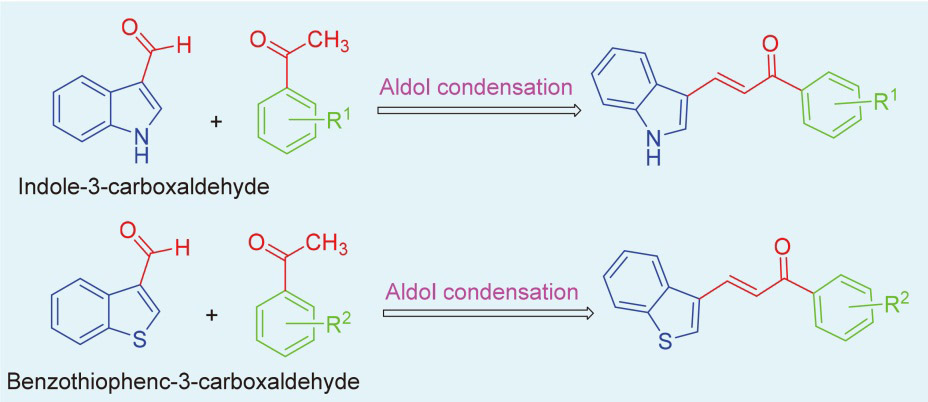
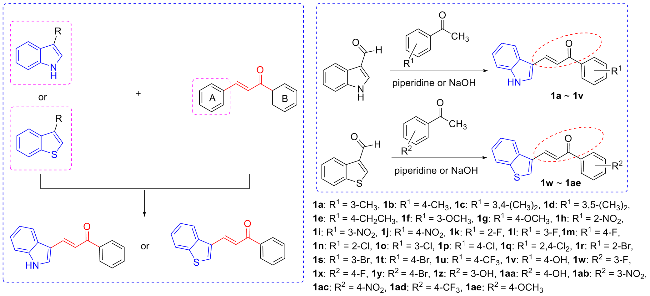
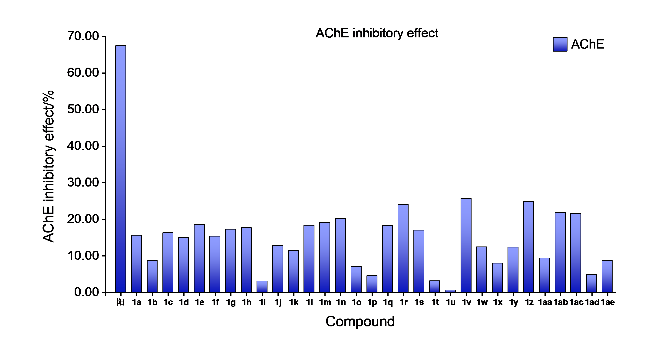
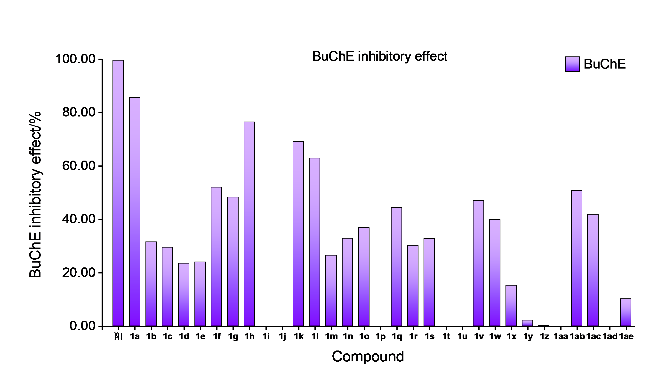
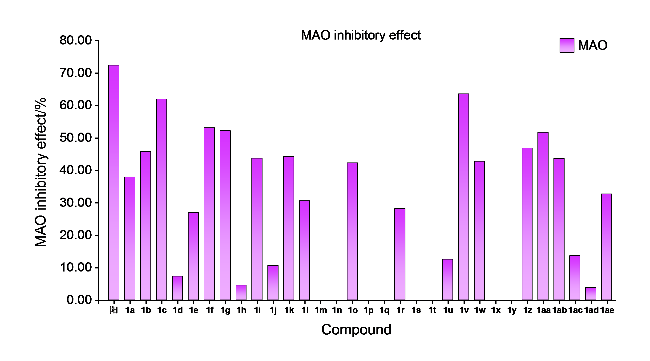
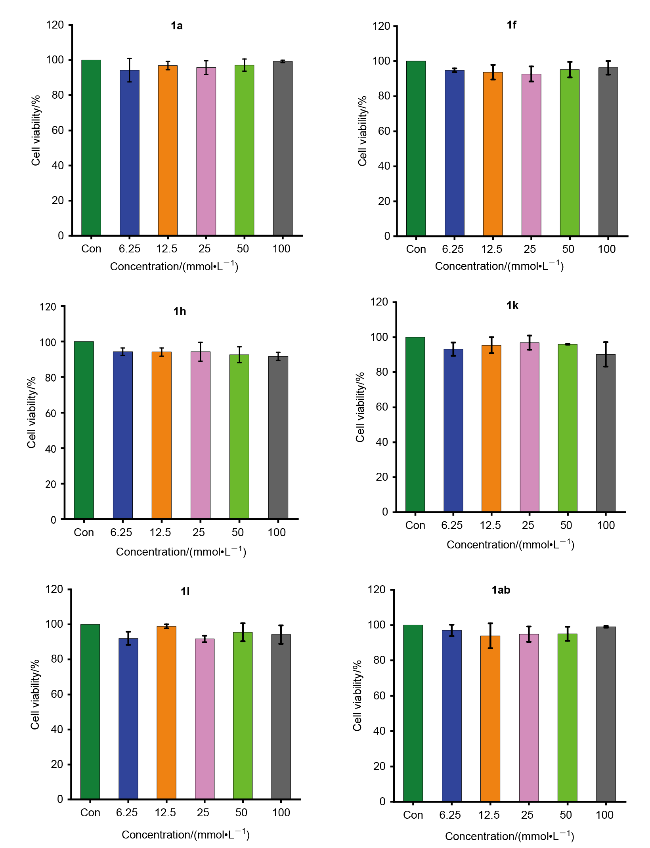
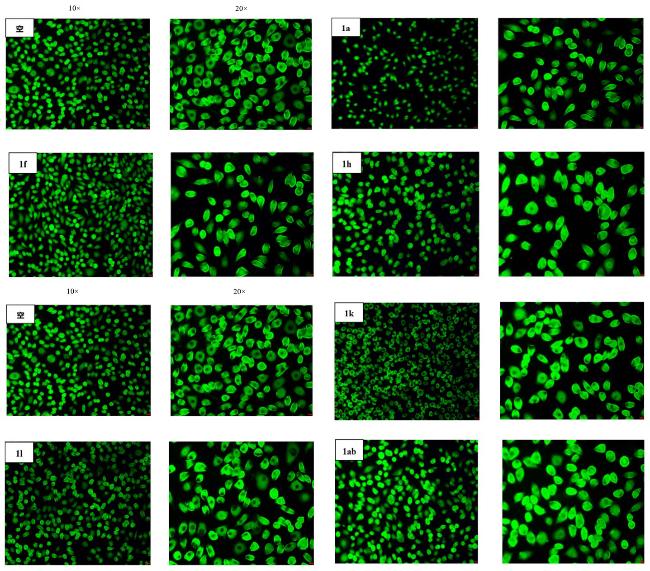
取哌啶1.3 g (1.5 mmol)和不同取代基的苯乙酮(3 mmol)于装有15 mL无水乙醇的圆底烧瓶中. 常温条件下搅拌4 h后, 加入0.435 g吲哚-3-甲醛(3 mmol), 待其全部溶解并反应稳定后, 加热回流过夜. 使用薄层色谱(TLC)监测反应进展, 待反应完成后, 冷却至室温, 将溶液倒入冰水中进行淬灭, 析出黄色固体. 过滤使固液分离, 用冷无水乙醇或正己烷洗涤漏斗中的滤饼2~3次, 洗涤后的滤饼用无水乙醇或甲醇进行重结晶以获得黄色固体产物.
(E)-3-(1H-吲哚-3-基)-1-(间甲苯基)丙-2-烯-1-酮 (1a): HPLC/Purity: 99.56% (tR=8.020 min), yield 41.13%. m.p. 143.2~144.5 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.92 (s, 1H, NH), 8.13 (s, 1H, NCH=C for indol ring), 8.06 (d, J=15.0 Hz, 1H, COCH=CH), 7.91~8.08 (m, 3H, C6H3), 7.61 (s, 1H, C6H1), 7.49 (d, J=15.0 Hz, 1H, COCH=CH), 7.22~7.55 (m, 4H, C6H4), 2.44 (s, 3H, CH3); 13C NMR (75 MHz, DMSO-d6) δ: 189.42, 139.36, 139.03, 138.51, 137.97, 133.62, 133.48, 129.04, 128.96, 125.86, 125.64, 123.17, 121.63, 120.82, 115.99, 113.24, 112.92, 21.46; HRMS calcd for C18H15NNaO [M+Na]+ 284.1051, found 284.1154.
