

Chinese Journal of Organic Chemistry >
Copper Mediated sp3-C—N Bond Cleavage for Synthesizing (Diarylmethyl)diarylphosphine Oxides
Received date: 2024-05-31
Revised date: 2024-09-05
Online published: 2024-09-19
Supported by
National Natural Science Foundation of China(22378106); National Natural Science Foundation of China(21706058); National Natural Science Foundation of China(21878072); National Natural Science Foundation of China(22002169); Natural Science Foundation of Hunan Province(2020JJ2011); China Postdoctoral Science Foundation(2019M662774)
Organic phosphorus compounds containing sp3-C—P(O) bonds are increasingly widely applied in catalysis, pharmaceuticals, materials, pesticides, and other fields, and their synthesis has become a research hotspot in chemistry. Diarylmethyl phosphine oxides are important organic phosphorus compounds containing sp3-C—P(O) bonds, but their synthesis is limited. Traditional methods for their synthesis require the use of halogenated compounds and harsh reaction conditions. A new method for the copper-mediated synthesis of (diarylmethyl)diarylphosphine oxides has been developed. This method involves the cleavage of the sp3-C—N bond in N-diarylmethylsulfonamides, leading to the formation of diarylmethyl carbocations. The carbocations then react with diarylphosphine oxides to construct sp3-C—P(O) bonds. Our method only requires the addition of stoichiometric, inexpensive CuBr2 and produces a series of target compounds in satisfactory yields. Thus, it provides a convenient, and cost-effective pathway for the synthesis of diarylmethyl phosphine oxides.
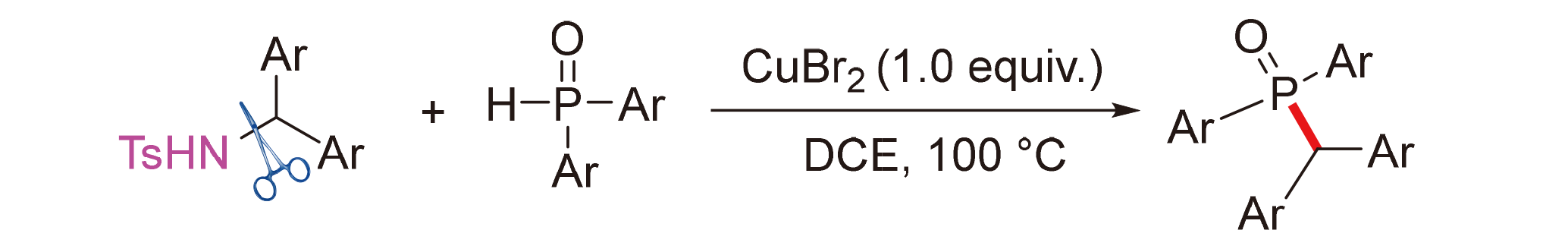
Jianyu Dong , Jie Huo , Ying Guo , Dan Zhou , Yongbo Zhou . Copper Mediated sp3-C—N Bond Cleavage for Synthesizing (Diarylmethyl)diarylphosphine Oxides[J]. Chinese Journal of Organic Chemistry, 2024 , 44(12) : 3713 -3719 . DOI: 10.6023/cjoc202405051
| [1] | (a) Baslé O.; Li C. J. Chem. Commun. 2009, 4124. |
| [1] | (b) Yang J.; Xiao J.; Zhou Y.; Chen T.; Yin S.; Han L. Chin. J. Org. Chem. 2017, 37, 1055 (in Chinese). |
| [1] | (杨佳, 肖晶, 周永波, 陈铁桥, 尹双凤, 韩立彪, 有机化学, 2017, 37, 1055.) |
| [1] | (c) Mo J. N.; Sun S.; Xu H.; Shu H.; Zhao J. Org. Lett. 2024, 26, 2197. |
| [1] | (d) Hirota E.; Hirashima S. I.; Morita R.; Takase J.; Matsushima Y.; Nakashima K.; Akutsu H.; Miura T. Org. Lett. 2024, 26, 1797. |
| [1] | (e) Xiong B.; Shi C.; Ren Y.; Xu W.; Liu Y.; Zhu L.; Cao F.; Tang K. W.; Yin S. F. J. Org. Chem. 2024, 89, 3033. |
| [1] | (f) Cheng Y.; Zhen J.; Chai L.; Wang J.; Yin J.; Zhu L.; Li C. Angew. Chem. Int. Ed. 2024, 63, e202316764. |
| [1] | (g) Yin Y.; Yang J.; Yan K.; Zeng T.; Lin H.; Ling J.; Wang S.; Wen J. Green Chem. 2024, 26, 832. |
| [2] | (a) Chen Q.; Wang X.; Yu G.; Wen C.; Huo Y. Org. Chem. Front. 2018, 5, 2652. |
| [2] | (b) Lv Y.; Xie J.; Pu W.; Wang X.; Zhang H.; Li X.; Liu Y.; Chen F.; Xu Y. Adv. Synth. Catal. 2023, 365, 4170. |
| [2] | (c) Liu L.; Liu M.; Liu B.; Wang Q.; Li Y.; Feng K.; Qiu R.; Zhou Y. Tetrahedron Lett. 2023, 133, 154823. |
| [2] | (d) Chen L.; Zhou Z.; Zhang S.; Li X.; Ma X.; Dong J. Chem. Commun. 2019, 55, 13693. |
