

Chinese Journal of Organic Chemistry >
Synthesis and Antibacterial Activities of Osthole Derivatives Containing Phosphonateptide Fragment
Received date: 2024-08-19
Revised date: 2024-10-21
Online published: 2024-10-29
Supported by
Guizhou Provincial Science and Technology Plan (Qiankehe Foundation ZK[2024]265); National College Students? Innovation and Entrepreneurship Training Program(202310661006)
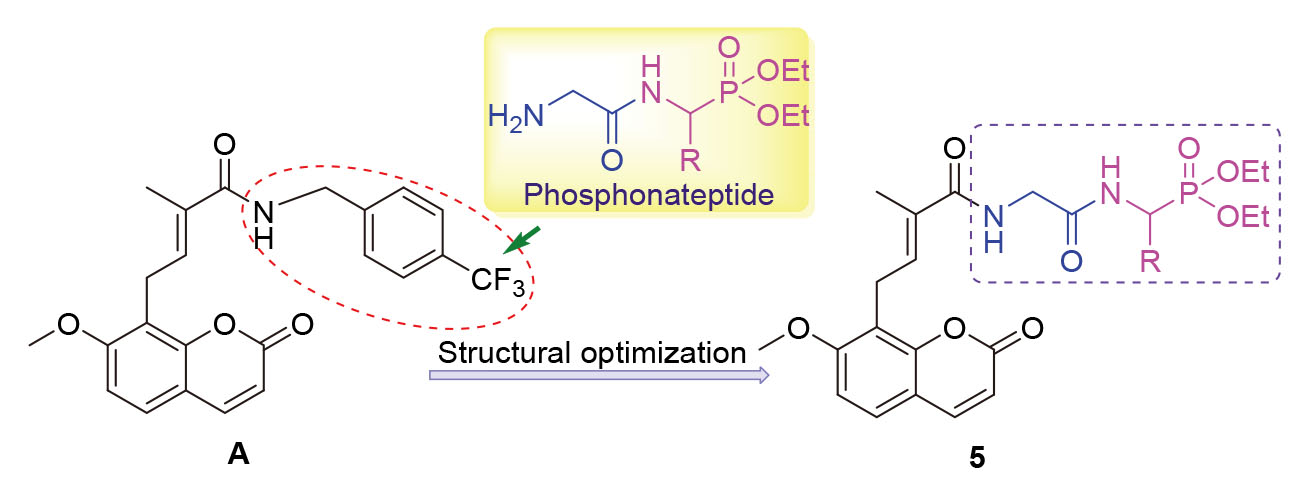
Based on previous research, the structures of osthole derivatives with potential antibacterial activities were optimized. Twenty-one novel derivatives containing phosphonateptide were designed and synthesized. The antibacterial activities against S. aureus, E. coli, methicillin-resistant S. aureus (MRSA) and fluoroquinolone-resistant E. coli (FREC) were evaluated. The results showed that the minimum inhibitory concentrations (MICs) of diethyl (E)-((2-(4-(7-methoxy-2-oxo-2H-chromen- 8-yl)-2-methylbut-2-enamido)acetamido)(3,4,5-trimethoxy phenyl)methyl)phosphonate (5f) were 0.25, 1.00, 1.00, 2.00 μg/mL respectively, and the MICs of diethyl (E)-((2-(4-(7-methoxy-2-oxo-2H-chromen-8-yl)-2-methylbut-2-enamido)acetamido)- (4-methylthiazol-2-yl)methyl)phosphonate (5u) were 0.25, 2.00, 1.00, 2.00 μg/mL, respectively. Its antibacterial activities were superior to the control drugs of oxacillin and norfloxacin, which will be further developed as novel candidates for antibacterial drugs. The binding with phosphonateptide can enhance their anti-S. aureus and anti-MRSA activities, and keep significant anti-E. coli and anti-FREC activities.
Jiaqiang Yang , Xurong Zhou , Yangmi Chen , Huixian She . Synthesis and Antibacterial Activities of Osthole Derivatives Containing Phosphonateptide Fragment[J]. Chinese Journal of Organic Chemistry, 2025 , 45(4) : 1306 -1314 . DOI: 10.6023/cjoc202407015
| [1] | Peng, X. M.; Damu, G. L. V.; Zhou, H. C. Curr. Pharm. Design 2013, 19, 3884. |
| [2] | Khursheed, A.; Jain, V. Nat. Prod. J. 2021, 11, 648. |
| [3] | Xia, D. G.; Liu, H.; Cheng, X.; Maraswami, M.; Chen, Y. T.; Lv, X. H. Curr. Top. Med. Chem. 2022, 22, 269. |
| [4] | Yi, J. L.; Shi, K. Q.; Wu, B. L.; Li, W. S.; Chen, G. Y. Chin. J. Org. Chem. 2023, 43, 295 (in Chinese). |
| [4] | (易继凌, 施康琦, 吴冰林, 黎婉珊, 陈光英, 有机化学, 2023, 43, 295.) |
| [5] | Figueroa, M.; Rivero-Cruz, I.; Rivero-Cruz, B.; Bye, R.; Navarrete, A.; Mata, R. J. Ethnopharmacol. 2007, 113, 125. |
| [6] | Rosselli, S.; Maggio, A.; Bellone, G.; Formisano, C.; Basile, A.; Cicala, C.; Alfieri, A.; Mascolo, N.; Bruno, M. Planta Med. 2007, 73, 116. |
| [7] | (a) Yang, J. Q.; Wu, X. J.; Lu, Z. C.; Chen, Y. M.; She, H. X.; Liu, H. J. Chin. J. Org. Chem. 2024, 44, 3541 (in Chinese). |
| [7] | (杨家强, 吴学姣, 卢子聪, 陈阳密, 佘慧娴, 刘海军, 有机化学, 2024, 44, 3541.) |
