

Chinese Journal of Organic Chemistry >
Kinetic Resolution of Racemic P-Chiral α-Hydroxymethylphos-phonates Catalyzed by Lipase from Porcine Pancreas
Received date: 2022-12-05
Revised date: 2023-02-25
Online published: 2023-04-23
Supported by
The State Key Laboratory of Nuclear Biological and Chemical Protection for Civilian(SKLNBC2021-09)
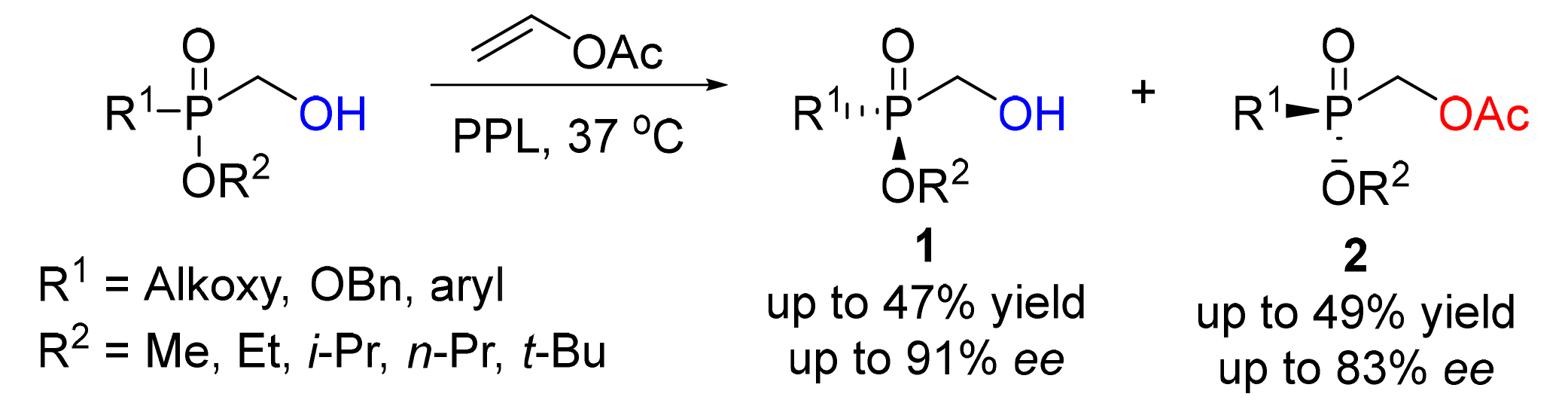
The development of enzyme-catalyzed methods for the synthesis of P-chiral phosphine derivatives has important implications. Herein this work presents a direct biocatalytic transesterification of racemic α-hydroxyphosphonates and vinyl acetate, which provides a rapid access to P-chiral hydroxymethylphosphonates. Catalyzed by lipase from porcine pancreas (PPL), which is commercially available, the reactions proceed efficiently with a wide array of reaction partners to deliver various tertiary phosphine oxides in up to 49% yield and 83% ee under very mild conditions. The enzyme-substrate binding mode was established and the high enantioselectivity of PPL was revealed through docking simulations.
