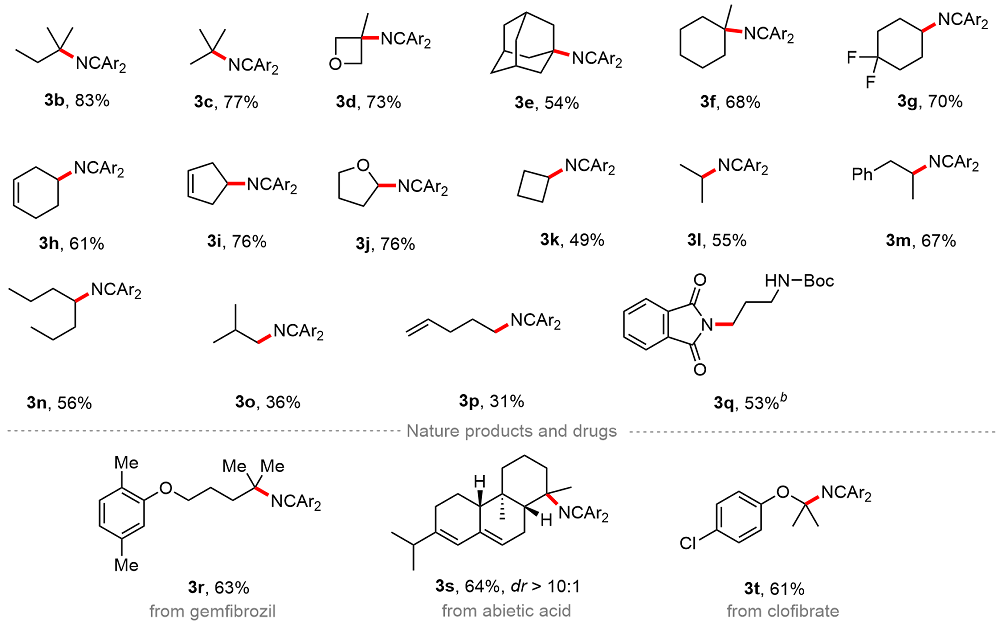
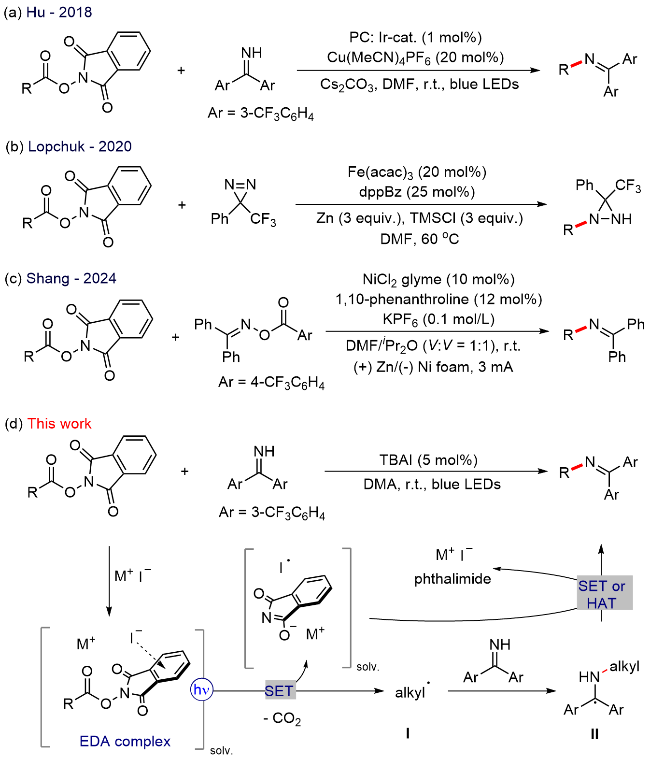
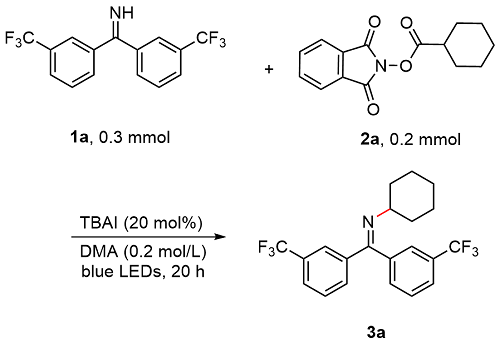
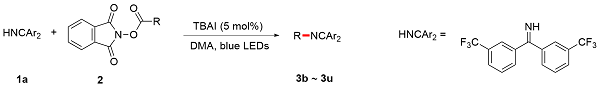
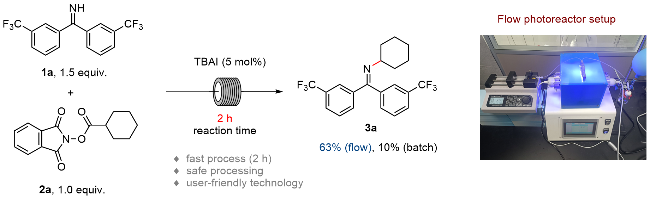
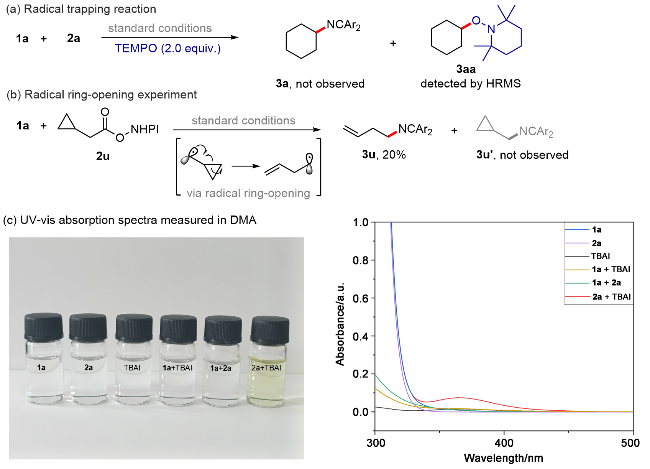
A 100 mL round bottom flask equipped with a magnetic stir bar was charged with 1,3-dioxoisoindolin-2-yl cyclohexanecarboxylate (5.0 mmol, 1.0 equiv.), bis(3-(trifluoro-methyl)phenyl)methanimine (7.5 mmol, 1.5 equiv.) and TBAI (0.25 mmol, 5 mol%). The reagents were dissolved in anhydrous DMA and the total volume of the solution was adjusted to 25 mL. The resulting mixture bubbled with an argon balloon for 20 min. After that, the reaction solution was introduced to the flow apparatus. The flow apparatus was purged with degassed argon to remove the air first. The syringe pump was then connected to the reaction mixture and 3.6 mL of polyfluoroalkoxy (PFA) microreactor coil (internal diameter of 1.0 