(E)-3-(1H-吲哚-3-基)-1-(对甲苯基)丙-2-烯-1-酮 (1b): HPLC/Purity: 96.93% (tR=8.074 min), yield 53.13%. m.p. 167.3~167.4 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.90 (s, 1H, NH), 8.33 (s, 1H, NCH=C for indol ring), 8.10 (d, J=15.0 Hz, 1H, COCH=CH), 8.03~8.12 (m, 4H, C6H4), 7.49 (d, J=15.0 Hz, 1H, COCH=CH), 7.21~7.46 (m, 4H, C6H4), 2.41 (s, 3H, CH3); 13C NMR (75 MHz, DMSO-d6) δ: 188.80, 143.10, 139.12, 137.97, 136.39, 133.51, 129.73, 128.73, 125.63, 123.15, 121.60, 120.82, 115.87, 113.25, 112.91, 21.63; HRMS calcd for C18H16NO [M+H]+ 262.1232, found 262.1234.
(E)-1-(3,4-二甲基苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮(1c): HPLC/Purity: 97.31% (tR=10.642 min), yield 66.84%. m.p. 167.9~168.1 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.94 (s, 1H, NH), 8.53 (s, 1H, NCH=C for indol ring), 8.10 (d, J=15.0 Hz, 1H, COCH=CH), 7.82~8.13 (m, 3H, C6H3), 7.70 (d, J=15.0 Hz, 1H, COCH=CH), 7.25~7.51 (m, 4H, C6H4), 2.37 (s, 3H, CH3), 2.33 (s, 3H, CH3); 13C NMR (75 MHz, DMSO-d6) δ: 188.98, 141.92, 138.93, 137.96, 137.15, 136.79, 133.33, 130.18, 129.54, 126.35, 125.68, 123.13, 121.58, 120.80, 116.02, 113.27, 112.90, 20.04, 19.88; HRMS calcd for C19H18NO [M+H]+ 276.1388, found 276.1390.
(E)-1-(3,5-二甲基苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮(1d): HPLC/Purity: 98.86% (tR=9.708 min), yield 38.55%. m.p. 167.7~172.1 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.92 (s, 1H, NH), 8.14 (s, 1H, NCH=C for indol ring), 8.07 (d, J=15.0 Hz, 1H, COCH=CH), 7.73~8.13 (m, 3H, C6H3), 7.64 (d, J=15.0 Hz, 1H, COCH=CH), 7.24~7.53 (m, 4H, C6H4), 2.51 (s, 3H, CH3), 2.39 (s, 3H, CH3); 13C NMR (75 MHz, DMSO-d6) δ: 189.53, 139.17, 138.30, 137.96, 134.21, 133.43, 126.30, 125.68, 123.14, 121.60, 120.79, 116.14, 113.25, 112.90, 21.36; HRMS calcd for C19H17NNaO [M+Na]+ 298.1208, found 298.1205.
(E)-1-(4-乙基苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮 (1e): HPLC/Purity: 98.86% (tR=18.944 min), yield 52.10%. m.p. 165.4~165.5 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.91 (s, 1H, NH), 8.43 (s, 1H, NCH=C for indol ring), 8.06 (d, J=15.0 Hz, 1H, COCH=CH), 7.51~7.95 (m, 4H, C6H4), 7.55 (d, J=15.0 Hz, 1H, COCH=CH), 7.21~7.55 (m, 4H, C6H4), 2.73 (q, J=7.0 Hz, 2H, CH2), 1.32 (t, J=7.0 Hz, 3H, CH3); 13C NMR (75 MHz, DMSO-d6) δ: 188.90, 149.19, 139.12, 137.98, 136.69, 133.52, 128.82, 128.55, 125.63, 123.16, 121.61, 120.82, 115.96, 113.25, 112.92, 28.66, 15.76; HRMS calcd for C19H18NO [M+H]+ 276.1388, found 276.1393.
