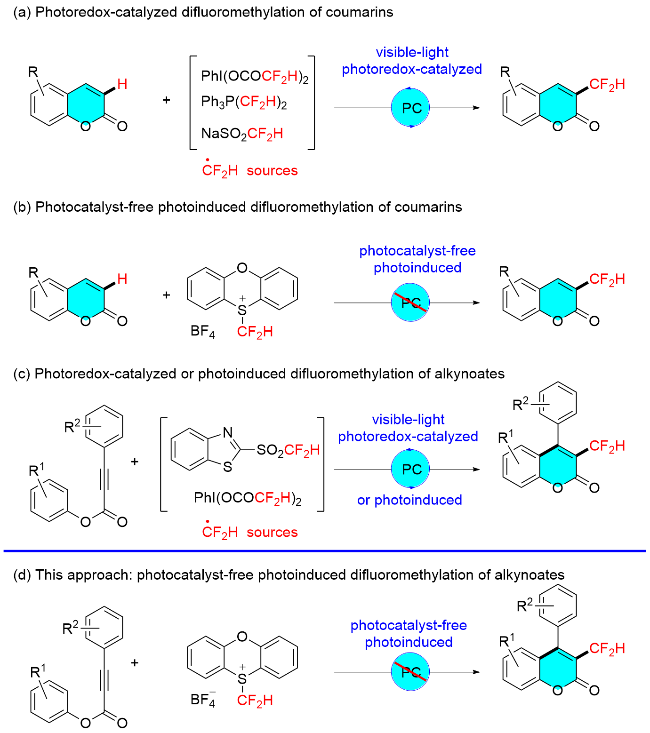
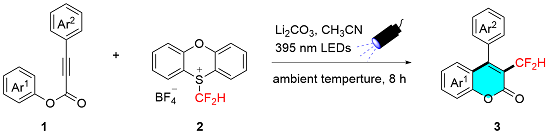
To a 10 mL Schlenk tube equipped with a magnetic stir bar were added alkynoate
1 (0.2 mmol, 1.0 equiv.), PT-
$\mathrm{CF}_{2} \mathrm{H}^{+} \mathrm{BF}_{4}^{-}$ 2 (0.6 mmol, 202.8 mg, 3.0 equiv.) and Li
2CO
3 (0.5 mmol, 36.95 mg, 2.5 equiv.). The flask was flushed with argon, followed by the addition of acetonitrile (4.0 mL). The tube was placed at a distance of 2~3 cm away from UV LED lamps (
λ=395 nm, 20 W×2), then the reaction mixture was stirred under irradiation of UV LEDs for 8 h at ambient temperature. The mixture was evaporated
in vacuo after reaction accomplished, and the residue was purified by flash column chromatography on silica gel (petrol ether/EtOAc,
V∶
V=12∶1) to give desired product
3. All known products
3a~
3o and
3r~
3t were prepared according to the general procedure, and analytical data are in agreement with those reported in the previous literatures.
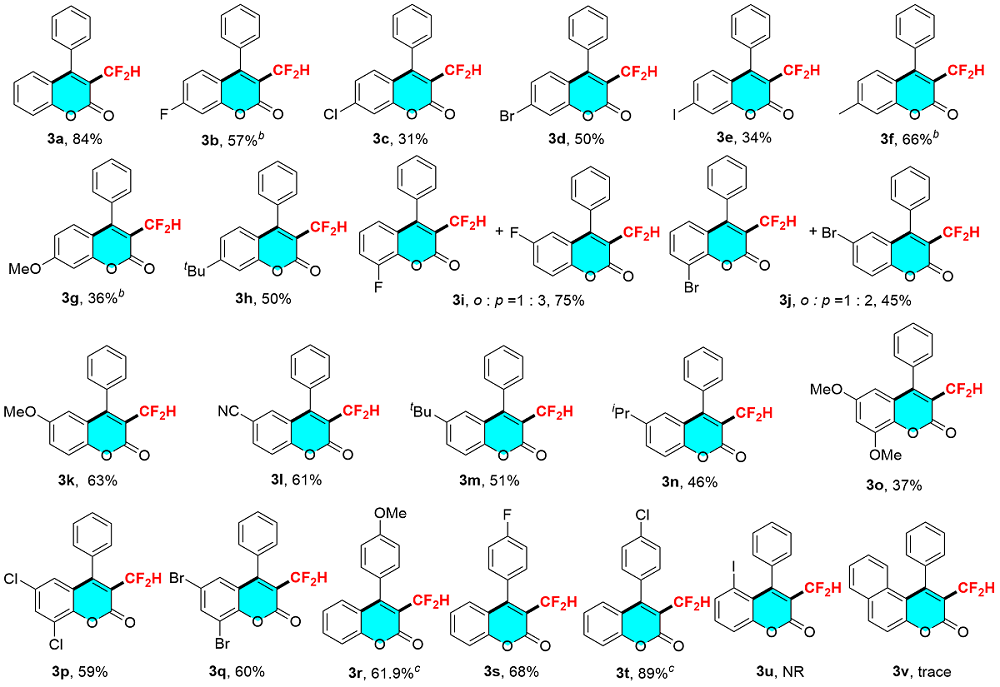
[12-13]3-(Difluoromethyl)-4-phenyl-2H-chromen-2-one (3a): Yellow solid (45.7 mg, 84% yield). 1H NMR (400 MHz, CDCl3) δ: 7.63 (t, J=8.6 Hz, 1H), 7.60~7.53 (m, 3H), 7.43 (d, J=8.4 Hz, 1H), 7.40~7.31 (m, 2H), 7.23 (t, J=8.2 Hz, 1H), 7.13 (d, J=8.1 Hz, 1H), 6.55 (t, J=53.3 Hz, 1H); 19F NMR (377 MHz, CDCl3) δ: -114.79 (d, J=52.7 Hz, 2F).