Huijuan Hu , Qiaoli Yan , Xiaogang Lu , Qifan Yang , Chengxin Pei , Hongmei Wang , Runli Gao . Kinetic Resolution of Racemic P-Chiral α-Hydroxymethylphos-phonates Catalyzed by Lipase from Porcine Pancreas[J]. Chinese Journal of Organic Chemistry, 2023 , 43(8) : 2815 -2825 . DOI: 10.6023/cjoc202212005
| [1] | Kazemi, M.; Tahmasbi, A. M.; Valizadeh, R.; Naserian, A. A.; Soni, A. Agric. Sci. Res. J. 2012, 2, 512. |
| [2] | Lamberth, C. Tetrahedron 2010, 66, 7239. |
| [3] | De Clercq, E. Clin. Microbiol. Rev. 2003, 16, 569. |
| [4] | Akiyama, T. Chem. Rev. 2007, 107, 5744. |
| [5] | Milo, A.; Neel, A. J.; Toste, F. D.; Sigman, M. S. Science 2015, 347, 737. |
| [6] | Duffy, M. P.; Delaunay, W.; Bouit, P. A.; Hissler, M. Chem. Soc. Rev. 2016, 45, 5296. |
| [7] | Zhu, R. Y.; Liao, K.; Yu, J. S.; Zhou, J. Acta Chim. Sinica 2020, 78, 193. (in Chinese) |
| [7] | ( 朱仁义, 廖奎, 余金生, 周剑, 化学学报, 2020, 78, 193.) |
| [8] | Tang, W.; Zhang, X. Chem. Rev. 2003, 103, 3029. |
| [9] | Fernández-Pérez, H.; Etayo, P.; Panossian, A.; Vidal-Ferran, A. Chem. Rev. 2011, 111, 2119. |
| [10] | Dutartre, M.; Bayardon, J.; Juge, S. Chem. Soc. Rev. 2016, 45, 5771. |
| [11] | Matsukawa, S.; Sugama, H.; Imamoto, T. Tetrahedron Lett. 2000, 41, 6461. |
| [12] | Wolfgang, R. G.; Dagfinn, O. J. Biol. Chem. 1990, 265, 10484. |
| [13] | Methot, J. L.; Roush, W. R. Adv. Synth. Catal. 2004, 346, 1035. |
| [14] | Seayad, J.; List, B. Org. Biomol. Chem. 2005, 3, 719. |
| [15] | Connon, S. J. Angew. Chem., Int. Ed. 2006, 45, 3909. |
| [16] | Guo, H. F.; Sun, Z.; Wu, Y.; Kwon, O. Chem. Rev. 2018, 118, 10049. |
| [17] | Iseki, K.; Kuroki, Y.; Takahashi, M.; Kobayashi, Y. Tetrahedron Lett. 1996, 37, 5149. |
| [18] | Iseki, K.; Kuroki, Y.; Takahashi, M.; Kishimoto, S.; Kobayashi, Y. Tetrahedron 1997, 53, 3513. |
| [19] | Schulze, C. J.; Navarro, G.; Ebert, D.; DeRisi, J.; Linington, R. G.; Salinipostins, A. K. J. Org. Chem. 2015, 80, 1312. |
| [20] | Clarion, L.; Pirat, J. L.; Bakalara, N. J. Med. Chem. 2012, 55, 2196. |
| [21] | Baraniak, J.; Kinas, R. W.; Lesiak, K.; Stec, W. J. J. Chem. Soc. 1979, 940. |
| [22] | Dostmann, W. R. G.; Taylor, S. S.; Genieser, H. G.; Jastorff, B.; D?skeland, S. O.; ?greid, D. J. Biol. Chem. 1990, 265, 10484. |
| [23] | Vineyard, B. D.; Knowles, W. S.; Sabacky, M. J.; Bachman, G. L.; Weinkauff, D. J. J. Am. Chem. Soc. 1977, 99, 5946. |
| [24] | Korpiun, O.; Lewis, R. A.; Chickos, J.; Mislow, K. J. Am. Chem. Soc. 1968, 90, 4842. |
| [25] | Gwon, D.; Lee, D.; Kim, J.; Park, S.; Chang, S. Chem.-Eur. J. 2014, 20, 12421. |
| [26] | Han, Z. S.; Goyal, N.; Wang, G.; Senanayake, C. H. J. Am. Chem. Soc. 2013, 135, 2474. |
| [27] | Berger, O. Montchamp, J. L. Angew. Chem., Int. Ed. 2013, 52, 11377. |
| [28] | Cooper, D. B.; Inch, T. D.; Lewis, G. J. J. Chem. Soc., Perkin Trans 1 1974, 1043. |