(E)-3-(1H-吲哚-3-基)-1-(3-甲氧基苯基)丙-2-烯-1-酮(1f): HPLC/Purity: 98.17% (tR=17.669 min), yield 79.40%. m.p. 130.5~132.4 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.96 (s, 1H, NH), 8.37 (s, 1H, NCH=C for indol ring), 8.10 (d, J=15.0 Hz, 1H, COCH=CH), 7.76~8.16 (m, 4H, C6H4), 7.76 (d, J=15.0 Hz, 1H, COCH=CH), 7.21~7.59 (m, 4H, C6H4), 3.87 (s, 3H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 189.10, 159.96, 140.51, 139.58, 137.98, 133.70, 130.31, 125.66, 123.20, 121.68, 121.10, 120.80, 118.78, 115.97, 113.26, 113.16, 112.94, 55.76; HRMS calcd for C18H16NO2 [M+H]+ 278.1181, found 278.1184.
(E)-3-(1H-吲哚-3-基)-1-(4-甲氧基苯基)丙-2-烯-1-酮(1g): HPLC/Purity: 98.12% (tR=7.872 min), yield 91.08%. m.p. 173.1~174.3 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.91 (s, 1H, NH), 8.46 (s, 1H, NCH=C for indol ring), 8.07 (d, J=15.0 Hz, 1H, COCH=CH), 7.72~8.18 (m, 4H, C6H4), 7.57 (d, J=15.0 Hz, 1H, COCH=CH), 7.09~7.52 (m, 4H, C6H4), 3.87 (s, 3H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 187.71, 163.15, 138.61, 137.96, 133.24, 131.72, 130.89, 125.66, 123.12, 121.54, 120.82, 115.84, 114.37, 113.27, 112.90, 55.93, 55.76; HRMS calcd for C18H16NO2 [M+H]+ 278.1181, found 278.1186.
(E)-3-(1H-吲哚-3-基)-1-(2-硝基苯基)丙-2-烯-1-酮 (1h): HPLC/Purity: 91.17% (tR=6.053 min), yield 50.53%. m.p. 162.6~164.1 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 12.00 (s, 1H, NH), 8.29 (s, 1H, NCH=C for indol ring), 8.13 (d, J=15.0 Hz, 1H, COCH=CH), 7.78~8.18 (m, 4H, C6H4), 7.74 (d, J=15.0 Hz, 1H, COCH=CH), 7.04~7.51 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 191.95, 147.62, 142.00, 138.08, 136.53, 134.52, 131.41, 129.58, 125.31, 124.92, 123.40, 121.89, 120.70, 119.67, 113.06, 112.62; HRMS calcd for C17H13- N2O3 [M+H]+ 293.0926, found 293.0929.
(E)-3-(1H-吲哚-3-基)-1-(3-硝基苯基)丙-2-烯-1-酮 (1i): HPLC/Purity: 100.00% (tR=16.818 min), yield 76.52%. m.p. 204.7~205.1 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 12.05 (s, 1H, NH), 8.76 (s, 1H, NCH=C for indol ring), 8.50 (d, J=15.0 Hz, 1H, COCH=CH), 8.13~8.62 (m, 4H, C6H4), 7.70 (d, J=15.0 Hz, 1H, COCH=CH), 7.25~7.72 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 187.34, 148.59, 141.11, 140.21, 138.05, 134.84, 132.15, 130.91, 127.00, 125.62, 123.37, 122.89, 121.83, 120.95, 114.94, 113.38, 113.02; HRMS calcd for C17H12N2NaO3 [M+Na]+ 315.0746, found 315.0745.
(E)-3-(1H-吲哚-3-基)-1-(4-硝基苯基)丙-2-烯-1-酮 (1j): HPLC/Purity: 99.45% (tR=8.017 min), yield 81.26%. m.p. 227.1~228.4 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 12.05 (s, 1H, NH), 8.76 (s, 1H, NCH=C for indol ring), 8.17 (d, J=15.0 Hz, 1H, COCH=CH), 8.07~8.39 (m, 4H, C6H4), 7.70 (d, J=15.0 Hz, 1H, COCH=CH), 7.24~7.62 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.15, 149.82, 144.04, 141.25, 138.09, 134.91, 129.91, 125.53, 124.23, 123.41, 121.87, 121.02, 115.41, 113.40, 113.05; HRMS calcd for C17H13N2O3 [M+H]+ 293.0926, found 293.0925.
