

Chinese Journal of Organic Chemistry >
Base-Catalyzed Chemoselective Substitution of β-Ketonitrile with Azodicarboxylates
Received date: 2024-08-06
Revised date: 2024-10-27
Online published: 2024-11-28
Supported by
National Natural Science Foundation of China(21563025); National Natural Science Foundation of China(22162022)
Base-catalyzed nucleophilic substitution reactions of β-ketonitrile with azodicarboxylates have been developed. A series of disubstituted C—N coupling products were obtained in good to excellent yields under Et3N catalysis. Monosubstitution C—N bond formation reaction catalyzed by K2CO3 also gave novel enol-based target products. This method is simple and mild, with good chemoselectivity, excellent substrate compatibility and tolerance for various functional groups, and achieves gram-scale synthesis. The reaction is a nucleophilic substitution process without the involvement of free radicals.
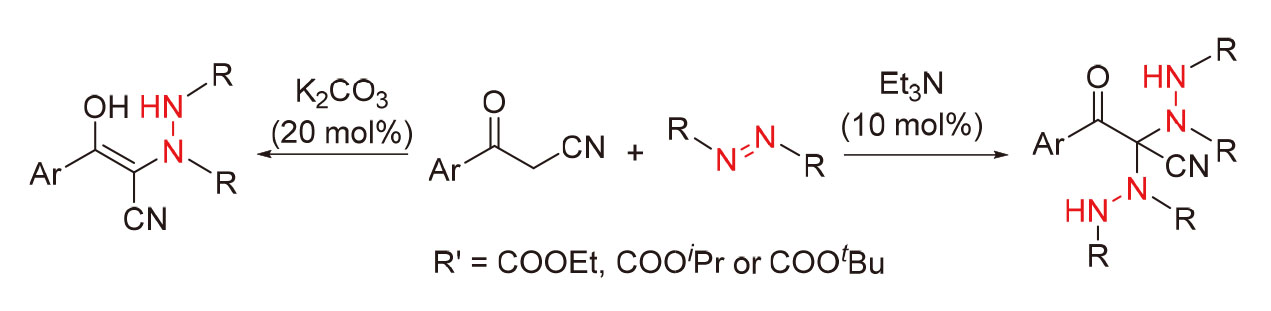
Key words: base-catalysis; β-ketonitrile; selective substitution; azodicarboxylate
Yishi Guo , Tian Sang , Yan Liu , Ping Liu . Base-Catalyzed Chemoselective Substitution of β-Ketonitrile with Azodicarboxylates[J]. Chinese Journal of Organic Chemistry, 2025 , 45(4) : 1249 -1260 . DOI: 10.6023/cjoc202408005
| [1] | (a) Mitsunobu, O. Synthesis 1981, 1981, 1. |
| [1] | (b) Mitsunobu, O.; Takemasa, A.; Endo, R. Chem. Lett. 1984, 13, 855. |
| [2] | (a) Swamy, K. C. K.; Kumar, N. N. B.; Balaraman, E.; Kumar, K. V. P. P. Chem. Rev. 2009, 109, 2551. |
| [2] | (b) Fletcher, S. Org. Chem. Front. 2015, 2, 739. |
| [3] | Beddoe, R. H.; Sneddon, H. F.; Denton, R. M. Org. Biomol. Chem. 2018, 16, 7774. |
| [4] | Hughes, D. L. Org. React. 1992, 335. |
| [5] | Hirose, D.; Gazvoda, M.; Ko?mrlj, J.; Taniguchi, T. Chem. Sci. 2016, 7, 5148. |
| [6] | Usman, M.; Zhang, X. W.; Wu, D.; Guan, Z. H.; Liu, W. B. Org. Chem. Front. 2019, 6, 1905. |
| [7] | (a) Leblanc, Y.; Fitzsimmons, B. J.; Rokach, J.; Springer, J. P.; J. Am. Chem. Soc. 1989, 111, 2995. |
| [7] | (b) Lee, C. K.; Ahn, Y. M. J. Org. Chem. 1990, 55, 3957. |
| [7] | (c) Barluenga, J.; González, F. J.; Fustero, S.; Gotor, V. J. Chem. Soc. Chem. Commun. 1986, 1179. |
| [8] | (a) Varlet, T.; Levitre, G.; Retailleau, P.; Masson, G. Bioorg. Med. Chem. 2019, 27, 2438. |
| [8] | (b) Mane, V.; Pandey, J.; Ayyagari, N.; Dey, C.; Kale, R.; Namboothiri, I. N. N. Org. Biomol. Chem. 2016, 14, 2427. |
| [9] | (a) List, B.; Am, J. Chem. Soc. 2002, 124, 5656. |
| [9] | (b) B?gevig, A.; Juhl, K.; Kumaragurubaran, N.; Zhuang, W.; J?rgensen, K. A. Angew. Chem., Int. Ed. 2002, 41, 1790. |
| [10] | (a) Ni, J.; Li, J.; Fan, Z.; Zhang, A. Org. Lett. 2016, 18, 5960. |
| [10] | (b) Ling, F.; Ai, C.; Lv, Y.; Zhong, W. Adv. Synth. Catal. 2017, 359, 3707. |
| [11] | Xu, N.; Li, D.; Zhang, Y.; Wang, L. Org. Biomol. Chem. 2015, 13, 9083. |
| [12] | (a) Nair, V.; Biju, A. T.; Mathew, S. C.; Babu, B. P. Chem.-Asian J. 2008, 3, 810. |
| [12] | (b) Samzadeh-Kermani, A. Tetrahedron Lett. 2016, 57, 463. |
| [13] | (a) Trost, B. M.; Tracy, J. S. Org. Lett. 2017, 19, 2630. |
| [13] | (b) Vallribera, A.; Maria Sebastian, R.; Shafir, A. Curr. Org. Chem. 2011, 15, 1539. |
| [14] | Lu, Q.-T.; Du, Y.-B.; Xu, M.-M.; Xie, P-P.; Cai, Q. J. Am. Chem. Soc. 2024, 146, 21535. |
| [15] | Jiang, Y. S.; Chen, D. N.; Jiang, H.; Xia, P. J. Green Synth. Catal. 2025, doi: 10.1016/j.gresc.2024.07.003. |
| [16] | (a) Hari, D. P.; Schroll, P.; K?nig, B. J. Am. Chem. Soc. 2012, 134, 2958. |
| [16] | (b) Batra, A.; Singh, K. N. Eur. J. Org. Chem. 2020, 2020, 6676. |
| [16] | (c) Singh, P.; Batra, A.; Singh, K. N.; Mritunjay, M. Synthesis 2021, 53, 1556. |
| [16] | (d) Yeung, C. S.; Dong, V. M. Chem. Rev. 2011, 111, 1215. |
| [16] | (e) Batra, A.; Singh, P.; Singh, K. N. Asian J. Org. Chem. 2021, 10, 1024. |
| [17] | Zhou, Y. Q.; Wang, N. X.; Xing, Y. L.; Wang, Y. J.; Hong, X. W.; Zhang, J. X.; Chen, D. D.; Geng, J. B.; Dang, Y. F.; Wang, Z. X. Sci. Rep. 2013, 3, 1058. |
| [18] | Zeng, G.; Liu, J.; Shao, Y.; Zhang, F.; Chen, Z.; Lv, N.; Chen, J.; Li, R. J. Org. Chem. 2021, 86, 861. |
/
| 〈 |
|
〉 |