

Chinese Journal of Organic Chemistry >
Strategy to Construct Functionalized Tetrazepine Derivatives via [4+3] Annulation Reaction of Azomethine Imine with Azadiene Precursor
Received date: 2023-03-21
Revised date: 2023-05-15
Online published: 2023-06-07
Supported by
Guizhou Provincial Science and Technology Plan Program(QKHJC-ZK[2023]-495); Chongqing Natural Science Foundation(cstc2021jcyj-bsh0010)
Tetrazepine derivative exhibits good biological activities. Only a limited method has been devoted to constructing these frameworks to date. Therefore, an efficient and novel way was established for the synthesis of functionalized tetrazepine scaffolds through the cycloaddition reaction of azomethine imine with 1,2-azadiene formed in situ by α-halogeno hydrazones in the presence of KOH. This transformation shows excellent functional group tolerance and substrate scope.
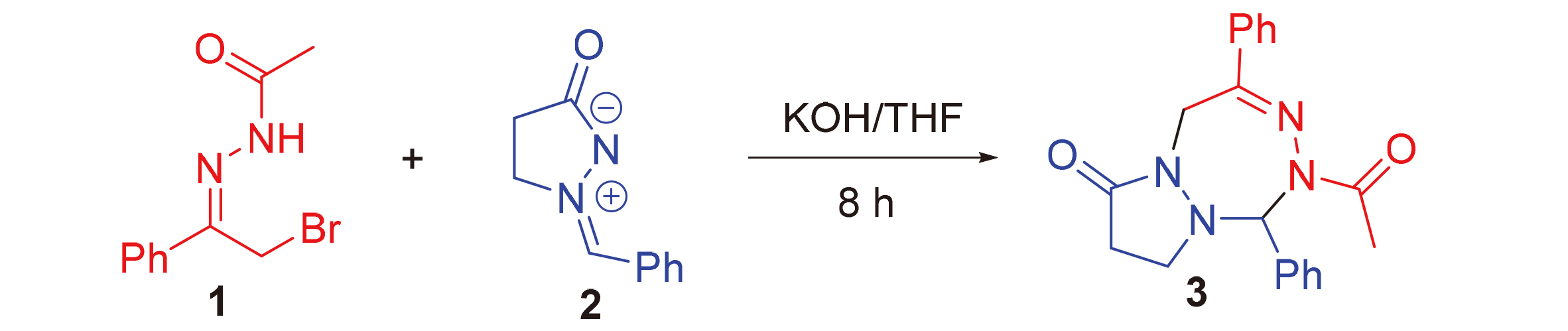
Xiaoke Zhang , Xiangru Zheng , Chaoyong Wang . Strategy to Construct Functionalized Tetrazepine Derivatives via [4+3] Annulation Reaction of Azomethine Imine with Azadiene Precursor[J]. Chinese Journal of Organic Chemistry, 2023 , 43(9) : 3180 -3187 . DOI: 10.6023/cjoc202303031
| [1] | For some selected examples: (a) Hu X.; Li, D.; Chu, C.; Li, X.; Wang, X. H.; Jia, Y.; Hua, H. M.; Xu, F. X. Int. J. Mol. Sci. 2018, 19, 3403. |
| [1] | (b) Xiao Y.; Gong Q.; Wang W. H.; Liu F.; Kong Q. H.; Pan F.; Zhang X. K.; Yu C. Y.; Hu S. S.; Fan F.; Li S. H.; Liu Y. Cancer Cell Int. 2020, 20, 371. |
| [1] | (c) Dong G. Q.; Liu Y.; Wu Y.; Tu J.; Chen S. Q.; Liu N.; Sheng C. Q. Chem. Commun. 2018, 54, 13535. |
| [1] | (d) Fu D. J.; Li J.; Yu B. Eur. J. Med. Chem. 2021, 214, 113254. |
| [1] | (e) Abdel-Halim M.; Sigler S.; Racheed N. A. S.; Hefnawy A.; Fathalla R. K.; Hammam M. A.; Maher A.; Maxuitenko Y. L.; Keeton A. B.; Hartmann R. W.; Engel M.; Piazza G. A.; Abadi A. H. J. Med. Chem. 2021, 64, 4462. |
| [2] | (a) Krueger B. W.; Fischer R.; Bertram H. J.; Bretschneider T.; Boehm S.; Krebs A.; Schenke T.; Santel H. J.; Lurssen K.; Schmidt R. R.; Erdelen C.; Wachendorff-Neumann U.; Stendel, W. EP0508126, 1992. |