mm) with a 34.5 kPa back-pressure regulator (BPR). The reaction was placed under 450 nm blue LEDs. The flow apparatus itself was set up with residence time (tR)=2 h, flow rate=1.8 mL/h. When the syringe was fully empty, a crude sample (1 mL) was taken from the collected solution and analyzed by determined through GC analysis. The yield of product 3 was determined to be 63%.
N-Cyclohexyl-1,1-bis(3-(trifluoromethyl)phenyl)meth-animine (
3a):
[16] 51.9 mg, 65% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.96 (s, 1H), 7.74 (d,
J=7.9 Hz, 1H), 7.67~7.59 (m, 3H), 7.46~7.41 (m, 2H), 7.37 (d,
J=7.6 Hz, 1H), 3.21~3.10 (m, 1H), 1.80~1.73 (m, 2H), 1.66~1.56 (m, 5H), 1.32~1.22 (m, 1H), 1.21~1.10 (m, 2H);
13C NMR (151 MHz, Chloroform-
d)
δ: 162.4, 140.3, 137.1, 131.7, 131.3, 131.0, 130.8, 129.3, 128.6, 126.5, 125.5, 124.7, 124.4, 124.1 (q,
J=272.5 Hz), 123.9 (q,
J=272.5 Hz), 61.9, 33.8, 25.5, 24.2;
19F NMR (564 MHz, Chloroform-
d)
δ: -63.0, -63.1.
