

Chinese Journal of Organic Chemistry >
Copper-Based Solid Wastes Promoted Cross-Coupling Reactions of Terminal Alkynes
Received date: 2024-01-12
Revised date: 2024-03-07
Online published: 2024-03-28
Supported by
National Natural Science Foundation of China(21961037); National Natural Science Foundation of China(22161044); National Natural Science Foundation of China(22201241); National Natural Science Foundation of China(22361044); Tianshan Talents Program for Leading Talents in Science and Technology Innovation(2022TSYCLJ0016); Key Program of Natural Science Foundation of Xinjiang Uygur Autonomous Region(2022D01D06)
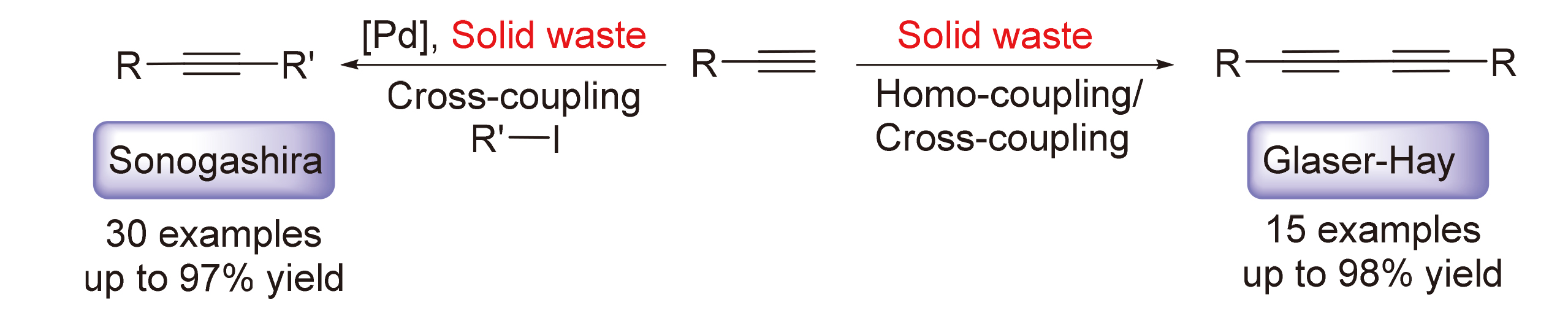
An approach to the Sonogashira and Glaser-Hay coupling reactions utilizing copper-based solid wastes instead of traditional copper salts has been developed. This method enables the efficient synthesis of various disubstituted alkynes under relatively mild conditions. For the Sonogashira coupling reaction, diverse substituted phenylacetylenes and aryl iodides exhibit excellent reactivity, resulting in diaryl acetylene compounds with yields ranging from 57% to 97%. The Glaser-Hay coupling reaction allows for the high-yield production of symmetrically disubstituted 1,3-butadiyne derivatives using a variety of terminal alkynes. Additionally, moderate yields can be achieved for cross-coupling products. Gram-scale and cycling experiments demonstrate promising prospects for the application of solid waste in organic synthesis.
Tong Xu , Ning Zhang , Yonghong Zhang , Bin Wang , Yu Xia , Weiwei Jin , Pinru Jin , Chenjiang Liu . Copper-Based Solid Wastes Promoted Cross-Coupling Reactions of Terminal Alkynes[J]. Chinese Journal of Organic Chemistry, 2024 , 44(7) : 2341 -2349 . DOI: 10.6023/cjoc202401012
| [1] | (a) Chinchilla, R.; Carmen Najera, C. Chem. Rev. 2014, 114, 1783. |
| [1] | (b) Stang, P. J.; Diederich, F. Modern Acetylene Chemistry, VCH, Weinheim, 1995. |
| [1] | (c) Diederich, F.; Stang, P. J. Acetylene Chemistry, Ed.: Tykwinski, R. R., Wiley-VCH, Weinheim, 2005. |
| [1] | (d) Feng, J.; Zhang, F; Shu, C.-Y.; Zhu, G.-G. Chin. J. Chem. 2022, 40, 1667. |
| [1] | (e) Hao, T.-G.; Shi, M.; Wei, Y. Chin. J. Chem. 2023, 41, 301. |
| [1] | (f) Batchu, V. R.; Subramanian, V.; Parasuraman, K.; Swamy, N. K.; Kumar, S.; Pal, M. Tetrahedron 2005, 61, 9869. |
| [2] | (a) Armstrong, K. M.; Lalic, G. J. Am. Chem. Soc. 2019, 141, 6173. |
| [2] | (b) Hamasaka, G.; Roy, D.; Tazawa, A.; Uozumi, Y. ACS Catal. 2019, 9, 11640. |
| [2] | (c) Liu, L.; Dan Zhou, D.; Liu, M.; Zhou, Y.-B.; Chen, T.-Q. Org. Lett. 2018, 20, 2741. |