(E)-1-(2-氟苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮(1k): HPLC/Purity: 98.18% (tR=8.759 min), yield 48.53%. m.p. 149.7~150.1 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.97 (s, 1H, NH), 8.09 (s, 1H, NCH=C for indol ring), 7.63~7.92 (m, 4H, C6H4), 7.90 (d, J=15.0 Hz, 1H, COCH=CH), 7.76 (d, J=15.0 Hz, 1H, COCH=CH), 7.22~7.49 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.75, 140.54, 138.12, 134.57, 133.94, 130.83, 128.13, 125.37, 123.34, 121.86, 120.55, 120.02, 119.96, 117.14, 116.84, 113.10, 112.90; HRMS calcd for C17H13FNO [M+H]+ 266.0981, found 266.0988.
(E)-1-(3-氟苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮(1l): HPLC/Purity: 99.40% (tR=7.414 min), yield 58.27%. m.p. 177.5~178.6 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 12.00 (s, 1H, NH), 8.53 (s, 1H, NCH=C for indol ring), 8.15 (d, J=15.0 Hz, 1H, COCH=CH), 7.88~8.19 (m, 4H, C6H4), 7.68 (d, J=15.0 Hz, 1H, COCH=CH), 7.24~7.65 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 187.98, 164.45, 161.21, 141.38, 138.00, 134.17, 131.35, 125.62, 124.80, 123.27, 121.72, 120.99, 119.84, 115.37, 114.90, 113.32, 112.94; HRMS calcd for C17H12FNNaO [M+Na]+ 288.0801, found 288.0804.
(E)-1-(4-氟苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮 (1m): HPLC/Purity: 94.68% (tR=8.118 min), yield 72.85%. m.p. 182.4~184.1 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.97 (s, 1H, NH), 8.49 (s, 1H, NCH=C for indol ring), 8.11 (d, J=15.0 Hz, 1H, COCH=CH), 7.98~8.27 (m, 4H, C6H4), 7.68 (d, J=15.0 Hz, 1H, COCH=CH), 7.26~7.54 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 187.82, 166.77, 163.45, 139.75, 138.01, 135.60, 133.91, 131.56, 125.61, 123.23, 121.66, 120.93, 116.23, 115.49, 113.69, 113.28, 112.94; HRMS calcd for C17H13FNO [M+H]+ 266.0981, found 266.0986.
(E)-1-(2-氯苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮(1n): HPLC/Purity: 100.00% (tR=11.820 min), yield 41.03%. m.p. 157.1~157.4 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.99 (s, 1H, NH), 8.35 (s, 1H, NCH=C for indol ring), 7.65 (d, J=15.0 Hz, 1H, COCH=CH), 7.58~8.05 (m, 4H, C6H4), 7.54 (d, J=15.0 Hz, 1H, COCH=CH), 7.02~7.59 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 193.37, 142.11, 140.11, 138.11, 134.44, 131.62, 130.41, 130.27, 129.53, 127.79, 125.33, 123.37, 121.91, 120.74, 120.56, 113.07, 112.68; HRMS calcd for C17H12ClNNaO [M+Na]+ 304.0505, found 304.0508.
(E)-1-(3-氯苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮 (1o): HPLC/Purity: 100.00% (tR=20.879 min), yield 88.61%. m.p. 166.8~168.0 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.96 (s, 1H, NH), 8.38 (s, 1H, NCH=C for indol ring), 7.90 (d, J=15.0 Hz, 1H, COCH=CH), 7.83~8.18 (m, 4H, C6H4), 7.64 (d, J=15.0 Hz, 1H, COCH=CH), 7.22~7.58 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 187.97, 140.90, 140.37, 137.98, 134.17, 132.55, 131.14, 128.13, 127.31, 125.64, 123.28, 121.73, 120.93, 115.31, 113.32, 112.95; HRMS calcd for C17H12Cl- NNaO [M+Na]+ 304.0505, found 304.0507.
(E)-1-(4-氯苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮 (1p): HPLC/Purity: 100.00% (tR=22.183 min), yield 68.15%. m.p. 187.5~187.8 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.99 (s, 1H, NH), 8.19 (s, 1H, NCH=C for indol ring), 7.65 (d, J=15.0 Hz, 1H, COCH=CH), 7.58~8.05 (m, 4H, C6H4), 7.54 (d, J=15.0 Hz, 1H, COCH=CH), 7.02~7.59 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.12, 140.09, 138.03, 137.74, 137.63, 134.10, 130.53, 129.21, 125.61, 123.26, 121.70, 120.94, 115.39, 113.32, 112.97, 56.54, 19.04; HRMS calcd for C17H12ClNNaO [M+Na]+ 304.0505, found 304.0506.