N-
tert-Pentyl-1,1-bis(3-(trifluoromethyl)phenyl)methan-imine (
3b):
[9] 64.3 mg, 83% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.94 (s, 1H), 7.71 (d,
J=7.8 Hz, 1H), 7.64~7.53 (m, 3H), 7.48 (s, 1H), 7.40 (t,
J=9.2 Hz, 2H), 1.61 (q,
J=7.4 Hz, 2H), 1.03 (s, 6H), 0.97 (t,
J=7.4 Hz, 3H);
13C NMR (151 MHz, Chloroform-
d)
δ: 160.1, 142.0, 140.1, 131.6, 131.3, 130.7 (q,
J=32.7 Hz), 130.6 (q,
J=32.3 Hz), 128.7, 128.5, 126.2 (q,
J=3.6 Hz), 125.2 (q,
J=3.8 Hz), 125.0 (q,
J=3.6 Hz), 124.4 (q,
J=4.0 Hz), 124.1 (q,
J=272.3 Hz), 123.8 (q,
J=272.5 Hz), 60.0, 38.4, 28.3, 9.0;
19F NMR (564 MHz, Chloroform-
d)
δ: -62.7, -62.8.
N-
tert-Butyl-1,1-bis(3-(trifluoromethyl)phenyl)methani-mine (
3c):
[9] 57.4 mg, 77% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.95 (s, 1H), 7.71 (d,
J=7.9 Hz, 1H), 7.62~7.56 (m, 2H), 7.52 (d,
J=7.9 Hz, 1H), 7.48 (s, 1H), 7.44~7.36 (m, 2H), 1.17 (s, 9H);
13C NMR (151 MHz, Chloroform-
d)
δ: 160.1, 141.9, 139.8, 131.7, 131.4, 130.8 (q,
J=33.0 Hz), 130.6 (q,
J=32.7 Hz), 128.7, 128.5, 126.2 (q,
J=3.9 Hz), 125.2 (q,
J=4.0 Hz), 125.1 (q,
J=3.8 Hz), 124.4 (q,
J=4.2 Hz), 124.1 (q,
J=272.1 Hz), 123.8 (q,
J=272.2 Hz), 57.6, 31.5;
19F NMR (564 MHz, Chloroform-
d)
δ: -62.7, -62.8.
N-(3-methyloxetan-3-yl)-1,1-bis(3-(trifluoromethyl)phe-nyl)methanimine (3d): 56.5 mg, 73% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 7.94 (s, 1H), 7.75 (d, J=7.9 Hz, 1H), 7.67 (d, J=8.2 Hz, 1H), 7.62 (t, J=7.8 Hz, 1H), 7.56 (d, J=7.9 Hz, 1H), 7.49~7.43 (m, 2H), 7.41 (d, J=7.7 Hz, 1H), 4.54 (d, J=5.7 Hz, 2H), 4.02 (d, J=6.4 Hz, 2H), 1.74 (s, 3H); 13C NMR (151 MHz, Chloroform-d) δ: 162.5, 140.1, 137.7, 131.7, 131.3, 131.2 (q, J=33.1 Hz), 130.9 (q, J=32.6 Hz), 129.3, 128.8, 127.1 (q, J=3.7 Hz), 126.2 (q, J=4.0 Hz), 124.8 (q, J=3.5 Hz), 124.7 (q, J=3.8 Hz), 124.3 (q, J=272.5 Hz), 123.6 (q, J=272.7 Hz), 83.7, 62.2, 26.6; 19F NMR (564 MHz, Chloroform-d) δ: -62.7, -62.9. HRMS (ESI) calcd for C19H16F6NO [M+H]+ 388.1131, found 388.1129.
N-(Adamantan-1-yl)-1,1-bis(3-(trifluoromethyl)phenyl)-methanimine (
3e):
[9] 48.7 mg, 54% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.94 (s, 1H), 7.71 (d,
J=7.9 Hz, 1H), 7.62~7.54 (m, 2H), 7.51 (d,
J=7.9 Hz, 1H), 7.47 (s, 1H), 7.43~7.36 (m, 2H), 1.99 (s, 3H), 1.70 (s, 6H), 1.65~1.49 (m, 6H);
13C NMR (151 MHz, Chloroform-
d)
δ: 159.4, 142.0, 140.3, 131.5, 131.4, 130.6 (q,
J=32.3 Hz), 130.5 (q,
J=32.6 Hz), 128.6, 128.5, 126.2, 125.2, 124.9, 124.4, 124.0 (q,
J=272.5 Hz), 123.8 (q,
J=272.5 Hz), 58.7, 44.2, 36.2, 29.6;
19F NMR (564 MHz, Chloroform-
d)
δ: -62.6, -62.7.