| [2] | (b) Muehlebach M.; Cederbaum F.; Cornes D.; Friedmann A. A.; Glock J.; Hall G.; Indolese A. F.; Kloer D. P.; Goupil G. L.; Maetzke T.; Meier H.; Schneider R.; Stoller A.; Szczepanski H.; Wendeborna S.; Widmer H. Pest. Manage. Sci. 2011, 67, 1499. |
| [2] | (c) Kamata M.; Yamashita T.; Tokkyo Koho, J. K. JP 2009196966, 2009. |
| [2] | (d) Kamata M.; Yamashita T.; Kina A.; Tawada M.; Endo S.; Mizukami A.; Sasaki M.; Tani A.; Nakano Y.; Watanabe Y.; Furuyama N.; Funami M.; Amano N.; Fukatsu K. Bioorg. Med. Chem. Lett. 2012, 22, 4769. |
| [3] | (a) Kodato S. I.; Wada H.; Saito S.; Takeda M.; Nishibata Y.; Aoe K.; Date T.; Onoda Y.; Tamaki H. Chem. Pharm. Bull. 1987, 35, 80. |
| [3] | (b) Mashevskaya I. V.; Makhmudov R. R.; Kuslina L. V.; Mokrushin I. G.; Shurov S. N.; Maslivets A. N. Pharm. Chem. J. 2012, 45, 660. |
| [3] | (c) Ibrahim S. M.; Baraka M. M.; El-Sabbagh O. I.; Kothayer H. Med. Chem. Res. 2013, 22, 1488. |
| [4] | Almerico A. M.; Mingoia F.; Diana P.; Barraja P.; Lauria A.; Montalbano A.; Cirrincione G.; Dattolo G. J. Med. Chem. 2005, 48, 2859. |
| [5] | (a) Gupta C. M.; Bhaduri A. P.; Khanna N. M. J. Med. Chem. 1968, 11, 392. |
| [5] | (b) Ghorab M. M.; Heiba H. I.; El-gawish M. A. Phosphorus, Sulfur Silicon Relat. Elem. 1995, 106, 85. |
| [5] | (c) Hamama W. S.; El-Bana G. G.; Shaaban S.; Zoorob H. H. J. Heterocycl. Chem. 2018, 55, 971. |
| [6] | (a) Maggio B.; Raffa D.; Raimondi M. V.; Cascioferro S.; Plescia F.; Tolomeo M.; Barbusca E.; Cannizzo G.; Mancuso S.; Daidone G. Eur. J. Med. Chem. 2008, 43, 2386. |
| [6] | (b) Maggio B.; Raffa D.; Raimondi M. V. ARKIVOC 2006, xv, 120. |
| [6] | (c) Vahedi H.; Rajabzadeh G.; Farvandi F. Chin. Chem. Lett. 2010, 21, 1419. |
| [6] | (d) Darehkordi A.; Khorasani F. N.; Mohammadi M.; Kazemi E. Monatsh. Chem. 2020, 151, 1835. |
| [7] | For selected examples, see: (a) Zhou M. B.; Song, R. J.; Wang, C. Y.; Li, J. H. Angew. Chem., Int. Ed. 2013, 52, 10805. |
| [7] | (b) Feng J. J.; Lin T. Y.; Wu H. H.; Zhang J. L. J. Am. Chem. Soc. 2015, 137, 3787. |
| [7] | (c) Liu L. L.; Chiu P. Chem. Commun. 2011, 47, 3416. |
| [7] | (d) Gao H. Y.; Wu X. X.; Zhang J. L. Chem. Commun. 2010, 46, 8764. |