(E)-1-(2,4-二氯苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮 (1q): HPLC/Purity: 99.11% (tR=11.361 min), yield 53.69%. m.p. 191.7~192.8 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 12.01 (s, 1H, NH), 8.38 (s, 1H, NCH=C for indol ring), 7.67 (d, J=15.0 Hz, 1H, COCH=CH), 7.58~8.07 (m, 3H, C6H3), 7.26 (d, J=15.0 Hz, 1H, COCH=CH), 7.01~7.49 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 192.43, 142.71, 138.92, 138.14, 135.32, 134.80, 131.55, 130.95, 129.96, 128.04, 125.31, 123.42, 121.95, 120.68, 120.42, 113.09, 112.77; HRMS calcd for C17H11Cl2NNaO [M+Na]+ 338.0115, found 338.0117.
(E)-1-(2-溴苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮(1r): HPLC/Purity: 100.00% (tR=11.913 min), yield 27.75%. m.p. 172.0~172.3 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.99 (s, 1H, NH), 8.35 (s, 1H, NCH=C for indol ring), 7.62 (d, J=15.0 Hz, 1H, COCH=CH), 7.73~8.05 (m, 4H, C6H4), 7.54 (d, J=15.0 Hz, 1H, COCH=CH), 6.99~7.49 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 194.33, 142.32, 142.16, 138.12, 134.42, 133.48, 131.64, 129.41, 128.24, 125.33, 123.38, 121.92, 120.56, 119.13, 113.07, 112.70; HRMS calcd for C17H12BrNNaO [M+ Na]+ 348.0000, found 348.0001.
(E)-1-(3-溴苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮(1s): HPLC/Purity: 98.68% (tR=8.661 min), yield 78.62%. m.p. 173.4~173.6 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 12.05 (s, 1H, NH), 8.36 (s, 1H, NCH=C for indol ring), 8.54 (d, J=15.0 Hz, 1H, COCH=CH), 7.59~8.05 (m, 4H, C6H4), 7.44 (d, J=15.0 Hz, 1H, COCH=CH), 7.00~7.47 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 194.34, 142.35, 142.16, 138.12, 134.45, 133.48, 131.64, 129.42, 128.24, 125.33, 123.38, 121.92, 120.56, 119.14, 113.08, 112.71; HRMS calcd for C17H12BrNNaO [M+ Na]+ 348.0000, found 348.0003.
(E)-1-(4-溴苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮 (1t): HPLC/Purity: 99.66% (tR=10.838 min), yield 53.70 %. m.p. 200.2~202.3 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 12.00 (s, 1H, NH), 8.30 (s, 1H, NCH=C for indol ring), 8.08~8.17 (m, 4H, C6H4), 8.11 (d, J=15.0 Hz, 1H, COCH=CH), 7.66 (d, J=15.0 Hz, 1H, COCH=CH), 7.26~7.55 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.34, 140.14, 138.03, 137.96, 134.12, 132.16, 130.67, 126.84, 125.60, 123.28, 121.72, 120.92, 115.37, 113.32, 112.98, 56.55, 19.03; HRMS calcd for C17H12BrNNaO [M+Na]+ 348.0000, found 348.0004.
(E)-3-(1H-吲哚-3-基)-1-(4-(三氟甲基)苯基)丙-2-烯- 1-酮(1u): HPLC/Purity: 99.57% (tR=11.230 min), yield 77.22%. m.p. 195.3~196.1 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 12.01 (s, 1H, NH), 8.49 (s, 1H, NCH=C for indol ring), 7.92~8.32 (m, 4H, C6H4), 8.14 (d, J=15.0 Hz, 1H, COCH=CH), 7.66 (d, J=15.0 Hz, 1H, COCH=CH), 7.26~7.54 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.64, 142.39, 140.82, 138.07, 134.58, 132.44, 129.36, 126.23, 126.10, 126.06, 125.55, 123.35, 122.62, 121.81, 120.97, 115.52, 113.02, 56.51; HRMS calcd for C18H12F3NNaO [M+Na]+ 338.0769, found 338.0773.
(E)-1-(4-羟基苯基)-3-(1H-吲哚-3-基)丙-2-烯-1-酮 (1v): HPLC/Purity: 90.54% (tR=5.619 min), yield 44.70%. m.p. 190.4~194.6 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 11.88 (s, 1H, NH), 9.96 (s, 1H, OH), 8.29 (s, 1H, NCH=C for indol ring), 8.08 (d, J=15.0 Hz, 1H, COCH=CH), 7.79~8.13 (m, 4H, C6H4), 7.55 (d, J=15.0 Hz, 1H, COCH=CH), 6.92~7.70 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 187.53, 185.47, 162.30, 138.11, 137.94, 132.96, 131.13, 130.19, 125.65, 123.94, 123.06, 122.61, 121.48, 120.79, 115.96, 115.82, 113.27, 112.86; HRMS calcd for C17H14NO2 [M+H]+ 264.1025, found 264.1030.