N-(1-Methylcyclohexyl)-1,1-bis(3-(trifluoromethyl)-phenyl)methanimine (
3f):
[16] 56.2 mg, 68% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.95 (s, 1H), 7.71 (d,
J=7.9 Hz, 1H), 7.64~7.55 (m, 3H), 7.49 (s, 1H), 7.45~7.38 (m, 2H), 1.69 (d,
J=12.6 Hz, 2H), 1.61~1.53 (m, 3H), 1.50~1.43 (m, 2H), 1.31 (t,
J=10.2 Hz, 3H), 1.02 (s, 3H);
13C NMR (151 MHz, Chloroform-
d)
δ: 160.3, 142.0, 140.2, 131.3, 131.3, 130.7 (q,
J=32.5 Hz). 130.6 (q,
J=32.2 Hz), 128.7, 128.5, 126.2 (q,
J=3.6 Hz), 125.2 (q,
J=3.6 Hz), 124.7 (q,
J=3.9 Hz), 124.3 (q,
J=3.9 Hz), 124.0 (q,
J=272.5 Hz), 123.8 (q,
J=272.5 Hz), 59.3, 40.3, 28.1, 25.8, 22.8;
19F NMR (564 MHz, Chloroform-
d)
δ: -63.0, -63.1.
N-(4,4-Difluorocyclohexyl)-1,1-bis(3-(trifluoromethyl)-phenyl)methanimine (3g): 60.9 mg, 70% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 7.93 (s, 1H), 7.77 (d, J=7.9 Hz, 1H), 7.66 (q, J=7.9 Hz, 3H), 7.46 (t, J=7.8 Hz, 1H), 7.42 (s, 1H), 7.37 (d, J=7.5 Hz, 1H), 3.37~3.30 (m, 1H), 2.32~2.20 (m, 2H), 1.94~1.84 (m, 2H), 1.81~1.68 (m, 4H); 13C NMR (151 MHz, Chloroform-d) δ: 163.7, 139.8, 136.7, 131.7, 131.5 (q, J=33.0 Hz), 130.8 (q, J=32.9 Hz), 130.8, 129.6, 128.8, 126.9 (q, J=3.6 Hz), 125.8 (q, J=3.9 Hz), 124.7 (q, J=3.8 Hz), 124.1 (q, J=3.7 Hz), 124.0 (q, J=272.5 Hz), 123.9 (q, J=272.5 Hz), 57.9, 30.9 (t, J=24.5 Hz), 29.8 (t, J=4.9 Hz), 18.4; 19F NMR (564 MHz, Chloroform-d) δ: -65.3, -65.3, -99.2. HRMS (ESI) calcd for C21H18F8N [M+H]+ 436.1306, found 436.1306.
N-(Cyclohex-3-en-1-yl)-1,1-bis(3-(trifluoromethyl)phe-nyl)methanimine (3h): 48.4 mg, 61% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 7.97 (s, 1H), 7.74 (d, J=7.9 Hz, 1H), 7.63 (t, J=8.1 Hz, 3H), 7.50~7.42 (m, 2H), 7.38 (d, J=7.5 Hz, 1H), 5.71~5.60 (m, 2H), 3.47~3.33 (m, 1H), 2.40~2.27 (m, 1H), 2.17 (d, J=14.9 Hz, 1H), 2.03~1.82 (m, 3H), 1.74~1.65 (m, 1H); 13C NMR (151 MHz, Chloroform-d) δ: 163.2, 140.1, 137.0, 131.7, 131.3 (q, J=32.5 Hz), 131.0, 130.8 (q, J=32.4 Hz), 129.4, 128.7, 126.8, 126.6 (q, J=3.8 Hz), 125.6 (q, J=3.9 Hz), 124.8 (q, J=3.8 Hz), 124.6, 124.3 (q, J=4.0 Hz), 124.0 (q, J=272.6 Hz), 123.8 (q, J=272.6 Hz), 58.2, 32.4, 29.9, 23.7; 19F NMR (564 MHz, Chloroform-d) δ: -62.6, -62.7. HRMS (ESI) calcd for C21H17F6NNa [M+Na]+420.1157, found 420.1157.