| [29] | Uziel, J.; Stephan, M.; Kaloun, E. B.; Genet, J. P.; Juge, S. Bull. Soc. Chim. Fr. 1997, 134, 379. |
| [30] | Drabowicz, J.; Lopusinski, A.; Heugebaert, T. A.; Stevens, C. V. Top. Heterocycl. Chem. 2010, 21, 103. |
| [31] | Pietrusiewicz, K. M.; Zablocka, M. Chem. Rev. 1994, 94, 1375. |
| [32] | Grabulosa, A.; Granell, J.; Muller, G. Coord. Chem. Rev. 2007, 251, 25. |
| [33] | Lurh, S.; Holz, J.; Borner, A. ChemCatChem 2011, 3, 1708. |
| [34] | Kolodiazhnyi, O. I.; Kukhar, V. P.; Kolodiazhna, A. O. Tetrahedron: Asymmetry 2014, 25, 865. |
| [35] | Ao, Y. F.; Wang, Q. Q.; Wang, D. X. Chin. J. Org. Chem. 2016, 36, 2333. (in Chinese) |
| [35] | ( 敖宇飞, 王其强, 王德先, 有机化学, 2016, 36, 2333.) |
| [36] | Wu, Z.; Liu, C.; Zhang, Z.; Zheng, R.; Zheng, Y. Biotechnol. Adv. 2020, 43, 107574. |
| [37] | Wang, M. X. Acc. Chem. Res. 2015, 48, 602. |
| [38] | Hu, H. J.; Wang, Q. Q.; Wang, D. X.; Ao, Y. F. Green Synth. Catal. 2021, 2, 324. |
| [39] | Hu, H. J.; Wang, Q. Q.; Wang, D. X.; Ao, Y. F. Adv. Synth. Catal. 2021, 363, 4538. |
| [40] | Ao, Y. F.; Hu, H. J.; Zhao, C. X.; Chen, P.; Huang, T.; Chen, H.; Wang, Q. Q.; Wang, D. X.; Wang, M. X. ACS Catal. 2021, 11, 6900. |
| [41] | Hu, H. J.; Chen, P.; Ao, Y. F.; Wang, Q. Q.; Wang, D. X.; Wang, M. X. Org. Chem. Front. 2019, 6, 808. |
| [42] | Sabbioni, G.; Shea, M. L.; Jones, J. B. J. Chem. Soc., Chem. Commun. 1984, 236. |
| [43] | Kobayashi, S.; Kamiyama, K.; Iimori, T.; Ohno, M. Tetrahedron Lett. 1984, 25, 2557. |
| [44] | Heiss, L.; Gais, H. J. Tetrahedron Lett. 1995, 36, 3833. |
| [45] | Sousa, H. A.; Afonso, C. A. M.; Mota, J. P. B.; Crespo, J. G. Ind. Eng. Chem. Res. 2003, 42, 5516. |
| [46] | Suss, P.; Illner, S.; Langermann, J. V.; Borchert, S.; Bornscheuer, U. T.; Wardenga, R.; Kragl, U. Org. Process Res. Dev. 2014, 18, 897. |
| [47] | Suss, P.; Borchert, S.; Hinze, J.; Illner, S.; Langermann, J. V.; Kragl, U.; Bornscheuer, U. T.; Wardenga, R. Org. Process Res. Dev. 2015, 19, 2034. |
| [48] | Meissner, M. P.; Suss, P.; Brundiek, H.; Woodley, J. M.; Langermann, J. V. Org. Process Res. Dev. 2018, 22, 1518. |
| [49] | Lutz, S. Tetrahedron: Asymmetry 2004, 15, 2743. |
| [50] | Kie?basinski, P.; Albrycht, M.; Luczak, J.; Miko?ajczyk, M. Tetrahedron: Asymmetry 2002, 13, 735. |
| [51] | Matsuda, T.; Harada, T.; Nakamura, K.; Ikariya, T. Tetrahedron: Asymmetry 2005, 16, 909. |
| [52] | Albrycht, M.; Kielbasinski, P.; Drabowicz, J.; Miko?ajczyk, M.; Matsuda, T.; Harada, T.; Nakamura, K. Tetrahedron: Asymmetry 2005, 16, 2015. |
| [53] | Kielbasinski, P.; Omelanczuk, J.; Miko?ajczyk, M. Tetrahedron: Asymmetry 1998, 9, 3283. |
| [54] | Chen, C. S.; Fujimoto, Y.; Girdaukas, G.; Sih, C. J. J. Am. Chem. Soc. 1982, 104, 7294. |
| [55] | Chen, C. S.; Wu, S. H.; Girdaukas, G.; Sih, C. J. J. Am. Chem. Soc. 1987, 109, 2812. |
/
| 〈 |
|
〉 |