3-(Difluoromethyl)-6-fluoro-4-phenyl-2H-chromen-2-one (3b): Yellow solid (26.1 mg, 45% yield). 1H NMR (400 MHz, CDCl3) δ: 7.88~7.50 (m, 3H), 7.44~7.29 (m, 2H), 7.18~ 7.09 (m, 2H), 6.96 (td, J=8.5, 2.6 Hz, 1H), 6.51 (t, J=53.2 Hz, 1H); 19F NMR (377 MHz, CDCl3) δ: -102.34 (d, J=6.9 Hz), -114.74 (d, J=52.7 Hz, 2F).
3-(Difluoromethyl)-7-chloro-4-phenyl-2H-chromen-2-one (3c): Colorless oil (9.4 mg, 31% yield). 1H NMR (400 MHz, CDCl3) δ: 7.64~7.55 (m, 3H), 7.45 (d, J=2.0 Hz, 1H), 7.36~7.31 (m, 2H), 7.20 (dd, J=8.6, 2.0 Hz, 1H), 7.06 (d, J=8.6 Hz, 1H), 6.53 (t, J=53.2 Hz, 1H); 19F NMR (377 MHz, CDCl3) δ: -114.80 (d, J=53.4 Hz, 2F).
3-(Difluoromethyl)-7-bromo-4-phenyl-2H-chromen-2-one (3d): Orange solid (17.9 mg, 51% yield). 1H NMR (500 MHz, CDCl3) δ: 7.61 (d, J=1.8 Hz, 1H), 7.59 (dd, J=5.0, 1.7 Hz, 2H), 7.35~7.32 (m, 2H), 7.18~7.08 (m, 1H), 7.05~7.00 (m, 1H), 6.98 (d, J=8.5 Hz, 1H), 6.52 (t, J=53.2 Hz, 1H); 19F NMR (377 MHz, CDCl3) δ: -114.80 (d, J=53.4 Hz, 2F).
3-(Difluoromethyl)-7-iodo-4-phenyl-2H-chromen-2-one (3e): Reddish brown solid (34.3 mg, 34% yield). 1H NMR (500 MHz, CDCl3) δ: 7.81 (d, J=1.7 Hz, 1H), 7.63~7.53 (m, 4H), 7.35~7.30 (m, 2H), 6.81 (d, J=8.4 Hz, 1H), 6.52 (t, J=53.2 Hz, 1H); 19F NMR (471 MHz, CDCl3) δ: -114.84 (d, J=52.9 Hz, 2F).
3-(Difluoromethyl)-7-methyl-4-phenyl-2H-chromen-2-one(3f): Yellow solid (37.8 mg, 66% yield). 1H NMR (500 MHz, CDCl3) δ: 7.62~7.53 (m, 3H), 7.34 (dd, J=6.6, 3.1 Hz, 2H), 7.23 (s, 1H), 7.05~6.98 (m, 2H), 6.53 (t, J=53.3 Hz, 1H), 2.48 (s, 3H); 19F NMR (471 MHz, CDCl3) δ: -114.54 (d, J=53.8 Hz, 2F).
3-(Difluoromethyl)-7-methoxy-4-phenyl-2H-chromen-2- one (3g): Yellow solid (21.4 mg, 35.4% yield). 1H NMR (400 MHz, CDCl3) δ: 7.60~7.53 (m, 3H), 7.33 (dd, J=6.6, 2.9 Hz, 2H), 7.04~7.02 (m, 1H), 6.90 (d, J=2.5 Hz, 1H), 6.78 (dd, J=9.0, 2.5 Hz, 1H), 6.51 (t, J=53.4 Hz, 1H), 3.91 (s, 3H); 19F NMR (377 MHz, CDCl3) δ: -114.12 (d, J=53.4 Hz, 2F).