N-(Cyclopent-3-en-1-yl)-1,1-bis(3-(trifluoromethyl)phe- nyl)methanimine (3i): 58.2 mg, 76% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 7.96 (s, 1H), 7.74 (d, J=7.9 Hz, 1H), 7.67~7.60 (m, 3H), 7.52~7.43 (m, 2H), 7.38 (d, J=7.6 Hz, 1H), 5.73 (s, 2H), 4.11~3.95 (m, 1H), 2.53 (d, J=6.4 Hz, 4H); 13C NMR (151 MHz, Chloroform-d) δ: 163.3, 140.1, 137.2, 131.8, 131.3, 131.3 (q, J=32.7 Hz), 130.8 (q, J=32.4 Hz), 129.3, 129.1, 128.7, 126.6 (q, J=4.0 Hz), 125.6 (q, J=3.8 Hz), 124.8 (q, J=3.8 Hz), 124.6 (q, J=3.6 Hz), 124.0 (q, J=272.5 Hz), 123.8 (q, J=272.5 Hz), 61.5, 41.4; 19F NMR (564 MHz, Chloroform-d) δ: -62.6, -62.8. HRMS (ESI) calcd for C20H16F6N [M+H]+ 384.1181, found 384.1200.
N-(Tetrahydrofuran-2-yl)-1,1-bis(3-(trifluoromethyl)-phenyl)methanimine (3j): 58.8 mg, 76% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 8.00 (s, 1H), 7.75 (d, J=7.9 Hz, 1H), 7.69~7.60 (m, 3H), 7.49~7.43 (m, 2H), 7.41 (d, J=7.7 Hz, 1H), 5.09 (dd, J=6.4, 3.7 Hz, 1H), 4.22 (q, J=7.4 Hz, 1H), 3.96~3.90 (m, 1H), 2.23~2.14 (m, 1H), 2.03~1.96 (m, 1H), 1.94~1.80 (m, 2H); 13C NMR (151 MHz, Chloroform-d) δ: 163.9, 139.5, 136.5, 132.1, 131.3, 131.3 (q, J=33.1 Hz), 130.9 (q, J=32.9 Hz), 129.4, 128.7, 127.2 (q, J=3.7 Hz), 126.0 (q, J=3.6 Hz), 125.2 (q, J=3.9 Hz), 124.7 (q, J=3.7 Hz), 124.0 (q, J=272.6 Hz), 123.8 (q, J=272.5 Hz), 91.5, 68.2, 34.1, 25.4; 19F NMR (564 MHz, Chloroform-d) δ: -62.7, -62.8. HRMS (ESI) calcd for C19H16F6NO [M+H]+ 388.1131, found 388.1125.
N-Cyclobutyl-1,1-bis(3-(trifluoromethyl)phenyl)metha-nimine (
3k):
[9] 36.4 mg, 49% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.97 (s, 1H), 7.74 (d,
J=8.0 Hz, 1H), 7.66~7.61 (m, 3H), 7.45 (t,
J=7.8 Hz, 1H), 7.38 (s, 1H), 7.32 (d,
J=7.6 Hz, 1H), 3.96~3.89 (m, 1H), 2.35~2.25 (m, 2H), 2.12~2.06 (m, 2H), 1.92~1.85 (m, 1H), 1.76~1.65 (m, 1H);
13C NMR (151 MHz, Chloroform-
d)
δ: 163.4, 140.0, 137.3, 131.7, 131.2 (q,
J=32.4 Hz), 131.1, 130.8 (q,
J=32.7 Hz), 129.2, 128.7, 126.7 (q,
J=3.8 Hz), 125.7 (q,
J=3.7 Hz), 124.8 (q,
J=4.0 Hz), 124.6 (q,
J=4.0 Hz), 124.0 (q,
J=272.4 Hz), 123.8 (q,
J=272.6 Hz), 57.4, 31.3, 16.1;
19F NMR (564 MHz, Chloroform-
d)
δ: -62.6, -62.8.
N-Isopropyl-1,1-bis(3-(trifluoromethyl)phenyl)methani-mine (
3l):
[9] 39.5 mg, 55% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.96 (s, 1H), 7.73 (d,
J=8.0 Hz, 1H), 7.66~7.59 (m, 3H), 7.46~7.41 (m, 2H), 7.37 (d,
J=7.6 Hz, 1H), 3.52~3.45 (m, 1H), 1.2 (s, 3H), 1.2 (s, 3H);
13C NMR (151 MHz, Chloroform-
d)
δ: 162.3, 140.2, 137.1, 131.7, 131.3 (q,
J=32.7 Hz), 131.0, 130.8 (q,
J=32.4 Hz), 129.4, 128.7, 126.6, 125.6, 124.8, 124.3 (q,
J=3.8 Hz), 124.0 (q,
J=272.7 Hz), 123.8 (q,
J=272.5 Hz), 53.5, 23.8;
19F NMR (564 MHz, Chloroform-
d)
δ: -62.6, -62.8.