将不同取代基的苯乙酮(3 mmol, 0.12 g)和氢氧化钠(3 mmol)溶解于15 mL无水乙醇中, 并在室温下搅拌1 h. 称取苯并噻吩-3-甲醛(3 mmol, 0.486 g)添加到圆底烧瓶中, 待全部溶解并使反应稳定后, 将烧瓶移至65 ℃的油浴中, 加热回流过夜反应. 使用薄层色谱法监测反应进程. 当TLC完全跟踪反应后, 将圆底烧瓶从油浴中取出. 待其冷却至室温后, 将溶液倒入冰水中进行淬火, 析出黄色固体. 过滤使固液分离, 用冷无水乙醇洗涤漏斗中的滤饼2~3次, 洗涤后的滤饼用三氯甲烷或甲醇进行重结晶, 以获得干净的黄色固体.
(E)-3-(苯并[b]噻吩-3-基)-1-(3-氟苯基)丙-2-烯-1-酮(1w): HPLC/Purity: 96.60% (tR=14.74 min), yield 28.62%. m.p. 93.0~94.0 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 8.26 (s, 1H, SCH=C for thiophen ring), 8.06~8.23 (m, 4H, C6H4), 8.13 (d, J=15.0 Hz, 1H, COCH=CH), 7.56 (d, J=15.0 Hz, 1H, COCH=CH), 7.47~7.96 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.41, 164.46, 161.21, 140.38, 140.30, 137.52, 136.22, 131.99, 126.53, 125.76,124.31, 123.80, 123.62, 122.67, 120.38, 115.60, 115.30; HRMS calcd for C17H11FNaOS [M+Na]+ 305.0412, found 305.0414.
(E)-3-(苯并[b]噻吩-3-基)-1-(4-氟苯基)丙-2-烯-1-酮(1x): HPLC/Purity: 96.55% (tR=22.809 min), yield 69.39%. m.p. 120.4~120.5 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 8.67 (s, 1H, SCH=C for thiophen ring), 8.06~8.30 (m, 4H, C6H4), 8.26 (d, J=15.0 Hz, 1H, COCH=CH), 7.50 (d, J=15.0 Hz, 1H, COCH=CH), 7.40~7.55 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.15, 140.37, 137.52, 135.78, 132.04, 131.90, 131.35, 125.75, 125.66, 123.81, 122.67, 122.58, 116.47, 116.18; HRMS calcd for C17H11FNaOS [M+Na]+ 305.0412, found 305.0416.
(E)-3-(苯并[b]噻吩-3-基)-1-(4-溴苯基)丙-2-烯-1-酮(1y): HPLC/Purity: 98.07% (tR=19.586 min), yield 27.74%. m.p. 132.7~134.6 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 8.67(s, 1H, SCH=C for thiophen ring), 8.10~8.25 (m, 4H, C6H4), 8.09 (d, J=15.0 Hz, 1H, COCH=CH), 7.55 (d, J=15.0 Hz, 1H, COCH=CH), 7.46~7.83 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.74, 140.37, 137.50, 137.07, 136.10, 132.37, 132.01, 131.59, 131.00, 127.78, 125.77, 125.68, 123.82, 122.68, 122.44; HRMS calcd for C17H12BrOS [M+H]+ 342.9792, found 342.9787.
(E)-3-(苯并[b]噻吩-3-基)-1-(3-硝基)丙-2-烯-1-酮(1z): HPLC/Purity: 98.21% (tR=13.432 min), yield 71.8%. m.p. 147.9~148.9 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 8.73 (s, 1H, SCH=C for thiophen ring), 8.52~8.83 (m, 4H, C6H4), 8.49 (d, J=15.0 Hz, 1H, COCH=CH), 7.55 (d, J=15.0 Hz, 1H, COCH=CH), 7.47~7.92 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 187.85, 152.29, 147.92, 144.38, 138.41, 138.11, 133.38, 132.92, 132.32, 125.82, 125.70, 124.38, 123.65, 121.74, 124.52; HRMS calcd for C17H12NO3S [M+H]+ 310.0538, found 310.0529.
