

Chinese Journal of Organic Chemistry >
Pd-Catalyzed C(2)—H Arylation of 3-(2-Aminopyrimidin-4-yl)indoles
Received date: 2023-04-09
Revised date: 2023-05-24
Online published: 2023-07-06
Supported by
Natural Science Foundation of Heilongjiang Province(LH2022B003); the Programme of Introducing Talents of Discipline to Universities (111 Project)(B20088)
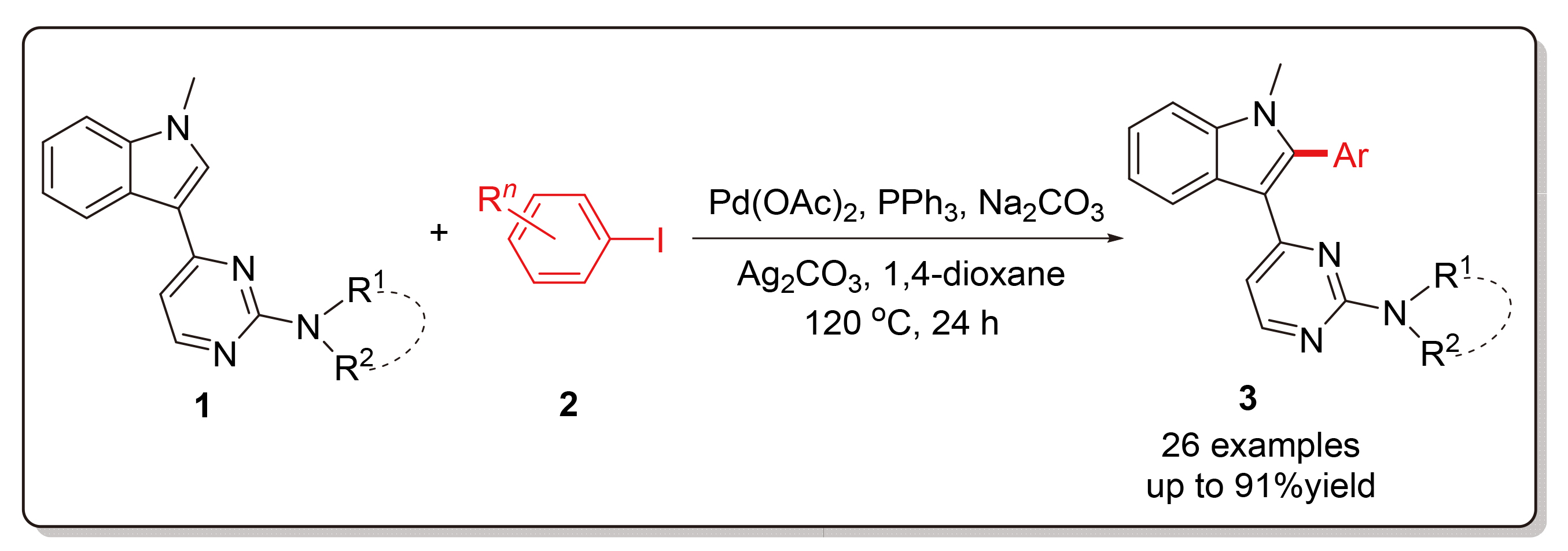
Using N-methyl-3-(2-aminopyrimidin-4-yl)indole derivatives and aryl iodides as starting materials, palladium- catalyzed selective C—H bond activation/arylation at the C-2 position of indole was investigated in detail. With palladium acetate as the catalyst, triphenylphosphine as the ligand, silver carbonate as the additive and sodium carbonate as the base, various 2-arylindole products were obtained in high yields in 1,4-dioxane at 120 ℃ for 24 h. Under these standard conditions, the scope and limitation of the synthetic method were explored by changing the structure of two substrates, and the synthesized indole derivatives were characterized by 1H NMR, 13C NMR and HRMS spectra.