N-(1-Phenylpropan-2-yl)-1,1-bis(3-(trifluoromethyl)-phenyl)methanimine (3m): 58.3 mg, 67% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 7.91 (s, 1H), 7.63 (d, J=7.8 Hz, 2H), 7.55 (d, J=7.8 Hz, 1H), 7.49~7.38 (m, 2H), 7.22~7.15 (m, 3H), 7.00 (d, J=6.4 Hz, 2H), 6.76 (d, J=35.7 Hz, 2H), 3.61~3.42 (m, 1H), 2.99~2.91 (m, 1H), 2.87~2.79 (m, 1H), 1.30 (d, J=6.2 Hz, 3H); 13C NMR (151 MHz, Chloroform-d) δ: 163.4, 139.9, 139.5, 137.0, 131.6, 131.0 (q, J=32.1 Hz), 130.7 (q, J=32.1 Hz), 130.8, 129.6, 129.0, 128.7, 128.2, 126.6 (q, J=4.1 Hz), 126.1, 125.3 (q, J=3.8 Hz), 124.7 (q, J=4.1 Hz), 124.1 (q, J=4.0 Hz), 124.0 (q, J=272.2 Hz), 123.7 (q, J=272.6 Hz), 60.3, 44.7, 22.2; 19F NMR (564 MHz, Chloroform-d) δ: -62.6, -62.6. HRMS (ESI) calcd for C24H20F6N [M+H]+ 436.1494, found 436.1494.
N-(Heptan-4-yl)-1,1-bis(3-(trifluoromethyl)phenyl)me-thanimine (3n): 46.5 mg, 56% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) 7.94 (s, 1H), 7.72 (d, J=8.7 Hz, 1H), 7.68~7.59 (m, 3H), 7.45 (t, J=7.8 Hz, 1H), 7.42 (s, 1H), 7.34 (d, J=7.6 Hz, 1H), 3.21 (dt, J=8.2, 4.6 Hz, 1H), 1.66~1.48 (m, 4H), 1.32~1.22 (m, 2H), 1.20~1.08 (m, 2H), 0.82 (t, J=7.3 Hz, 6H); 13C NMR (151 MHz, Chloroform-d) δ: 163.0, 140.2, 137.5, 131.6, 131.4, 131.1 (q, J=32.6 Hz), 130.8 (q, J=32.4 Hz), 129.2, 128.7, 126.5 (q, J=3.5 Hz), 125.3 (q, J=3.9 Hz), 124.8 (q, J=3.6 Hz), 124.7 (q, J=4.4 Hz), 124.0 (q, J=272.5 Hz), 123.9 (q, J=293.8 Hz), 62.2, 38.8, 19.7, 14.2; 19F NMR (564 MHz, Chloroform-d) δ: -62.6, -62.8. HRMS (ESI) calcd for C22H24F6N [M+H]+ 416.1807, found 416.1808.
N-Isobutyl-1,1-bis(3-(trifluoromethyl)phenyl)methani-mine (
3o):
[9] 26.9 mg, 36% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.95 (s, 1H), 7.74 (d,
J=7.9 Hz, 1H), 7.69~7.61 (m, 3H), 7.46 (t,
J=7.8 Hz, 1H), 7.41 (s, 1H), 7.36 (d,
J=7.6 Hz, 1H), 3.18 (d,
J=6.6 Hz, 2H), 2.11~2.02 (m, 1H), 0.94 (s, 3H), 0.93 (s, 3H);
13C NMR (151 MHz, Chloroform-
d)
δ: 164.6, 140.0, 137.0, 131.6, 131.3, 131.3 (q,
J=33.0 Hz), 130.8 (q,
J=32.4 Hz), 129.4, 128.7, 126.7, 125.6, 124.7, 124.6, 124.0 (q,
J=272.4 Hz), 123.7 (q,
J=253.8 Hz), 61.8, 30.1, 20.7;
19F NMR (564 MHz, Chloroform-
d)
δ: -62.7, -62.7.
