

Chinese Journal of Organic Chemistry >
Design, Synthesis, and Properties of 1,3-Di-tert-butylazulene Derivatives
Received date: 2024-07-09
Revised date: 2024-09-27
Online published: 2024-10-29
Supported by
National Natural Science Foundation of China(22225506); Strategic Priority Research Program of the Chinese Academy of Sciences(XDB0610000)
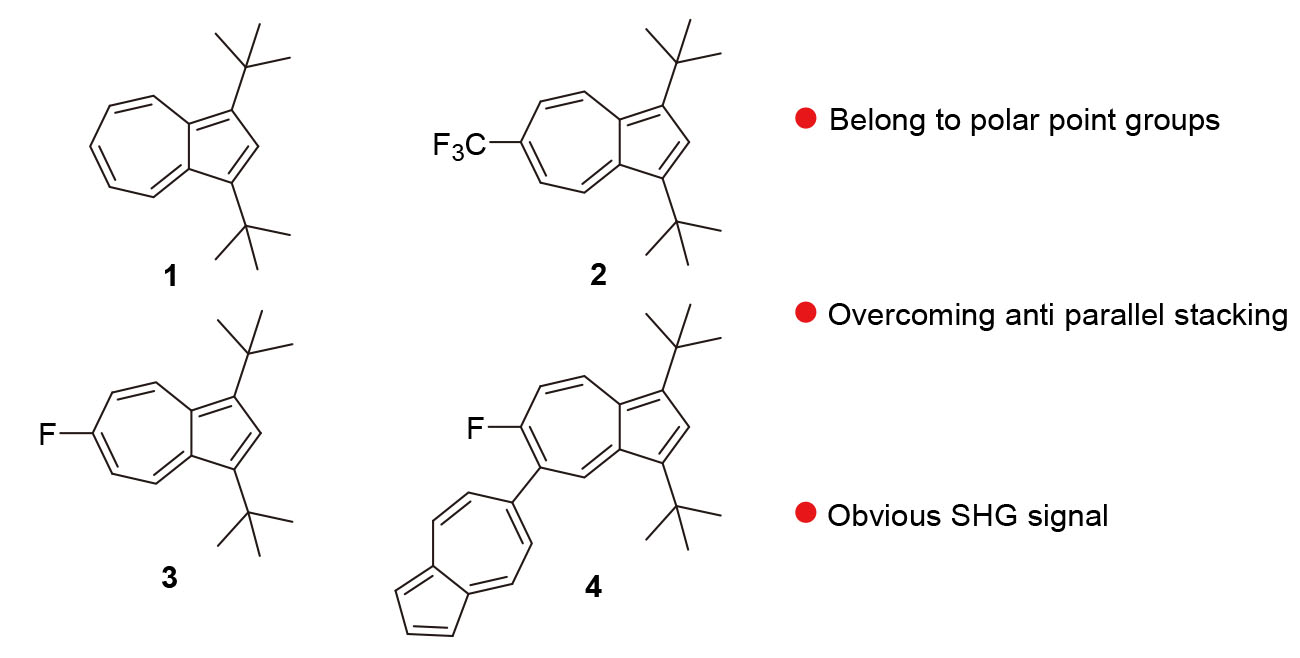
Azulene is one of the few all carbon dipole molecules that is expected to achieve ferroelectricity through the superposition of molecular dipoles. Through rational analysis, four 1,3-di-tert-butylazulene derivatives were synthesized, namely 1,3-di-tert-butylazulene (1), 1,3-di-tert-butyl-6-trifluoromethylazulene (2), 1,3-di-tert-butyl-6-fluoroazulene (3), and 1,3-di-tert-butyl-5-(6'-azulene)-6-fluoroazulene (4). The single crystal structures of these compounds are Aba2, Fdd2, Pna21, and Cc space groups all belong to 10 polar point groups and overcome antiparallel stacking in the crystal, exhibiting macroscopic polarization. Compound 4 is stacked in a molecular dipole consistent manner. This indicates that the introduction of large steric hindrance tert-butyl groups effectively reduces intermolecular dipole-dipole interactions. Compounds 1 and 4 exhibited significant second harmonic generation (SHG) signals at 300 K, which were approximately 1/4 and 2/3 of the typical inorganic ferroelectric potassium dihydrogen phosphate (KDP). These research results indicate that fluoro-group substitution and 5-site modification on azulene units are effective strategies for obtaining azulene derivatives with 10 polar point groups, providing ideas for the development of new azulene organic ferroelectrics.