Meijiao Sun , Jing Tan , Yu Tan , Jinsong Peng , Chunxia Chen . Pd-Catalyzed C(2)—H Arylation of 3-(2-Aminopyrimidin-4-yl)indoles[J]. Chinese Journal of Organic Chemistry, 2023 , 43(11) : 3945 -3959 . DOI: 10.6023/cjoc202304012
| [1] | (a) Dorababu, A. RSC Med. Chem. 2020, 11, 1335. |
| [1] | (b) Han, Y.; Dong, W.; Guo, Q.; Li, X; Huang, L. Eur. J. Med. Chem. 2020, 203, 112506. |
| [1] | (c) Qin, H. L.; Liu, J.; Fang, W. Y.; Ravindar, L; Rakesh, K. P. Eur. J. Med. Chem. 2020, 194, 112245. |
| [1] | (d) Colenda, B. E.; Lee, H. S.; Reibenspies, J. H.; Hancock, R. D. Inorg. Chim. Acta. 2018, 482, 478. |
| [1] | (e) Zhao, X. H.; Jia, Y. X.; Li, J. J.; Dong, R.H.; Zhang, J.J.; Ma, C. X.; Wang, H.; Rui, Y.K.; Jiang, X.Y. ACS Appl. Mater. Inter. 2018, 35, 29398. |
| [1] | (f) Mohbiya, D. R.; Sekar, N. Comput. Theor. Chem. 2018, 1139, 90. |
| [1] | (g) Zhao, J. F.; Dong, H.; Zheng, Y. J. J. Lumin. 2018, 195, 228. |
| [2] | Zhang, M. Z.; Chen, Q.; Yang, G. F. Eur. J. Med. Chem. 2015, 89, 42. |
| [3] | (a) Austin, O.; Chelsea, S.; Patrick, S.; Kent, A. C. ACS Med. Chem. Lett. 2018, 9, 901. |
| [3] | (b) Cong, H.; Zhao, X. H.; Castle, B. T.; Pomeroy, E. J.; Zhou, B.; John, L.; Wang, Y.; Bian, T. F.; Miao, Z. Y.; Zhang, W. N.; Sham, Y. Y.; David, J. O.; Craig, E. E.; Xing, C. G.; Zhuang, C. L. Mol. Pharmaceut. 2018, 15, 3892. |
| [3] | (c) Singh, P. K.; Silakari, O. Bioorg. Chem. 2018, 79, 163. |
| [4] | Kaushik, N. K.; Kaushik, N.; Attri, P.; Kumar, N.; Kim, C. H.; Verma, A. K.; Choi, E. H. Molecules 2013, 18, 6620. |
| [5] | Ding, C. F.; Ma, H. X.; Yang, J.; Qin, X. J.; Guy, S. S.; Yu, H. F.; Wei, X.; Liu, Y. P.; Huang, W. Y.; Yang, Z. F.; Wang, X. H.; Luo, X. D.; Org. Lett. 2018, 20, 2702. |
| [6] | (a) Zhang, Z.; Tanaka, K.; Yu, J. Q. Nature. 2017, 543, 538. |
| [6] | (b) Zhang, F. L.; Hong, K.; Li, T. J.; Yu, J. Q. Science 2016, 351, 252. |
| [7] | Dalpozzo, R.; Bartoli, G.; Bencivenni, G. Chem. Soc. Rev. 2012, 41, 7247. |
| [8] | Kumari, A.; Singh, R. K. Bioorg. Chem. 2019, 89, 103021. |
| [9] | (a) Zhang, S. S.; Tan, Q. W.; Guan, L. P. Mini-Rev. Med. Chem. 2021, 21, 2285. |
| [9] | (b) Okada, M.; Sugita, T.; Wong, C. P.; Wakimoto, T.; Abe, I. J. Nat. Prod. 2017, 80, 1205. |
| [9] | (c) Hong, W.; Li, J.; Chang, Z; Tan, X. L.; Yang, Y.; Ouyang, Y. F.; Yang, Y. H.; Kaur, S.; Paterson, I. C.; Ngeow, Y. F.; Wang, H. J. Antibiot. 2017, 70, 832. |
| [9] | (d) Kherkhache, H.; Benabdelaziz, I.; Silva, A. M. S.; Lahrech, M. B.; Benalia, M.; Haba, H. Nat. Prod. Res. 2020, 34, 1528. |