N-(Pent-4-en-1-yl)-1,1-bis(3-(trifluoromethyl)phenyl)-methanimine (3p): 23.9 mg, 31% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 7.95 (s, 1H), 7.74 (d, J=7.9 Hz, 1H), 7.68~7.61 (m, 3H), 7.46 (t, J=7.8 Hz, 1H), 7.42 (s, 1H), 7.36 (d, J=7.6 Hz, 1H), 5.88~5.69 (m, 1H), 5.25~4.62 (m, 2H), 3.37 (t, J=6.9 Hz, 2H), 2.12 (q, J=6.7 Hz, 2H), 1.85~1.77 (m, 2H); 13C NMR (151 MHz, Chloroform-d) δ: 164.9, 140.0, 138.2, 136.8, 131.6, 131.4 (q, J=32.6 Hz), 131.2, 130.9 (q, J=32.5 Hz), 129.4, 128.7, 126.7 (q, J=3.8 Hz), 125.7 (q, J=3.8 Hz), 124.7 (q, J=4.2 Hz), 124.5 (q, J=4.0 Hz), 124.0 (q, J=272.5 Hz), 123.7 (q, J=253.8 Hz), 114.8, 53.4, 31.5, 30.2; 19F NMR (564 MHz, Chloroform-d) δ: -62.7, -62.7. HRMS (ESI) calcd for C20H18F6N [M+H]+ 386.1338, found 386.1338.
tert-Butyl (3-(1,3-dioxoisoindolin-2-yl)propyl)carba-mate (3q): 32.2 mg, 53% yield, pale yellow oil; 1H NMR (600 MHz, Chloroform-d) δ: 7.83 (dd, J=5.4, 3.0 Hz, 2H), 7.71 (dd, J=5.5, 3.0 Hz, 2H), 5.06 (s, 1H), 3.75 (t, J=6.6 Hz, 2H), 3.14~3.10 (m, 2H), 1.87~1.80 (m, 2H), 1.40 (s, 9H); 13C NMR (151 MHz, Chloroform-d) δ: 168.6, 155.9, 134.1, 132.0, 123.3, 79.2, 37.4, 35.1, 28.8, 28.4. HRMS (ESI) calcd for C16H21N2O4 [M+H]+ 305.1496, found 305.1491.
N-(5-(2,5-Dimethylphenoxy)-2-methylpentan-2-yl)-1,1-bis(3-(trifluoromethyl)phenyl)methan-imine (
3r):
[9] 65.7 mg, 63% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.92 (s, 1H), 7.73 (d,
J=7.8 Hz, 1H), 7.66~7.56 (m, 3H), 7.50 (s, 1H), 7.46~7.39 (m, 2H), 7.03 (d,
J=7.4 Hz, 1H), 6.68 (d,
J=7.5 Hz, 1H), 6.65 (s, 1H), 4.00 (t,
J=6.3 Hz, 2H), 2.32 (s, 3H), 2.22 (s, 3H), 2.03~1.94 (m, 2H), 1.89~1.76 (m, 2H), 1.10 (s, 6H);
13C NMR (151 MHz, Chloroform-
d)
δ: 160.4, 157.1, 141.8, 139.9, 136.5, 131.6, 131.3, 130.8 (q,
J=32.8 Hz), 130.6 (q,
J=32.3 Hz), 130.3, 128.8, 128.6, 126.3 (q,
J=3.6 Hz), 125.3 (q,
J=3.7 Hz), 125.0 (q,
J=3.9 Hz), 124.4 (q,
J=4.0 Hz), 124.1 (q,
J=272.6 Hz), 123.8 (q,
J=272.7 Hz), 123.6, 120.7, 112.1, 68.4, 59.6, 42.8, 28.8, 25.0, 21.4, 15.8;
19F NMR (564 MHz, Chloroform-
d)
δ: -62.6, -62.7.
