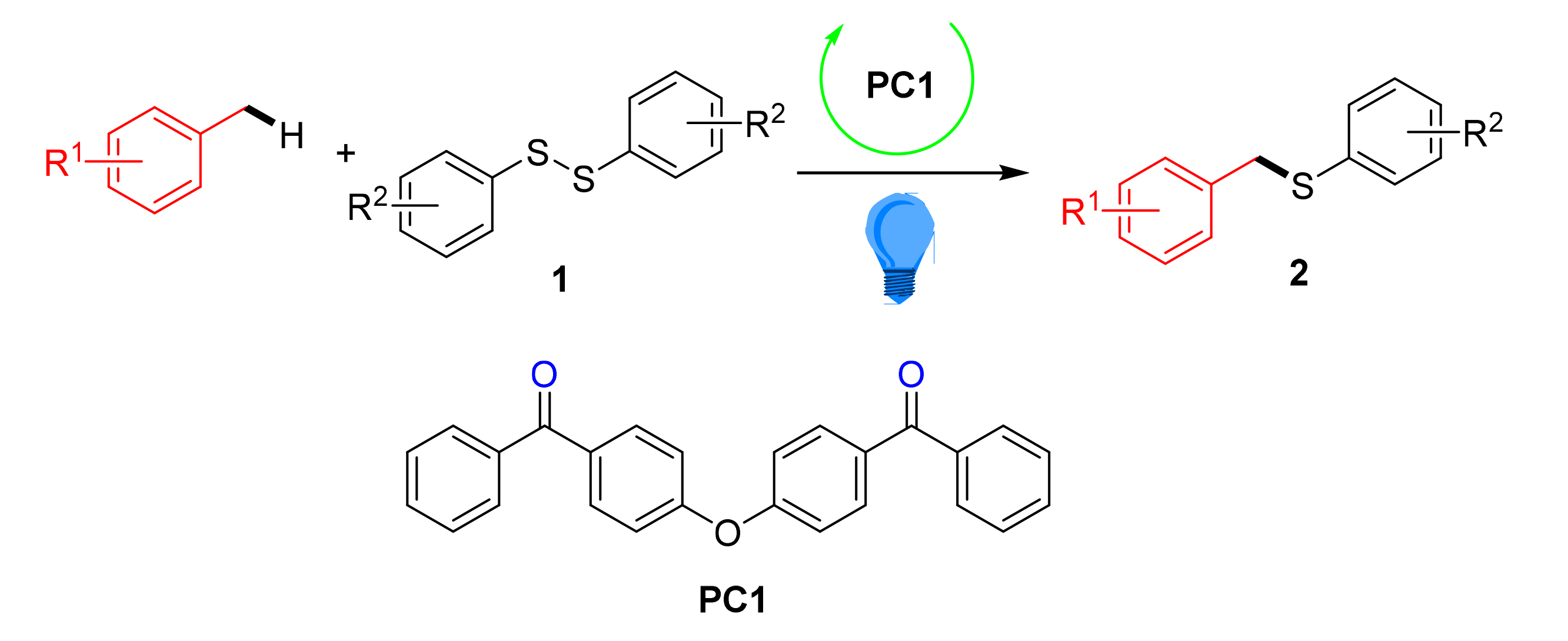
The photocatalytic C(sp3)—H bond activated thioetherification of toluene and its derivatives was carried out with diaryl disulfide as sulfur source, and a series of thioether compounds with 45%~93% yields. Among them, the catalysis of new hydrogen atom transfer (HAT) photosensitizer of (oxybis(4,1-phenylene))bis(phenylmethanone) showed excellent light conversion efficiency, up to 9.3×10-1 mmol•kW-1•h-1. Compared with benzophenone (1.7×10-1 mmol•kW-1•h-1), the light conversion efficiency was 5 times higher. In addition, through the study of the reaction mechanism, the possible mechanism of the benzophenone analogs in the three-line excited state grab hydrogen atoms of toluene methyl to obtain benzyl radicals, then diphenyl sulfide is homolytically cleaved by LEDs irradiation to obtain sulfur radicals, and the bond formation reaction is carried out to obtain sulfide compounds. At the same time, another part of the sulfur radicals under alkali auxiliary through single electron oxidation of benzyl alcohol free radicals returns to the state of benzophenone, completing the photocatalytic cycle.