(E)-3-(苯并[b]噻吩-3-基)-1-(4-硝基)丙-2-烯-1-酮 (1aa): HPLC/Purity: 99.49% (tR=13.432 min), yield 73.65%. m.p. 171.7~172.7 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 8.71 (s, 1H, SCH=C for thiophen ring), 8.13~8.42 (m, 4H, C6H4), 8.12 (d, J=15.0 Hz, 1H, COCH=CH), 7.54 (d, J=15.0 Hz, 1H, COCH=CH), 7.47~7.55 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 188.85, 150.29, 142.92, 140.38, 137.41, 137.11, 132.38, 131.92, 130.32, 125.82, 125.75, 124.38, 123.85, 122.74, 122.52; HRMS calcd for C17H12NO3S [M+H]+ 310.0538, found 310.0539.
(E)-3-(苯并[b]噻吩-3-基)-1-(3-羟基苯基)丙-2-烯-1-酮(1ab): HPLC/Purity: 97.04% (tR=8.231 min), yield 65.49%. m.p. 168.5~170.9 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 10.14 (s, 1H, OH), 8.65 (s, 1H, SCH=C for thiophen ring), 7.91~8.23 (m, 4H, C6H4), 8.06 (d, J=15.0 Hz, 1H, COCH=CH), 7.55 (d, J=15.0 Hz, 1H, COCH=CH), 7.08~7.67 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 189.53, 158.24, 140.38, 139.54, 137.56, 135.43, 132.05, 131.08, 130.38, 125.74, 125.64, 123.81, 123.01, 122.58, 120.79, 119.99, 115.09; HRMS calcd for C17H12NaO2S [M+Na]+ 303.0456, found 303.0455.
(E)-3-(苯并[b]噻吩-3-基)-1-(4-羟基苯基)丙-2-烯-1-酮(1ac): HPLC/Purity: 90.90% (tR=8.231 min), yield 54.06%. m.p. 206.0~209.3 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 10.07 (s, 1H, OH), 8.60 (s, 1H, SCH=C for thiophen ring), 8.07~8.23 (m, 4H, C6H4), 8.11 (d, J=15.0 Hz, 1H, COCH=CH), 7.55 (d, J=15.0 Hz, 1H, COCH=CH), 6.94~7.60 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 187.57, 162.73, 140.37, 137.61, 134.38, 132.24, 131.62, 131.17, 130.36, 129.61, 125.67, 125.57, 123.77, 123.00, 122.60, 115.93; HRMS calcd for C17H12- NaO2S [M+Na]+ 303.0456, found 303.0454.
(E)-3-(苯并[b]噻吩-3-基)-1-(4-(三氟甲基)苯基)丙- 2-烯-1-酮(1ad): HPLC/Purity: 97.40% (tR=17.321 min), yield 31.52%. m.p. 135.5~136.3 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 8.70(s, 1H, SCH=C for thiophen ring), 8.12~8.36 (m, 4H, C6H4), 8.13 (d, J=15.0 Hz, 1H, COCH=CH), 7.55 (d, J=15.0 Hz, 1H, COCH=CH), 7.47~7.58 (m, 4H, C6H4); 13C NMR (75 MHz, DMSO-d6) δ: 189.11, 141.37, 140.38, 137.45, 136.72, 133.08, 132.65, 132.04, 131.96, 129.73, 126.27, 126.22, 125.78, 125.70, 123.82, 122.70, 122.54; HRMS calcd for C18H12F3OS [M+H]+ 333.0561, found 333.0562.
(E)-3-(苯并[b]噻吩-3-基)-1-(4-甲氧基苯基)丙-2-烯- 1-酮(1ae): HPLC/Purity: 100.00% (tR=17.614 min), yield 53.33%. m.p. 149.1~149.7 ℃; 1H NMR (300 MHz, DMSO-d6) δ: 8.63 (s, 1H, SCH=C for thiophen ring), 7.48~8.23 (m, 4H, C6H4), 8.12 (d, J=15.0 Hz, 1H, COCH=CH), 7.55 (d, J=15.0 Hz, 1H, COCH=CH), 7.11~7.51 (m, 4H, C6H4), 3.37 (s, 3H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 187.79, 163.73, 140.36, 137.61, 134.74, 132.17, 131.36, 130.94, 130.63, 125.70, 125.61, 123.80, 122.89, 122.61, 114.56, 56.07; HRMS calcd for C18H14NaO2S [M+Na]+ 317.0612, found 317.0608.