| [2] | (d) Tian, Z.-Y.; Wang, S.-M.; Jia, S.-J.; Song, H.-X.; Zhang, C.-P. Org. Lett. 2017, 19, 5454. |
| [2] | (e) Liu, B.-Q.; Yan, Z.-F.; Quan, Z.-J. Chin. J. Org. Chem. 2018, 38, 3032. (in Chinese) |
| [2] | (刘伯渠, 燕中飞, 权正军, 有机化学, 2018, 38, 3032.) |
| [3] | (a) Ma, N.; Zeng, X.-H. Chin. J. Org. Chem. 2018, 38, 1556. (in Chinese) |
| [3] | (马楠, 曾祥华, 有机化学, 2018, 38, 1556.) |
| [3] | (b) Li, X.-W.; Liu, X.-H.; Chen, H.-J.; Wu, W.-Q.; Qi, C.-F.; Jiang, H.-F. Angew. Chem., Int. Ed. 2014, 53, 14485. |
| [3] | (c) Sakamoto, R.; Kato, T.; Sakurai, S.; Maruoka, K. Org. Lett. 2018, 20, 1400. |
| [3] | (d) Feng, L.-L.; Hu, T.-J.; Zhang, S.-S.; Xiong, H.-Y.; Zhang, G.-W. Org. Lett. 2019, 21, 9487. |
| [3] | (e) Lv, Y.-H.; Pu, W.-Y.; Shi, L.-H. Org. Lett. 2019, 21, 6034. |
| [3] | (f) Biswas, S.; Mullick, K.; Chen, S.-Y.; Kriz, D. A.; Shakil, M.; Kuo, C.-H.; Angeles-Boza, A. M.; Rossi, A. R.; Suib, S. L. ACS Catal. 2016, 6, 5069. |
| [4] | (a) Zhang, G.; Shao, X.-B.; Li, Q.-H.; Yang, X.-J. Chin. J. Org. Chem. 2018, 38, 1538. (in Chinese) |
| [4] | (张刚, 杓学蓓, 李清寒, 杨学军, 有机化学, 2018, 38, 1538.) |
| [4] | (b) Chen, H.; Yao, L.-C.; Guo, L.; Liu, Y. A.; Tian, B.-X.; Liao, X.-B. Cell Rep. Phys. Sci. 2023, 4, 101573. |
| [4] | (c) Li, Y.-Q.; Li, F.; Shi, S.-L. Chin. J. Chem. 2020, 38, 1035. |
| [5] | Islam, K. M. N. Renewable Sustainable Energy Rev. 2018, 81, 2472. |
| [6] | Martins, M. A. D. B.; Crispim, A.; Ferreira, M. L.; Dos Santos, I. F.; Melo, M. D. L. N.; Barros, R. M.; Filho, G. L. T. Cleaner Waste Systems 2023, 4, 100070 |
| [7] | Ding, Y.; Zhao, J.; Liu, J.; Zhou, J.; Cheng, L.; Zhao, J.; Shao, Z.; Iris, ?.; Pan, B.; Li, X.; Hu, Z. J. Clean. Prod. 2021, 293, 126144. |
| [8] | (a) Yu, H.-X.; Zahidi, I.; Liang, D.-F. J. Mater. Res. Technol. 2023, 23, 5733. |
| [8] | (b) Arenas, C.; Ríos, D. J.; Cifuentes, H.; Vilches, F. L.; Leiva, C. Eur. J. Environ. Civ. Eng. 2022, 9, 3805. |
| [8] | (c) Zhang, F.; Yu, W.; Liu, W.-Y.; Xu, Z.-Y. Front. Energy Res. 2020, 8, 50. |
| [8] | (d) Liu, B.-C.; Han, B.-R.; Liang, X.-Q.; Liu, Y.-F. Int. J. Hydrogen Energy 2024, 52, 1445. |
| [9] | (a) Dewan, A.; Sarmah, M.; Bora, U.; Thakur, A. J. Tetrahedron Lett. 2016, 57, 3760. |
| [9] | (b) Dewan, A.; Sarmah, M.; Bora, U.; Thakur, A. J. Appl. Organomet. Chem. 2017, 31, e3646. |
| [9] | (c) Dewan, A.; armah, M.; Thakur, A. J.; Bharali, P.; Bora, U. ACS Omega 2018, 3, 5327. |
| [9] | (d) Boruah, P.-R.; Ali, A.-A.; Saikia, B.; Sarma, D. Green Chem. 2015, 17, 1442. |
| [10] | (a) Isfahani, A. L.; Mohammadpoor-Baltork, I.; Mirkhani, V.; hosropour, A. R.; Moghadam, M.; Tangestaninejad, S. Eur. J. Org. Chem. 2014, 5603. |