| [3] | (a) Huang W. B.; Qiu L. Q.; Ren F. Y.; He L. N. Chem. Commun. 2021, 57, 9578. |
| [3] | (b) Reich D.; Noble A.; Aggarwal V. K. Angew. Chem. Int. Ed. 2022, 61, e202207063. |
| [3] | (c) Wang Y.; Wu X.; Yang L.; Liu W.; Zhang Z.; Xie X. Org. Biomol. Chem. 2023, 21, 2955. |
| [3] | (d) Backx S.; Dejaegere A.; Simoens A.; Van de Poel J.; Krasowska D.; Stevens C. V.; Mangelinckx S. Eur. J. Org. Chem. 2023, 26, e202300172. |
| [3] | (e) Ma C.; Li X.; Chen X.; He X.; Zhang S. T.; Jiang Y. Q.; Yu B. Org. Lett. 2023, 25, 8016. |
| [3] | (f) Guo Y.; Li N.; Li J.; Bi X.; Gao Z.; Duan Y. N.; Xiao J. Commun. Chem. 2023, 6, https://doi.org/10.1038/s42004-023-00826-4. |
| [4] | Yadav A.; Kumar D.; Mishra M. K.; Deeksha |
| [5] | (a) Hicks I.; McTague J.; Hapatsha T.; Teriak R.; Kaur P. Molecules 2020, 25, 290. |
| [5] | (b) Shen K.; Feng C.; Liu Y.; Yi D.; Lin P.; Li H.; Gong Y.; Wei S.; Fu Q.; Zhang Z. Org. Biomol. Chem. 2023, 21, 9341 |
| [6] | (a) Barney R. J.; Richardson R. M.; Wiemer D. F. J. Org. Chem. 2011, 76, 2875. |
| [6] | (b) Liu C.; Szostak M. Angew. Chem. Int. Ed. 2017, 56, 12718. |
| [6] | (c) Wen J.; Sun X.; Yan K.; Yan T.; Liu Z.; Li Y.; Yang J. Org. Chem. Front. 2024, 11, 796. |
| [6] | (d) Lei Z.; Zhang W.; Wu J. ACS Catal. 2023, 13, 16105. |
| [6] | (e) Li C. K.; Tao Z. K.; Shoberu A.; Zhang W.; Zou J. P. Org. Lett. 2022, 24, 6083. |
| [7] | (a) Zhang B.; Liu L.; Mao S.; Zhou M.; Wang H.; Li L. Eur. J. Org. Chem. 2019, 2019, 3898. |
| [7] | (b) Zhou X.; Wang J.; Shen Y.; Ma D.; Zhao Y.; Wu J. J. Org. Chem. 2023, 88, 17521. |
| [7] | (c) Zhang C.; Li Z.; Zhu L.; Yu L.; Wang Z.; Li C. J. Am. Chem. Soc. 2013, 135, 14082. |
| [7] | (d) Ahmed S.; Shafeeq Z.; Hussain F.; Ahmed Q. N. Chem. Commun. 2023, 59, 12334. |
| [7] | (e) Wang L. L.; Zhou H.; Cao Y. X.; Zhang C.; Ren Y. Q.; Li Z. L.; Gu Q. S.; Liu X. Y. Nat. Synth. 2023, 2, 430. |
| [8] | Aher Y. N.; Pawar A. B. Org. Biomol. Chem. 2019, 17, 7536. |
| [9] | (a) Niu M.; Fu H.; Jiang Y.; Zhao Y. Chem. Commun. 2007, 272. |
| [9] | (b) Rajeshwaran G. G.; Nandakumar M.; Sureshbabu R.; Mohanakrishnan A. K. Org. Lett. 2011, 13, 1270. |
| [10] | Pallikonda G.; Chakravarty M. Eur. J. Org. Chem. 2012, 2013, 944. |
| [11] | Lee K.-Y.; Lee H.-S.; Kim H.-S.; Kim J.-N. Bull. Korean Chem. Soc. 2008, 29, 1441. |
| [12] | Liu C.; Li M.; Yang C.; Tian S. Chem. Eur. J. 2008, 15, 793. |
| [13] | Liu C. R.; Yang F. L.; Jin Y. Z.; Ma X. T.; Cheng D. J.; Li N.; Tian S. K. Org. Lett. 2010, 12, 3832. |
| [14] | Yang C. F.; Wang J. Y.; Tian S. K. Chem. Commun. 2011, 47, 8343. |
| [15] | Liu C. R.; Wang T. T.; Qi Q. B.; Tian S. K. Chem. Commun. 2012, 48, 10913. |
| [16] | Hu C.; Hong G.; Qian X.; Kim K. R.; Zhu X.; Wang L. Org. Biomol. Chem. 2017, 15, 4984. |
| [17] | Dong J.; Liu L.; Ji X.; Shang Q.; Liu L.; Su L.; Chen B.; Kan R.; Zhou Y.; Yin S. F.; Han L. B. Org. Lett. 2019, 21, 3198. |
| [18] | Zhu M.; Liu J.; Yu J.; Chen L.; Zhang C.; Wang L. Org. Lett. 2014, 16, 1856. |
| [19] | Montel S.; Jia T.; Walsh P. J. Org. Lett. 2013, 16, 130. |
| [20] | Zhuang H.; Wan P.; Miao C.; Yang Y.; Liang S.; Han F. J. Org. Chem. 2024, 89, 2397. |
/
| 〈 |
|
〉 |