3-(Difluoromethyl)-7-(tert-butyl)-4-phenyl-2H-chromen-2-one (3h): Yellow solid (33 mg, 50% yield). 1H NMR (500 MHz, CDCl3) δ: 7.61~7.53 (m, 3H), 7.43 (d, J=1.8 Hz, 1H), 7.38 ~7.32 (m, 2H), 7.26 (dd, J=8.5, 1.9 Hz, 1H), 7.05 (d, J=8.5 Hz, 1H), 6.55 (t, J=53.3 Hz, 1H), 1.36 (s, 9H); 19F NMR (471 MHz, CDCl3) δ: -114.46 (d, J=52.9 Hz, 2F).
3-(Difluoromethyl)-6-fluoro-4-phenyl-2
H-chromen-2-one (
3i):
[15] Yellow oil (43.5 mg, 75% yield).
1H NMR (500 MHz, CDCl
3)
δ: 7.61~7.57 (m, 4H), 7.43 (d,
J=3.8 Hz, 0.3H), 7.43~7.40 (m, 1H), 7.39~7.31 (m, 4H), 6.90 (d,
J=8.2 Hz, 0.3H), 6.80 (dd,
J=8.8, 3.0 Hz, 1H), 6.55 (t,
J=53.2 Hz, 1H), 6.54 (t,
J=53.1 Hz, 0.3H);
19F NMR (471 MHz, CDCl
3)
δ: -114.96 (d,
J=56.5Hz, 2F), -115.79~ -115.84 (m, 1F).
3-(Difluoromethyl)-6-bromo-4-phenyl-2
H-chromen-2-one (
3j):
[15] Yellow oil (31.4 mg, 45% yield).
1H NMR (500 MHz, CDCl
3)
δ: 7.85 (dd,
J=7.5, 1.8 Hz, 0.5H), 7.71 (dd,
J=8.8, 2.3 Hz, 1H), 7.63~7.55 (m, 4.5H), 7.37~7.30 (m, 4H), 7.22 (d,
J=2.3 Hz, 1H), 7.12~7.06 (m, 1H), 6.54 (t,
J=53.2 Hz, 0.5H), 6.51 (t,
J=53.2 Hz, 1H);
19F NMR (471 MHz, CDCl
3)
δ: -114.89 (d,
J=53.8 Hz), -115.02 (d,
J=52.9 Hz, 2F)
3-(Difluoromethyl)-6-methoxy-4-phenyl-2H-chromen-2-one (3k): Yellow solid (38.1 mg, 63% yield). 1H NMR (400 MHz, CDCl3) δ: 7.60~7.55 (m, 3H), 7.37 (d, J=9.1 Hz, 2H), 7.20 (dd, J=9.1, 2.9 Hz, 1H), 6.68~6.41 (m, 2H), 3.69 (s, 3H); 19F NMR (377 MHz, CDCl3) δ: -114.72 (d, J=53.4 Hz, 2F).
3-(Difluoromethyl)-2-oxo-4-phenyl-2H-chromene-6-carbonitrile (3l): Reddish brown solid (36.2 mg, 61% yield). 1H NMR (500 MHz, CDCl3) δ: 7.91 (dd, J=7.6, 1.8 Hz, 1H), 7.61 (dd, J=5.0, 2.1 Hz, 3H), 7.39~7.31 (m, 4H), 6.53 (t, J=52.9 Hz, 1H); 19F NMR (471 MHz, CDCl3) δ: -115.08 (d, J=52.9 Hz, 2F).
3-(Difluoromethyl)-6-(tert-butyl)-4-phenyl-2H-chromen-2-one (3m): Yellow solid (33.7 mg, 51% yield). 1H NMR (500 MHz, CDCl3) δ: 7.67 (dd, J=8.7, 2.4 Hz, 1H), 7.62~7.55 (m, 3H), 7.40~7.33 (m, 3H), 7.08 (d, J=2.4 Hz, 1H), 6.55 (t, J=53.3 Hz, 1H), 1.21 (s, 9H); 19F NMR (471 MHz, CDCl3) δ: -114.64 (d, J=53.8 Hz, 2F).
3-(Difluoromethyl)-6-isopropyl-4-phenyl-2H-chromen-2-one (3n): Yellow solid (28.9 mg, 46% yield). 1H NMR (600 MHz, CDCl3) δ: 7.65~7.55 (m, 3H), 7.39~7.36 (m, 2H), 7.16~7.12 (m, 1H), 7.06~7.03 (m, 1H), 6.55 (t, J=53.3 Hz, 1H), 2.87 (p, J=6.9 Hz, 1H), 1.18 (d, J=6.9 Hz, 6H); 19F NMR (471 MHz, CDCl3) δ: -115.24 (d, J=53.8 Hz, 2F).