Key words: azulene; molecular dipole; ferroelectricity; tert-butyl; polar point group
Yulong Xie , Junjun Xiang , Xianjiang Song , Hanyue Zhang , Xike Gao . Design, Synthesis, and Properties of 1,3-Di-tert-butylazulene Derivatives[J]. Chinese Journal of Organic Chemistry, 2025 , 45(4) : 1297 -1305 . DOI: 10.6023/cjoc202407022
| [1] | (a) Anderson, A. G. Jr.; Steckler, B. M. J. Am. Chem. Soc. 1959, 81, 4941. |
| [1] | (b) Wang, Y.; Xiang, J.; Ge, C.; Gao, X. Acta Chim. Sinica 2023, 81, 1341 (in Chinese). |
| [1] | (汪洋, 向焌钧, 葛从伍, 高希珂, 化学学报, 2023, 81, 1341.) |
| [1] | (c) Xin, H.; Hou, B.; Gao, X. Acc. Chem. Res. 2021, 54, 1737. |
| [1] | (d) Xin, H.; Gao, X. ChemPlusChem 2017, 82, 945. |
| [1] | (e) Peng, P.; Li, J.; Hou, B.; Xin, H.; Cheng, T.; Gao, X. Chin. J. Org. Chem. 2020, 40, 3916 (in Chinese). |
| [1] | (彭培珍, 李晶, 侯斌, 辛涵申, 程探宇, 高希珂, 有机化学, 2020, 40, 3916.) |
| [1] | (f) Gao, H.; Yang, X.; Xin, H.; Gao, T.; Gong, H.; Gao, X. Chin. J. Org. Chem. 2018, 38, 2680 (in Chinese). |
| [1] | (高洪磊, 杨笑迪, 辛涵申, 高铁阵, 龚和贵, 高希珂, 有机化学, 2018, 38, 2680.) |
| [1] | (g) Xin, H.; Ge, C.; Fu, L.; Yang, X.; Gao, X. Chin. J. Org. Chem. 2017, 37, 711 (in Chinese). |
| [1] | (辛涵申, 葛从伍, 傅丽娜, 杨笑迪, 高希珂, 有机化学, 2017, 37, 711.) |
| [2] | Ou, L.; Zhou, Y.; Wu, B.; Zhu, L. Chin. Chem. Lett. 2019, 30, 1903. |
| [3] | (a) Schwarz, F.; Koch, M.; Kastlunger, G.; Berke, H.; Stadler, R.; Venkatesan, K.; L?rtscher, E. Angew. Chem., Int. Ed. 2016, 55, 11781. |
| [3] | (b) Yang, G.; Sangtarash, S.; Liu, Z.; Li, X.; Sadeghi, H.; Tan, Z.; Li, R.; Zheng, J.; Dong, X.; Liu, J.; Yang, Y.; Shi, J.; Xiao, Z.; Zhang, G.; Lambert, C.; Hong, W.; Zhang, D. Chem. Sci. 2017, 8, 7505. |
| [3] | (c) Freysoldt, C.; Merz, P.; Schmidt, M.; Mohitkar, S.; Felser, C.; Neugebauer, J.; Jansen, M. Angew. Chem., Int. Ed. 2019, 58, 1. |
| [3] | (d) Huang, C.; Jevric, M.; Borges, A.; Olsen, S. T.; Hamill, J. M.; Zheng, J.-T.; Yang, Y.; Rudnev, A.; Baghernejad, M.; Broekmann, P.; Petersen, A. U.; Wandlowski, T.; Mikkelsen, K. V.; Solomon, G. C.; Br?ndsted Nielsen, M.; Hong, W. Nat. Commun. 2017, 8, 15436. |
| [3] | (e) Zhang, C.; Cheng, J.; Wu, Q.; Hou, S.; Feng, S.; Jiang, B.; Lambert, C. J.; Gao, X.; Li, Y.; Li, J. J. Am. Chem. Soc. 2023, 145, 1617. |
| [4] | (a) Zieliński, T.; K?dziorek, M.; Jurczak, J. Chem.-Eur. J. 2008, 14, 838. |
| [4] | (b) Lichosyt, D.; Wasi?ek, S.; Dydio, P.; Jurczak, J. Chem.-Eur. J. 2018, 24, 11683. |
| [5] | (a) Yamaguchi, Y.; Maruya, Y.; Katagiri, H.; Nakayama, K.-I.; Ohba, Y. Org. Lett. 2012, 14, 2316. |
| [5] | (b) Yamaguchi, Y.; Ogawa, K.; Nakayama, K.-I.; Ohba, Y.; Katagiri, H. J. Am. Chem. Soc. 2013, 135, 19095. |