| [8] | Kumari Y. B. Heterocycl. Commun. 2007, 13, 177. |
| [9] | Li Z. F.; Li S. K.; Kan T. J.; Wang X. Y.; Xin X.; Hou Y. L.; Gong P. Adv. Synth. Catal. 2020, 362, 2626. |
| [10] | (a) Huisgen R.; Fleischmann R.; Eckell A. Tetrahedron Lett. 1960, 1, 1. |
| [10] | (b) Breugst M.; Reissig H. U. Angew. Chem., Int. Ed. 2020, 59, 12293. |
| [10] | (c) Yue G. Z.; Liu B. Chin. J. Org. Chem. 2020, 40, 3132. (in Chinese) |
| [10] | (乐贵洲, 刘波, 有机化学, 2020, 40, 3132.) |
| [11] | (a) Bakthadoss M.; Agarwal V. ChemistrySelect 2018, 3, 6960. |
| [11] | (b) Choi A.; Castle J.; Saruengkhanphasit R.; Coldham I. Synthesis 2020, 1273. |
| [11] | (c) Sandmeier T.; Sievertsen N.; Carreira E. M. Helv. Chim. Acta 2020, 103, e2000058. |
| [12] | (a) Li Z.; Yu H.; Liu Y.; Zhou L.; Sun Z.; Guo H. Adv. Synth. Catal. 2016, 358, 1880. |
| [12] | (b) Zhou L.; Yuan C.; Zhang C.; Zhang L.; Gao Z.; Wang C.; Liu H.; Wu Y.; Guo H. Adv. Synth. Catal. 2017, 359, 2316. |
| [12] | (c) Zhu C. Z.; Feng J. J.; Zhang J. Chem. Commun. 2017, 53, 4688. |
| [12] | (d) Yu L.; Zhong Y.; Yu J.; Gan L.; Cai Z.; Wang R.; Jiang X. Chem. Commun. 2018, 54, 2353. |
| [12] | (e) Cheng X.; Cao X.; Xuan J.; Xiao W. J. Org. Lett. 2018, 20, 52. |
| [13] | (a) Wei L.; Wang Z. F.; Yao L.; Qiu G. F.; Tao H. Y.; Li H.; Wang C. J. Adv. Synth. Catal. 2016, 358, 3955. |
| [13] | (b) Wang Y. F.; Zhu L. P.; Wang M. R.; Xiong J.; Chen N. N.; Feng X.; Xu Z. Q.; Jiang X. X. Org. Lett. 2018, 20, 6506. |
| [14] | (a) Li Z.; Feng Y. L.; Hou Z. F.; Zhang L.; Yang W. J.; Wu Y.; Xiao Y. M.; Guo H. C. RSC Adv. 2015, 5, 34481. |
| [14] | (b) Mei G. J.; Zhu Z. Q.; Zhao J. J.; Bian C. Y.; Chen J.; Chen R. W.; Shi F. Chem. Commun. 2017, 53, 2768. |
| [14] | (c) Li C.; Wang C. S.; Li T. Z.; Mei G. J.; Shi F. Org. Lett. 2019, 21, 598. |
| [14] | (d) Yuan C. H.; Zhou L. J.; Xia M. R.; Sun Z. H.; Wang D. Q.; Guo H. C. Org. Lett. 2016, 18, 5644. |
| [14] | (e) Na R.; Jing C.; Xu Q.; Jiang H.; Wu X.; Shi J. J. Am. Chem. Soc. 2011, 133, 13341. |
| [14] | (f) Wang M.; Huang Z. J.; Xu J. F.; Chi Y. R. J. Am. Chem. Soc. 2014, 136, 1214. |