| [10] | (b) Thathagar, M. B.; Beckers, J.; Rothenberg, G. Green Chem. 2004, 6, 215. |
| [10] | (c) Zhang, C.-T.; Peng, L.-J.; Song, B.-H.; Li, Z.-W.; Cao, X.-Q. Inorg. Chem. Commun. 2023, 158, 111471. |
| [10] | (d) Moghaddam, F. M.; Tavakoli, G.; Rezvani, H. R. Catal. Commun. 2015, 60, 82. |
| [10] | (e) Mohammadi, P.; Heravil, M. M.; Mohammadi, L.; Saljooqi, A. Sci. Rep. 2023, 13, 17375. |
| [11] | (a) Tang, S.-Y.; Li, L.-J.; Ren, X.-H.; Li, J.; Yang, G.-Y.; Li, H.; Yuan, B.-X. Green Chem. 2019, 21, 2899. |
| [11] | (b) Su, L.-B.; Dong, J.-Y.; Liu, L.; Sun, M.-L.; Qiu, R.-H.; Zhou, Y.-B.; Yin, S.-F. J. Am. Chem. Soc. 2016, 138, 12348. |
| [11] | (c) Kusuda, A.; Xu, X.-X.; Wang, X.; Tokunaga, E.; Shibata, N. Green Chem. 2011, 13, 843. |
| [11] | (d) Zhang, L.-Z.; Wei, C.-B.; Wu, J.-W.; Liu, D.; Yao, Y.-C.; Chen, Z.; Liu, J.-X.; Yao, C.-J.; Li, D.-H.; Yang, R.-J.; Xia, Z.-H. Chem. Sci. 2022, 13, 7475. |
| [12] | (a) Sun, Y.-J.; Wang, R.; Liu, T.-X.; Jin, W.-W.; Wang, B.; Zhang, Y.-H.; Xia, Y.; Liu, C.-J. Eur. J. Org. Chem. 2021, 2470. |
| [12] | (b) Sun, Y.-J.; Jin, W.-W.; Liu, C.-J. Molecules 2019, 24, 3838. |
| [13] | Denis, P.; Andy, W.; John, H.; Sneddon, H.; C. Robert, M.; Sarah, A. S.; Peter J. D. Green Chem. 2016, 18, 288. |
| [14] | Zhao, C.-Q.; Chen, Y.-G.; Qiu, H.; Wei, L.; Fang, P.; Mei, T.-S. Org. Lett. 2019, 21, 1412. |
| [15] | Stein, A.-L.; Bilheria, F.-N.; Zeni, G. Chem. Commun. 2015, 51, 15522. |
| [16] | (a) Qiu, S.-Z.; Zhang, C.-Y.; Qiu, R.; Yin, G.-D.; Huang, J.-K. Adv. Synth. Catal. 2018, 360, 313. |
| [16] | (b) Yu, S.-Y.; Wu, J.-X.; He, X.-W.; Shang, Y.-J. Appl. Organomet. Chem. 2018, 32, e4156. |
| [16] | (c) Topolov?an, N.; Hara, S.; Císa?ová, I.; To?ner, Z.; Kotora, M. Eur. J. Org. Chem. 2020, 2, 234. |
| [16] | (d) Jitendra, R. H.; Theodore, J. A.; Alwyn, T. G.; Garcia, M. T.; Robert, D. S.; Peter, J. S. Green Chem. 2010, 12, 650. |
| [16] | (e) Pati, A. K.; Mohapatra, M.; Ghosh, P.; Gharpure, S. J.; Mishra, A. K. J. Phys. Chem. A 2013, 117, 6548. |
| [16] | (f) Kurita, T.; Abe, M.; Maegawa, T.; Monguchi, Y.; Sajiki, H. Synlett 2007, 16, 2521. |
| [16] | (g) Chen, S.-N.; Wu, W.-Y.; Tsai, F.-Y. Green Chem. 2009, 11, 269. |
| [16] | (h) Batsanov, A. S.; Collings, J. C.; Fairlamb, I. J. S.; Holland, J. P.; Howard, J. A. K.; Lin, Z.-Y.; Marder, T. B.; Parsons, A. C.; Ward, R. M.; Zhu, J. J. Org. Chem. 2005, 70, 703. |
| [16] | (i) Li, X.; Li, D.-J.; Bai, Y.-N.; Zhang, C.-X.; Chang, H.-H.; Gao, W.-C.; Wei, W.-L. Tetrahedron 2016, 72, 6996. |
| [16] | (j) Zhang, S.-L.; Liu, X.-Y.; Wang, T.-Q. Adv. Synth. Catal. 2011, 353, 1463. |
| [16] | (k) Chinta, B. S.; Baire, B. RSC Adv. 2016, 6, 54449. |
/
| 〈 |
|
〉 |