| [5] | (c) Yamaguchi, Y.; Takubo, M.; Ogawa, K.; Nakayama, K.-I.; Koganezawa, T.; Katagiri, H. J. Am. Chem. Soc. 2016, 138, 11335. |
| [5] | (d) Yao, J.; Cai, Z.; Liu, Z.; Yu, C.; Luo, H.; Yang, Y.; Yang, S.; Zhang, G.; Zhang, D. Macromolecules 2015, 48, 2039. |
| [5] | (e) Shoji, T.; Ito, S. Sci. China: Chem. 2018, 61, 973. |
| [5] | (f) Schulz, F.; Takamaru, S.; Bens, T.; Hanna, J.-I.; Sarkar, B.; Laschat, S.; Iino, H. Phys. Chem. Chem. Phys. 2022, 24, 23481. |
| [6] | (a) Yang, C.-C.; Ma, J.-Y.; Su, X.; Zheng, X.-L.; Chen, J.; He, Y.-Y.; Tian, W.-Q.; Li, W.-Q.; Yang, L. FlatChem 2022, 33, 100362. |
| [6] | (b) Yang, C.-C.; Li, L.; Tian, W. Q.; Li, W.-Q.; Yang, L. Phys. Chem. Chem. Phys. 2022, 24, 13275. |
| [7] | (a) Wang, F.; Lin, T. T.; He, C.; Chi, H.; Tang, T.; Lai, Y.-H. J. Mater. Chem. 2012, 22, 10448. |
| [7] | (b) Ince, M.; Bartelmess, J.; Kiessling, D.; Dirian, K.; Martínez- Díaz, M. V.; Torres, T.; Guldi, D. M. Chem. Sci. 2012, 3, 1472. |
| [7] | (c) Yao, Y.; Zhang, Y.; Zhang, J.; Yang, X.; Ding, D.; Shi, Y.; Xu, H.; Gao, X. ACS Appl. Mater. Interfaces 2022, 14, 19192. |
| [7] | (d) Gao, H.; Yao, Y.; Li, C.; Zhang, J.; Yu, H.; Yang, X.; Shen, J.; Liu, Q.; Xu, R.; Gao, X.; Ding, D. Angew. Chem., Int. Ed. 2024, 63, e202400372. |
| [8] | (a) Seidel, J.; Fu, D.; Yang, S.-Y.; Alarcón-Lladó, E.; Wu, J.; Ramesh, R.; Ager, J. W. Phys. Rev. Lett. 2011, 107, 126805. |
| [8] | (b) Choi, T.; Lee, S.; Choi, Y. J.; Kiryukhin, V.; Cheong, S.-W. Science 2009, 324, 63. |
| [8] | (c) Scott, J. F. Science 2007, 315, 954. |
| [8] | (d) Scott, J. F.; Paz de Araujo, C. A. Science 1989, 246, 1400. |
| [8] | (e) Whatmore, R. W. Rep. Prog. Phys. 1986, 49, 1335. |
| [9] | Shi, P.-P.; Tang, Y.-Y.; Li, P.-F.; Liao, W.-Q.; Wang, Z.-X.; Ye, Q.; Xiong, R.-G. Chem. Soc. Rev. 2016, 45, 3811. |
| [10] | (a) Thomas, S.; Ramasesha, S.; Hallberg, K.; Garcia, D. Phys. Rev. B 2012, 86, 180403. |
| [10] | (b) Bühl, M.; Ko?miński, W.; Linden, A.; Nanz, D.; Sperandio, D.; Hansen, H.-J. Helv. Chim. Acta 1996, 79, 837. |
| [10] | (c) Dyker, G.; Borowski, S.; Heiermann, J.; K?rning, J.; Opwis, K.; Henkel, G.; K?ckerling, M. J. Organomet. Chem. 2000, 606, 108. |
| [10] | (d) Adachi, T.; Saitoh, H.; Yamamura, Y.; Hishida, M.; Ueda, M.; Ito, S.; Saito, K. Bull. Chem. Soc. Jpn. 2013, 86, 1022. |
| [10] | (e) Horiuchi, S.; Tokura, Y. Nat. Mater. 2008, 7, 357. |
| [10] | (f) Horiuchi, S.; Tokunaga, Y.; Giovannetti, G.; Picozzi, S.; Itoh, H.; Shimano, R.; Kumai, R.; Tokura, Y. Nature 2010, 463, 789. |