| [14] | (g) Na R.; Jing C. F.; Xu Q. H.; Jiang H.; Wu X.; Shi J. Y.; Zhong J. C.; Wang M.; Benitez D.; Tkatchouk E.; Goddard W. A.; Guo H. C.; Kwon O. J. Am. Chem. Soc. 2011, 133, 13337. |
| [14] | (h) Hu X. Q.; Chen J. R.; Gao S.; Feng B.; Lu L. Q.; Xiao W. J. Chem. Commun. 2013, 49, 7905. |
| [14] | (i) Yang W. J.; Yuan C. H.; Liu Y.; Mao B. M.; Sun Z. H.; Guo H. C. J. Org. Chem. 2016, 81, 7597. |
| [15] | (a) Jin Q.; Zhang J.; Jiang C.; Zhang D.; Gao M.; Hu S. J. Org. Chem. 2018, 83, 8410. |
| [15] | (b) Hu X. Q.; Chen J. R.; Gao S.; Feng B.; Lu L. Q.; Xiao W. J. Chem. Commun. 2013, 49, 7906. |
| [15] | (c) Zhi Y.; Zhao K.; Shu T.; Enders D. Synthesis 2016, 48, 240. |
| [15] | (d) Chen L.; Yang G. M.; Wang J.; Jia Q. F.; Wei J.; Du Z. Y. RSC Adv. 2015, 5, 76697. |
| [16] | For selected examples, see: (a) Zhang X. K.; Pan, Y.; Wang, H. B.; Liang, C.; Ma, X. F.; Jiao, W.; Shao, H. W. Adv. Synth. Catal. 2021, 363, 459. |
| [16] | (b) Chen J. R.; Dong W. R.; Candy M.; Pan F. F.; Jo?rres M.; Bolm C. J. Am. Chem. Soc. 2012, 134, 6924. |
| [16] | (c) Huang R.; Chang X.; Li J.; Wang C. J. J. Am. Chem. Soc. 2016, 138, 3998. |
| [16] | (d) Quan B. X.; Zhuo J. R.; Zhao J. Q.; Zhang M. L.; Zhou M. Q.; Zhang X. M.; Yuan W. C. Org. Biomol. Chem. 2020, 18, 1886. |
| [16] | (f) Chen B.; Chu W. D.; Liu Q. Z. RSC Adv. 2019, 9, 1487. |
| [16] | (g) Huang R.; Chang X.; Li J.; Wang C. J. J. Am. Chem. Soc. 2016, 138, 3998. |
| [16] | (h) Yin W. H.; Fang L.; Wang Z. Y.; Gao F.; Li Z. F.; Wang Z. Y. Org. Lett. 2020, 21, 7361. |
| [16] | (i) Cao W. B.; Xu X. P.; Ji S. J. Org. Biomol. Chem. 2017, 15, 1651. |
| [16] | (j) Zhong X. G.; Lv J.; Luo S. Org. Lett. 2015, 17, 1561. |
| [16] | (k) Deng Y. M.; Pei C.; Arman H.; Dong K. Y.; Xu X. F.; Doyle M. P. Org. Lett. 2016, 18, 5884. |
| [17] | Zhao H. W.; Pang H. L.; Tian T.; Li B.; Chen X. Q.; Song X. Q.; Meng W.; Yang Z.; Liu Y. Y.; Zhao Y. D. Adv. Synth. Catal. 2016, 358, 1826. |
| [18] | Cheng B.; Li Y. T.; Wang T. M.; Zhang X. P.; Li H.; Li Y.; Zhai H. B. Chem. Commun. 2019, 55, 14606. |
| [19] | Zhang X. K.; Wang H. B.; Li Z. W.; Shu Y.; Gan S. Zhang X. F.; Shao H. W.; Wang C. Y. ACS Omega 2022, 7, 40963. |
/
| 〈 |
|
〉 |