

Chinese Journal of Organic Chemistry >
Recognition of Bis-thiourea Tweezers to Neutral Molecules in Non-Polar Solvent
Received date: 2023-06-18
Revised date: 2023-07-21
Online published: 2023-08-30
Supported by
National Natural Science Foundation of China(21961042); Program of Yulin Normal University(G2021ZK16); Yulin Normal University High-Level Talent Project(G2023ZK12); Program of Yulin Normal University(S202210606118)
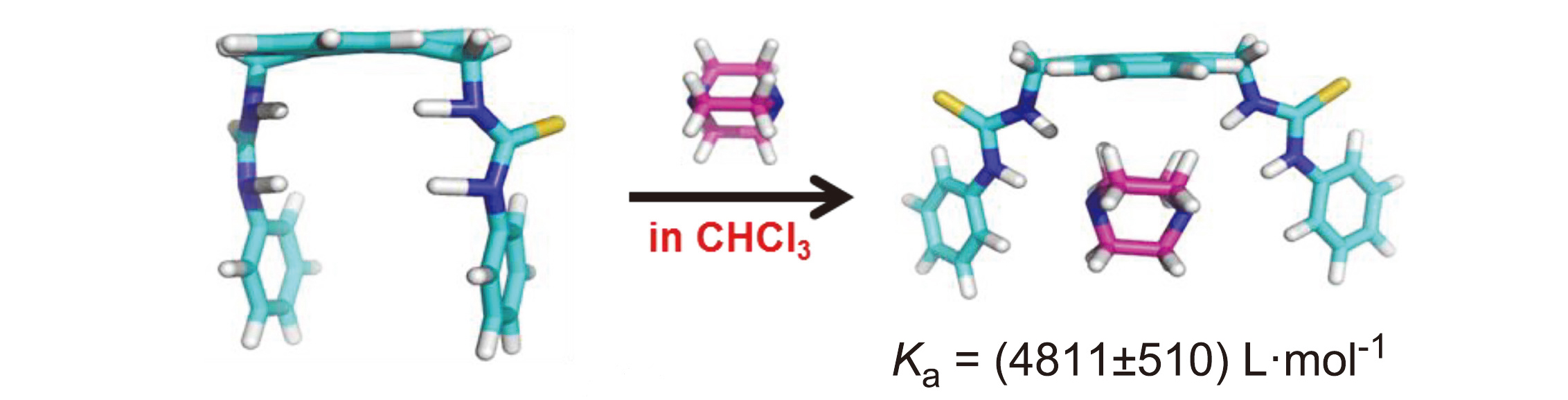
Three different molecular tweezers containing bis-thiourea groups were efficiently synthesized by a series of organic reactions using anthracene as raw materials. Their structures were confirmed and characterized by melting point, 1H NMR, 13C NMR and HRMS. The recognition behavior of molecular tweezers to several neutral molecules in non-polar environment was studied in detail by fluorescence spectroscopy. The results show that the molecular tweezer 9,10-bis- trifluoromethylphenyl-thioureamethyl anthracene (T-3) has good recognition ability for several guest molecules, and the synergistic effect between the hydrogen bonding and multiple C—H…π interactions on molecular recognition was discussed.
Key words: bis-thiourea tweezer; synthesis; recognition; neutral molecule
Shan Chen , Zhilin Chen , Qiong Hu , Yanshuang Meng , Yue Huang , Pingfang Tao , Liru Lu , Guobao Huang . Recognition of Bis-thiourea Tweezers to Neutral Molecules in Non-Polar Solvent[J]. Chinese Journal of Organic Chemistry, 2024 , 44(1) : 277 -281 . DOI: 10.6023/cjoc202306013
| [1] | Berthod, A. Anal. Chem. 2006, 78, 2093. |
| [2] | Pedersen, C. J. J. Am. Chem. Soc. 1967, 89, 7017. |
| [3] | Zhao, Z.-G.; Liu, X.-L.; Li, Q.-H.; Chen, S.-H. Chin. J. Org. Chem. 2009, 29, 1336 (in Chinese). |
| [3] | (赵志刚, 刘兴利, 李清寒, 陈淑华, 有机化学, 2009, 29, 1336.) |
| [4] | Salonen, L. M.; Ellermann, M.; Diederich, F. Angew. Chem., Int. Ed. 2011, 50, 4808. |
| [5] | Chen, C.-W.; Whitlock Jr., H. W. J. Am. Chem. Soc. 1978, 100, 4921. |
| [6] | Hardouin-Lerouge, M.; Hudhomme, P.; Salle, M. Chem. Soc. Rev. 2001, 40, 30. |
| [7] | Legouin, B.; Uriac, P.; Tomasi, S.; Toupet, L.; Bondon, A.; Vande Weghe, P. Org. Lett. 2009, 11, 745. |
| [8] | Colquhoun, H. M.; Zhu, Z.; Williams, D. J. Org. Lett. 2003, 5, 4353. |
| [9] | Canevet, D.; Sallé, M.; Zhang, G.; Zhang, D.; Zhu, D. Chem. Commun. 2009, 2245. |
| [10] | (a) Park, J.-S.; Le Derf, F.; Bejger, C.-M.; Lynch, V.-M.; Sessler, J.-L.; Nielsen, K.-A.; Johnsen, C.; Jeppesen, J.-O. Chem.-Eur. J. 2010, 16, 848. |
| [10] | (b) Dolensky, B.; Havlík, M.; Král, V. Chem. Soc. Rev. 2012, 41, 3839. |
| [10] | (c) Zimmerman, S. Beilstein J. Org. Chem. 2016, 12, 125. |
| [10] | (d) Kumar, R.; Srivastava, A. Chem.-Eur. J. 2016, 22, 3224. |
| [10] | (e) Fu, T.; Han, Y.; Ao, L.; Wang, F. Organometallics 2016, 35, 2850. |
| [10] | (f) Tsuchido, Y.; Suzaki, Y.; Ide, T.; Osakada, K. Chem.-Eur. J. 2014, 20, 4762. |
| [11] | (a) Gunnlaugsson, T.; Davis, A. P.; O'Briena, J. E.; Glynna, M. Org. Biomol. Chem. 2005, 3, 48. |
| [11] | (b) Zhang, Z.-Y.; Li, C. Acc. Chem. Res. 2022, 55, 916. |
| [11] | (c) Zhang, X; Wang, X.; Wang, B.; Ding, Z.; Li, C. Chin. Chem. Lett. 2020, 31, 3230. |
| [12] | (a) Huang, G.; He, Z.; Cai, C.; Pan, F.; Yang, D.; Rissanen, K.; Jiang, W. Chem. Commun. 2015, 15, 15490. |
| [12] | (b) Huang, G.; Valkonen, A.; Rissanen, K.; Jiang, W. Chem. Commun. 2016, 52, 9078. |
| [13] | Altava, B.; Burguete, M. I.; Escuder, B.; Luis, S. V.; García-Espa?a, E.; Mu?oz, M. C. Tetrahedron 1997, 53, 2629. |
/
| 〈 |
|
〉 |