| [7] | (b) Yang, J. Q.; Wu, X. J.; Lu, Z. C.; Chen, Y. M.; She, H. X.; Liu, O. L. Chem. J. Chin. Univ. 2024, 45, 20240124 (in Chinese). |
| [7] | (杨家强, 吴学姣, 卢子聪, 陈阳密, 佘慧娴, 刘讴灵, 高等学校化学学报, 2024, 45, 20240124.) |
| [8] | Zhou, X. R.; Yang, H.; Zheng, J.; Wu, X. J.; Yang, J. Q. Fine Chem. 2021, 38, 790 (in Chinese). |
| [8] | (周绪容, 杨红, 郑洁, 吴学姣, 杨家强, 精细化工, 2021, 38, 790.) |
| [9] | Huang, M. G.; Ruan, X. H.; Zhang, J. P.; Li, Q.; Wang, Y. H.; Chen, L. J.; Zhang, C.; Li, P.; Xue, W. Chin. J. Org. Chem. 2017, 37, 2145 (in Chinese). |
| [9] | (黄民国, 阮祥辉, 张菊平, 李琴, 王一会, 陈丽娟, 张橙, 李普, 薛伟, 有机化学, 2017, 37, 2145.) |
| [10] | Wang, J.; Ansari, M. F.; Lin, J. M.; Zhou, C. H. Chin. J. Chem. 2021, 39, 2251. |
| [11] | Liang, C. Q.; Li, Y. M. Chin. J. Chem. Ed. 2019, 40, 1 (in Chinese). |
| [11] | (梁楚翘, 李艳梅, 化学教育, 2019, 40, 1.) |
| [12] | Rodriguez, J. B.; Gallo-Rodriguez, C. ChemMedChem 2019, 14, 190. |
| [13] | Yu, H. X.; Yang, H.; Shi, E. X.; Tang, W. J. Med. Drug Discovery 2020, 8, 100063. |
| [14] | Yang, X. C.; Zeng, C. M.; Avula, S. R.; Peng, X. M.; Geng, R. X.; Zhou, C. H. Eur. J. Med. Chem. 2023, 245, 114891. |
| [15] | Coco, L. B.; Toci, E. M.; Chen, P. Y. T.; Drennan, C. L.; Freel- Meyers, C. L. ACS Infect. Dis. 2024, 10, 1312. |
| [16] | Li, W.; Separovic, F.; O'Brien-Simpson, N. M.; Wade, J. D. Chem. Soc. Rev. 2021, 50, 4932. |
| [17] | Gan, B. H.; Gaynord, J.; Rowe, S. M.; Deingruber, D.; Spring, D. R. Chem. Soc. Rev. 2021, 50, 7820. |
| [18] | Silva, A. R. P.; Guimar?es, M. S.; Rabelo, J.; Belén, L. H.; Perecin, C. J.; Farías, J. G.; Santos, J. H. P. M.; Rangel-Yagui, C. O. J. Mater. Chem. B 2022, 10, 3587. |
| [19] | (a) Chang, J. P.; Zhao, J. R.; Chen, S. J.; Meng, K.; Shi, W. N.; Li, R. F. Chem. J. Chin. Univ. 2019, 40, 705 (in Chinese). |
| [19] | (常俊朋, 赵佳瑞, 陈思佳, 孟凯, 石微妮, 李瑞芳, 高等学校化学学报, 2019, 40, 705.) |
| [19] | (b) Haoyang, W. W.; Zhang, M.; Hou, J. L. Chin. J. Chem. 2019, 37, 25. |
| [20] | Svendsen, J. S. M.; Grant, T. M.; Rennison, D.; Brimble, M. A.; Svenson, J. Acc. Chem. Res. 2019, 52, 749. |
| [21] | Zhong, C.; Zhang, F. Y.; Zhu, N. Y.; Zhu, Y. W.; Yao, J.; Gou, S. H.; Xie, J. Q.; Ni, J. M. Eur. J. Med. Chem. 2021, 212, 113138. |
| [22] | Ronc?evic?, T.; Vukic?evic?, D.; Ilic?, N.; Krce, L.; Gajski, G.; Tonkic?, M.; Goi?-Bari?i?, I.; Zorani?, L.; Sonavane, Y.; Benincasa, M.; Jureti?, D.; Maravi?, A.; Tossi, A. J. Med. Chem. 2018, 61, 2924. |
| [23] | Lombrea, A.; Semenescu, A. D.; Magyari-Pavel, I. Z.; Turks, M.; Lugi?ina, J.; Peipi??, U.; Muntean, D.; Dehelean, C. A.; Dinu, S.; Danciu, C. Plants 2023, 12, 1253 |
| [24] | Yu, C. J. Ph.D. Dissertation, Lanzhou University, Lanzhou, 2023 (in Chinese). |
| [24] | (余长俊. 博士论文, 兰州大学, 兰州, 2023.) |
| [25] | Shen, G. X. Microbiology and Immunology, People’s Medical Publishing House, Beijing, 2007, p. 326 (in Chinese). |
| [25] | (沈关心. 微生物与免疫学, 人民卫生出版社, 北京, 2007, p. 326.) |
/
| 〈 |
|
〉 |