Lingna Wang , Xiaoqing Liu , Gang Lin , Hongying Jin , Minjun Jiao , Xuefen Liu , Shuping Luo . Photocatalytic Activation of C(sp3)—H Bonds to Form C—S Bonds Catalyzed by (Oxybis(4,1-phenylene))bis(phenylmethanone)[J]. Chinese Journal of Organic Chemistry, 2023 , 43(8) : 2848 -2854 . DOI: 10.6023/cjoc202210018
| [1] | Llinás, A.; Page, M. I. Org. Biomol. Chem. 2004, 2, 651. |
| [2] | Feng, M.; Tang, B.; Liang, S. H.; Jiang, X. Curr. Top. Med. Chem. 2016, 16, 2000. |
| [3] | Li, N. S.; Frederiksen, J. K.; Piccirilli, J. A. Acc. Chem. Res. 2011, 44, 1257. |
| [4] | Syguda, A.; Gielnik, A.; Borkowski, A.; Wo?niak-Karczewska, M.; Parus, A.; Piechalak, A.; Olejnik, A.; Marecik, R.; ?awniczak, ?.; Chrzanowski, ?. New J. Chem. 2018, 42, 9819. |
| [5] | Llinás, A.; Page, M. I. Org. Biomol. Chem. 2004, 2, 651. |
| [6] | Cole, D. C.; Lennox, W. J., Lombardi, S.; Ellingboe, J. W.; Bernotas, R. C.; Tawa, G. J.; Mazandarani, H; Smith, D. L.; Zhang, G.-M.; Coupet, J.; Schechter, L. E. J. Med. Chem. 2005, 48, 353. |
| [7] | Huang, S.; Wang, M.; Jiang, X. Chem. Soc. Rev. 2022, 51, 8351. |
| [8] | Vásquez-Céspedes, S; Ferry, A; Candish, L; Frank, G. Angew. Chem. Int. Ed. 2015, 54, 5772. |
| [9] | (a) Capaldo, L.; Ravelli, D. Eur. J. Org. Chem. 2017, 15, 2056. |
| [9] | (b) Ravelli, D.; Dondi, D.; Fagnoni, M.; Albini, A. Chem. Soc. Rev. 2009, 38, 1999. |
| [10] | Yan, J.; Cheo, H.-W.; Teo, W.-K.; Shi, X.-C.; Wu, H.; Idres, S. B.; Deng, L.-W.; Wu, J. J. Am. Chem. Soc. 2020, 142, 11357. |
| [11] | Ravelli, D.; Protti, S.; Fagnoni, M. Acc. Chem. Res. 2016, 49, 2232. |
| [12] | Nasser, I.; Arash, S. Tetrahedron Lett. 2014, 55, 1212. |
| [13] | Reeves, J. T.; Camara, K.; Han, Z. S.; Xu, Y.; Lee, H. Org. Lett. 2014, 16, 1196. |
| [14] | Bahrami, K.; Mohammad, M. K.; Ahmad, K. Synthesis 2008, 2543. |
| [15] | Schwertz, G.; Frei, M. S.; Witschel, M. C.; Rottmann, M.; Leartsakulpanich, U.; Chitnumsub, P.; Jaruwat, A.; Ittarat, W.; Sch?fer, A.; Aponte, R. A.; Trapp, N.; Mark, K.; Chaiyen, P.; Diederich, F. Chem. Eur. J. 2017, 23, 14345. |
| [16] | Bahrami, K.; Khodaei, M. M.; Arab, M. S. J. Org. Chem. 2010, 75, 6208. |
| [17] | Tanaka, K.; Shoji, T.; Hirano, M. Eur. J. Org. Chem. 2007, 2687. |
| [18] | O'Mahony, G. E.; Ford, A.; Maguire, J. A. R. Org. Chem. 2012, 77, 3288. |
| [19] | Gevorgyan, A.; Mkrtchyan, S.; Grigoryan, T.; Iaroshenko, V. O. Org. Chem. Front. 2017, 4, 2437. |
| [20] | Pathak, A. K.; Pathak, V.; Seitz, L. E.; Suling, W. J.; Reynolds, R. C. J. Med. Chem. 2004, 47, 273. |
| [21] | Bahrami, K.; Khodaei, M. M.; Khodadoustan, N. Synlett 2011, 2206. |
| [22] | Clarke, A. K.; Parkin, A.; Taylor, R. J. K.; Unsworth, W. P.; Rossi-Ashton, J. A. ACS Catal. 2020, 10, 5814. |
| [23] | O’Mahony, G. E.; Eccles, K. S.; Morrison, R. E.; Ford, A.; Lawrence, S. E.; Maguire, A. R. Tetrahedron Lett. 2013, 69, 10168. |
/
| 〈 |
|
〉 |