3-(Difluoromethyl)-6,8-dimethoxy-4-phenyl-2H-chro-men-2-one (3o): Yellow solid (24.7 mg, 37.2% yield). 1H NMR (500 MHz, CDCl3) δ: 7.60~7.46 (m, 3H), 7.38~7.29 (m, 2H), 6.76 (d, J=2.6 Hz, 1H), 6.55 (t, J=53.3 Hz, 1H), 6.04 (d, J=2.7 Hz, 1H), 3.81 (d, J=170.9 Hz, 6H); 19F NMR (471 MHz, CDCl3) δ: -114.63 (d, J=52.9 Hz, 2F).
3-(Difluoromethyl)-6,8-dichloro-4-phenyl-2H-chromen-2-one (3p): Yellow solid (39.9 mg, 59% yield), m.p. 157~160 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.68 (d, J=2.4 Hz, 1H), 7.61 (dd, J=5.0, 2.1 Hz, 3H), 7.34~7.31 (m, 2H), 6.98 (d, J=2.4 Hz, 1H), 6.52 (t, J=53.0 Hz, 1H); 13C NMR (151 MHz, Chloroform-d) δ: 156.19, 155.06, 148.08, 133.34, 130.70, 130.32, 129.89, 129.12, 128.27, 126.44, 123.20, 121.66, 119.52 (t, J=22.5 Hz), 111.20 (t, J=240.0 Hz); 19F NMR (471 MHz, CDCl3) δ: -115.14 (d, J=52.9 Hz, 2F); HRMS (ESI) calcd for C16H8Cl2F2O2 [M+H]+ 341.1369, found 341.1348.
3-(Difluoromethyl)-6,8-dibromo-4-phenyl-2H-chromen-2-one (3q): Yellow solid (51.3 mg, 60% yield), m.p. 173~176 ℃; 1H NMR (600 MHz, Chloroform-d) δ: 7.96 (d, J=2.2 Hz, 1H), 7.63~7.57 (m, 3H), 7.32~7.28 (m, 2H), 7.14 (d, J=2.2 Hz, 1H), 6.49 (t, J=53.0 Hz, 1H); 13C NMR (151 MHz, Chloroform-d) δ: 156.22, 154.94, 149.48, 138.91, 130.57, 130.29, 130.09, 129.09, 128.23, 121.98, 119.46 (d, J=22.7 Hz), 117.29, 111.81, 111.12 (t, J=240.0 Hz); 19F NMR (471 MHz, CDCl3) δ: -115.11 (d, J=52.9 Hz, 2F); HRMS (ESI) calcd for C16H8Br2F2O2 [M+H]+ 430.0457, found 430.0428.
3-(Difluoromethyl)-4-(4-methoxyphenyl)-2H-chromen-2-one (3r): Yellow solid (37.4 mg, 61.9% yield). 1H NMR (500 MHz, CDCl3) δ: 7.65~7.59 (m, 1H), 7.42 (d, J=8.2 Hz, 1H), 7.30~7.27 (m, 2H), 7.23 (qd, J=8.1, 1.8 Hz, 2H), 7.09 (d, J=8.9 Hz, 2H), 6.56 (t, J=53.3 Hz, 1H), 3.93 (s, 3H); 19F NMR (471 MHz, CDCl3) δ: -114.76 (d, J=52.9 Hz, 2F).
3-(Difluoromethyl)-4-(4-fluorophenyl)-2H-chromen-2-one (3s): Yellow solid (39.4 mg, 68% yield). 1H NMR (500 MHz, CDCl3) δ: 7.64 (t, J=8.7 Hz, 1H), 7.43 (d, J=7.2 Hz, 1H), 7.35 (dd, J=8.9, 5.2 Hz, 2H), 7.30~7.23 (m, 3H), 7.12 (d, J=1.7 Hz, 1H), 6.62 (t, J=53.3 Hz, 1H); 19F NMR (471 MHz, CDCl3) δ: -110.60, -113.96 (d, J=52.9 Hz, 2F).
3-(Difluoromethyl)-4-(4-chlorophenyl)-2H-chromen-2-One (3t): brown solid (54.7 mg, 89.2 % yield). 1H NMR (500 MHz, CDCl3) δ: 7.64 (t, J=8.6 Hz, 1H), 7.56 (d, J=8.4 Hz, 2H), 7.43 (d, J=8.4 Hz, 1H), 7.30 (d, J=8.4 Hz, 2H), 7.25 (t, J=8.2 Hz, 1H), 7.12 (dd, J=6.7, 1.8 Hz, 1H), 6.64 (t, J=53.3 Hz, 1H); 19F NMR (471 MHz, CDCl3) δ: -113.72 (d, J=52.9 Hz, 2F).