N-((1R,4aR,4bR,10aR)-7-Isopropyl-1,4a-dimethyl-1,2,3,4,4a,4b,5,6,10,10a-decahydrophenanthren-1-yl)-1,1-bis-(3-(trifluoromethyl)phenyl)methanimine (3s): 73.4 mg, 64% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 7.78 (s, 1H), 7.69 (d, J=7.9 Hz, 1H), 7.57 (q, J=7.8 Hz, 3H), 7.44 (s, 1H), 7.41~7.36 (m, 2H), 5.84 (s, 1H), 5.53 (s, 1H), 2.51 (d, J=18.8 Hz, 1H), 2.28~2.20 (m, 1H), 2.10 (d, J=10.8 Hz, 2H), 2.06 (s, 1H), 1.99 (d,J=13.0 Hz, 1H), 1.80 (d, J=12.0 Hz, 2H), 1.52~1.37 (m, 4H), 1.30~1.20 (m, 2H), 1.14~1.07 (m, 1H), 1.03 (t, J=6.5 Hz, 6H), 0.97 (s, 3H), 0.82 (s, 3H); 13C NMR (151 MHz, Chloroform-d) δ: 159.0, 145.2, 142.3, 140.3, 135.4, 131.7, 131.2, 130.6 (q, J=32.4 Hz), 130.5 (q, J=32.5 Hz), 128.6, 128.4, 126.1 (q, J=3.8 Hz), 125.1 (q, J=4.0 Hz), 125.0 (q, J=4.7 Hz), 124.3 (q, J=4.0 Hz), 124.0 (q, J=272.4 Hz), 123.8 (q, J=272.7 Hz), 122.5, 121.8, 62.4, 52.9, 51.3, 40.4, 38.1, 35.5, 34.9, 27.6, 24.7, 22.7, 22.0, 21.4, 20.9, 19.4, 13.8; 19F NMR (564 MHz, Chloroform-d) δ: -62.6, -62.7. HRMS (ESI) calcd for C34H38F6N [M+H]+574.2903, found 574.2896.
N-(2-(4-Chlorophenoxy)propan-2-yl)-1,1-bis(3-(trifluo-romethyl)phenyl)methanimine (3t): 59.2 mg, 61% yield, colorless oil. 1H NMR (600 MHz, Chloroform-d) δ: 7.87 (s, 1H), 7.63 (d, J=7.7 Hz, 1H), 7.56 (dd, J=5.5, 3.0 Hz, 2H), 7.51~7.48 (m, 3H), 7.41 (t, J=7.8 Hz, 1H), 7.30 (d, J=7.6 Hz, 1H), 7.25 (s, 1H), 7.16 (t, J=7.8 Hz, 1H), 7.09 (d, J=7.9 Hz, 1H), 2.11 (s, 6H); 13C NMR (151 MHz, Chloroform-d) δ: 168.3, 159.5, 141.3, 137.1, 134.4, 133.7, 131.8, 131.1, 130.6 (q, J=32.5 Hz), 130.3 (q, J=33.0 Hz), 130.8, 128.6 (q, J=3.2 Hz), 126.8 (q, J=3.7 Hz), 125.1 (q, J=3.9 Hz), 124.8 (q, J=3.8 Hz), 124.0 (q, J=272.4 Hz), 123.9 (q, J=4.1 Hz), 123.9 (q, J=272.3 Hz), 122.4, 76.4, 30.4; 19F NMR (564 MHz, Chloroform-d) δ: -62.6, -63.0. HRMS (ESI) calcd for C24H19ClF6NO [M+H]+ 486.1054, found 486.1053.
N-(But-3-en-1-yl)-1,1-bis(3-(trifluoromethyl)phenyl)-methanimine (
3u):
[9] 14.8 mg, 20% yield, colorless oil.
1H NMR (600 MHz, Chloroform-
d)
δ: 7.95 (s, 1H), 7.74 (d,
J=7.9 Hz, 1H), 7.68~7.61 (m, 3H), 7.45 (t,
J=7.8 Hz, 1H), 7.43 (s, 1H), 7.36 (d,
J=7.6 Hz, 1H), 5.89~5.73 (m, 1H), 5.10~4.97 (m, 2H), 3.44 (t,
J=7.0 Hz, 2H), 2.47 (q,
J=7.4, 6.9 Hz, 2H);
13C NMR (151 MHz, Chloroform-
d)
δ: 165.1, 139.9, 136.7, 136.3, 131.6, 131.4 (q,
J=32.7 Hz), 130.9 (q,
J=32.6 Hz), 131.2, 129.4, 128.7, 126.8 (q,
J=3.9 Hz), 125.7 (q,
J=4.0 Hz), 124.7 (q,
J=4.0 Hz), 124.6 (q,
J=3.8 Hz), 124.0 (q,
J=272.4 Hz), 123.8 (q,
J=272.7 Hz), 116.2, 53.7, 35.4;
19F NMR (564 MHz, Chloroform-
d)
δ: -62.7, -62.7.
Supporting Information 1H NMR and
13C NMR spectra of compounds
3a~
3u, and the radical trapping experiments. The Supporting Information is available free of charge via the Internet at
http://sioc-journal.cn/.