| [9] | (e) Pulla, R. S.; Ummadi, N.; Gudi, Y.; Venkatapuram, P.; Adivireddy, P. J. Heterocyclic. Chem. 2018, 55, 115. |
| [10] | (a) Esvan, Y. J.; Giraud, F.; Pereira, E.; Suchaud, V.; Nauton, L.; Théry, V.; Dezhenkova, L. G.; Kaluzhny, D. N.; Mazov, V. N.; Shtil, A. A.; Anizon, F.; Moreau, P. Bioorg. Med. Chem. 2016, 24, 3116. |
| [10] | (b) Karimabad, M. N.; Mahmoodi, M.; Jafarzadeh, A.; Darekordi, A.; Hajizadeh, M. R.; Hassanshahi, G. Mini-Rev. Med. Chem. 2019, 19, 540. |
| [11] | Bian, M.; Wang, Z.; Xiong, X.; Sun, Y.; Matera, C.; Nicolaou, K. C.; Li, A. J. Am. Chem. Soc. 2012, 134, 8078. |
| [12] | Vasconcelos, S. N.; Meissner, K. A.; Ferraz, W. R.; Trossini, G. H.; Wrenger, C.; Stefani, H. A. Future Med. Chem. 2019, 11, 525. |
| [13] | Chadha, N.; Silakari, O. Eur. J. Med. Chem. 2017, 134, 159. |
| [14] | Singh, P.; Prasher, P.; Dhillon, P.; Bhatti, R. Eur. J. Med. Chem. 2015, 97, 104. |
| [15] | Sravanthi, T. V.; Manju, S. L. Eur. J. Pharm. Sci. 2016, 91, 1. |
| [16] | Ye, Q. Y.; Xu, Y.; Zhao, J.; Gao, X. X; Chen, M. J.; Pan, R. l.; Zhong, W.; Wang, M. Z. Transl. Oncol. 2023, 31, 101637. |
| [17] | Barnes, L.; Blaber, H.; Brooks, D. T. K.; Byers, L.; Buckley, D.; Byron, Z. C.; Chilvers, R. G.; Cochrane, L.; Cooney, E.; Damian, H. A.; Francis, L.; He, D. F.; Grace, J. M. J.; Green, H. J.; Hogarth, E. J. P.; Jusu, L.; Killalea, C. E.; King, O.; Lambert, J.; Lee, Z. J.; Lima, N. S.; Long, C. L.; Mackinnon, M.-L.; Mahdy, S.; Matthews-Wright, J.; Millward, M. J.; Meehan, M. F.; Merrett, C.; Morrison, L.; Parke, H. R. I.; Payne, C.; Payne, L.; Pike, C.; Seal, A.; Senior, A. J.; Smith, K. M.; Stanelyte, K.; Stillibrand, J.; Szpara, R.; Taday, F. F. H.; Threadgould, A. M.; Trainor, R. J.; Waters, J.; Williams, O.; Wong, C. K. W.; Wood, K.; Barton, N.; Gruszka, A.; Henley, Z.; Rowedder, J. E.; Cookson, R.; Jones, K. L.; Nadin, A.; Smith, I. E.; Macdonald, S. J. F.; Nortcliffe, A. J. Med. Chem. 2019, 62, 10402. |
| [18] | (a) Jian, H. H.; Xin, R. W.; Rui, N. D.; Xiao, Y. L.; Hong, M. L.; Tian, Y. Z.; Jun, Y. X.; Chen, H. L.; Yan, M.; Zhang, S. H.; Hou, W. F.; Tang, T. L.; Ya, D. C. J. Med. Chem. 2021, 64, 12548. |
| [18] | (b) Yuan, S.; Wang, B.; Dai, Q. Q.; Zhang, X. N.; Zhang, J. Y.; Zuo, J. H.; Liu, H.; Chen, Z. S.; Li, G. B.; Wang, S. M.; Liu, H. M.; Yu, B. J. Med. Chem. 2021, 64, 14895. |
| [19] | (a) Deng, H.; Jung, J.-K.; Liu, T.; Kuntz, K. W.; Snapper, M. L.; Hoveyda, A. H. J. Am. Chem. Soc. 2003, 125, 9032. |
| [19] | (b) Nicolaou, K. C.; Bheema Rao, P.; Hao, J.; Reddy, M. V.; Rassias, G.; Huang, X.; Chen, D. Y.; Snyder, S. A. Angew. Chem., Int. Ed. 2003, 42, 1753. |