| [10] | (g) Fu, D.-W.; Cai, H.-L.; Liu, Y.; Ye, Q.; Zhang, W.; Zhang, Y.; Chen, X.-Y.; Giovannetti, G.; Capone, M.; Li, J.; Xiong, R.-G. Science 2013, 339, 425. |
| [10] | (h) Ye, H.-Y.; Tang, Y.-Y.; Li, P.-F.; Liao, W.-Q.; Gao, J.-X.; Hua, X.-N.; Cai, H.; Shi, P.-P.; You, Y.-M.; Xiong, R.-G. Science 2018, 361, 151. |
| [11] | Danzmann, S.; Liebing, P.; Engelhardt, F.; Hilfert, L.; Edelmann, F. T. Inorg. Chem. Commun. 2019, 99, 176. |
| [12] | F?rster, S.; Seichter, W.; Kuhnert, R.; Weber, E. J. Mol. Struct. 2014, 1075, 63. |
| [13] | Voss, J.; Pesel, T.; Buddensiek, D.; Lehtivarjo, J. Z. Naturforsch., : J. Chem. Sci. 2015, 70, 441. |
| [14] | Tsuchiya, T.; Katsuoka, Y.; Yoza, K.; Sato, H.; Mazaki, Y. Chem- PlusChem 2019, 84, 1659. |
| [15] | Hou, B.; Li, J.; Xin, H.; Yang, X.; Gao, H.; Peng, P.; Gao, X. Acta Chim. Sinica 2020, 78, 788 (in Chinese). |
| [15] | (侯斌, 李晶, 辛涵申, 杨笑迪, 高洪磊, 彭培珍, 高希珂, 化学学报, 2020, 78, 788.) |
| [16] | (a) Chopra, D.; Row, T. N. G. CrystEngComm 2011, 13, 2175. |
| [16] | (b) Ai, Y.; Chen, X.-G.; Shi, P.-P.; Tang, Y.-Y.; Li, P.-F.; Liao, W.-Q.; Xiong, R.-G. J. Am. Chem. Soc. 2019, 141, 4474. |
| [16] | (c) Tang, Y.-Y.; Ai, Y.; Liao, W.-Q.; Li, P.-F.; Wang, Z.-X.; Xiong, R.-G. Adv. Mater. 2019, 31, 1902163. |
| [16] | (d) Liu, Y.-L.; Lu, S.-Q.; Tang, Y.-Y.; Chen, X.-G.; Gao, J.-X.; Li, H.-J.; Xiong, R.-G. Chem. Commun. 2019, 55, 10007. |
| [17] | (a) Nozoe, T.; Seto, S.; Matsumura, S.; Asano, T. Proc. Jpn. Acad. 1956, 32, 339. |
| [17] | (b) Nozoe, T.; Seto, S.; Matsumura, S.; Murase, Y. Bull. Chem. Soc. Jpn. 2006, 35, 1179. |
| [18] | (a) McDonald, R. N.; Richmond, J. M.; Curtis, J. R.; Petty, H. E.; Hoskins, T. L. J. Org. Chem. 1976, 41, 1811. |
| [18] | (b) Meher, S.; Sharma, N. K. New J. Chem. 2023, 47, 5593. |
| [19] | (a) Cowper, P.; Pockett, A.; Kociok-K?hn, G.; Cameron, P. J.; Lewis, S. E. Tetrahedron 2018, 74, 2775. |
| [19] | (b) Chen, M.; Buchwald, S. L. Angew. Chem., Int. Ed. 2013, 52, 11628. |
| [19] | (c) Frigola, J.; Colombo, A.; Más, J.; Parés, J. J. Heterocycl. Chem. 1987, 24, 399. |
| [19] | (d) Nishimura, H.; Eliseeva, M. N.; Wakamiya, A.; Scott, L. T. Synlett 2015, 26, 1578. |
| [20] | Gerson, F.; Scholz, M.; Hansen, H.-J.; Uebelhart, P. J. Chem. Soc., Perkin Trans. 2 1995, 215. |
| [21] | Pan, Q.; Liu, Z.-B.; Zhang, H.-Y.; Zhang, W.-Y.; Tang, Y.-Y.; You, Y.-M.; Li, P.-F.; Liao, W.-Q.; Shi, P.-P.; Ma, R.-W.; Wei, R.-Y.; Xiong, R.-G. Adv. Mater. 2017, 29, 1700831. |
| [22] | Zhang, H.-Y.; Chen, X.-G.; Tang, Y.-Y.; Liao, W.-Q.; Di, F.-F.; Mu, X.; Peng, H.; Xiong, R.-G. Chem. Soc. Rev. 2021, 50, 8248. |
/
| 〈 |
|
〉 |