| [19] | (c) Nicolaou, K. C.; Hao, J.; Reddy, M. V.; Rao, P. B.; Rassias, G.; Snyder, S. A.; Huang, X.; Chen, D. Y. K.; Brenzovich, W. E.; Giuseppone, N.; Giannakakou, P.; O'Brate, A. J. Am. Chem. Soc. 2004, 126, 12897. |
| [19] | (d) Sitnikov, N.; Velder, J.; Abodo, L.; Cuvelier, N.; Neudorfl, J.; Prokop, A.; Krause, G.; Fedorov, A. Y.; Schmalz, H. G. Chem. - Eur. J. 2012, 18, 12096. |
| [20] | Kumar, P.; Nagtilak, P. J.; Kapur, M. New J. Chem. 2021, 45, 13692. |
| [21] | (a) Chen, Z.; Wang, B.; Zhang, J.; Yu, W.; Liu, Z.; Zhang, Y. Org. Chem. Front. 2015, 2, 1107. |
| [21] | (b) Sandtorv, A. H. Adv. Synth. Catal. 2015, 357, 2403. |
| [21] | (c) Wen, J.; Shi, Z. Acc. Chem. Res. 2021, 54, 1723. |
| [21] | (d) Luo, J. F.; Xu, X.; Zheng, J. L. Chin. J. Org. Chem. 2018, 38, 363. (in Chinese) |
| [21] | (骆钧飞, 徐星, 郑俊良, 有机化学, 2018, 38, 363.) |
| [22] | (a) Islam, S.; Larrosa, I. Chem. Eur. J. 2013, 19, 15093. |
| [22] | (b) Zheng, J.; Zhang, Y.; Cui, S. L. Org. Lett. 2014, 16, 3560. |
| [22] | (c) Miao, T.; Li, P.; Wang, G. W.; Wang, L. Chem. Asian J. 2013, 8, 3185. |
| [23] | (a) Lu, M.-Z.; Lu, P.; Xu, Y.-H.; Loh, T.-P. Org. Lett. 2014, 16, 2614. |
| [23] | (b) Lu, M.-Z.; Ding, X.; Shao, C. D.; Hu, Z. S.; Luo, H. Q.; Zhi, S. Z.; Hu, H. Y.; Kan, Y. H.; Loh, T.-P. Org. Lett. 2020, 22, 2663. |
| [23] | (c) Tiwari, V. K.; Kamal, N.; Kapur, M. Org. Lett. 2015, 17, 1766. |
| [24] | (a) Sun, P.; Yang, J. J.; MO, B. C.; Chen, X.; Li, X.; Chen, C. X. J. Org. Chem. 2020, 85, 6761. |
| [24] | (b) Chen, X.; Sun, P.; MO, B. C.; Chen, C. X.; Peng, J. S. J. Org. Chem. 2021, 86, 352. |
| [24] | (c) Li, X.; Chen, X.; Wang, H.; Chen, C. X.; Sun, P.; MO, B. C.; Peng, J. S. Org. Biomol. Chem. 2019, 17, 4014. |
| [24] | (d) Li, X.; Bian, Y. Y.; Chen, X.; Zhang, H.; Wang, W.; Ren, S. D.; Yang, X. C.; Lu, C.; Chen, C. X.; Peng, J. S. Org. Biomol. Chem, 2019, 17, 321. |
| [24] | (e) Yue, Y. X.; Peng, J. S.; Wang, D. Q.; Bian, Y. Y.; Sun, P.; Chen, C. X. J. Org. Chem. 2017, 82, 5481. |
| [25] | Yanagisawa, S.; Itami, K. Tetrahedron. 2011, 67, 4425. |
| [26] | (a) Mota, A. J.; Dedieu, A.; Bour, C.; Suffert, J. J. Am. Chem. Soc. 2005, 127, 7171. |
| [26] | (b) Garcia-Cuadrado, D.; Braga, A. A. C.; Maseras, F.; Echavarren, A. M. J. Am. Chem. Soc. 2006, 128, 6798. |
| [27] | Jia, H. J.; Wu, K. Y.; Wang, Y. X. CN 108017620 2017 [Chem. Abstr. 2017, 10, 747229] |
| [28] | Zhang, X. X.; Chen, T. P.; Zhu, G. Y. CN 113563310 2021 [Chem. Abstr. 2021, 10, 709318] |
/
| 〈 |
|
〉 |