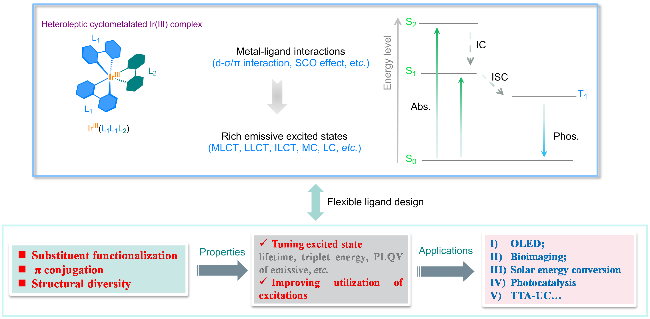
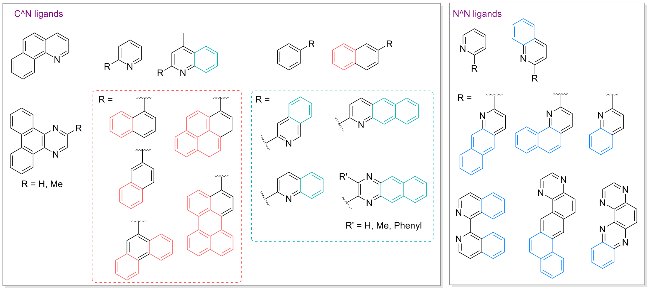
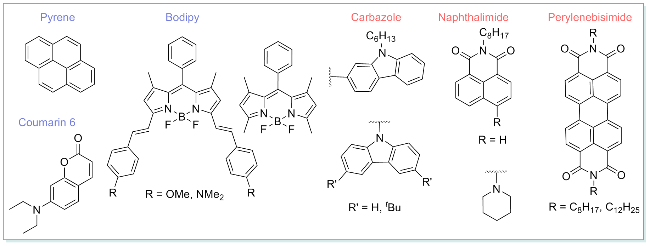
图1 杂环金属铱(III)配合物IrIII(L1L1L2)的分子设计策略及潜在应用Figure 1 Molecular design strategies and potential applications of heteroleptic iridium complex IrIII(L1L1L2) Abs., S1, S2, Phos., IC, and ISC represent absorption, first singlet excited state, second singlet excited state, phosphorescence, internal conversion, and intersystem crossing, respectively. |
1 [Ir(C^N)2(N^N)]+类环金属铱(III)配合物的光物理性质和调控
1.1 [Ir(C^N)2(N^N)]+类环金属铱(III)配合物的一般光物理性质
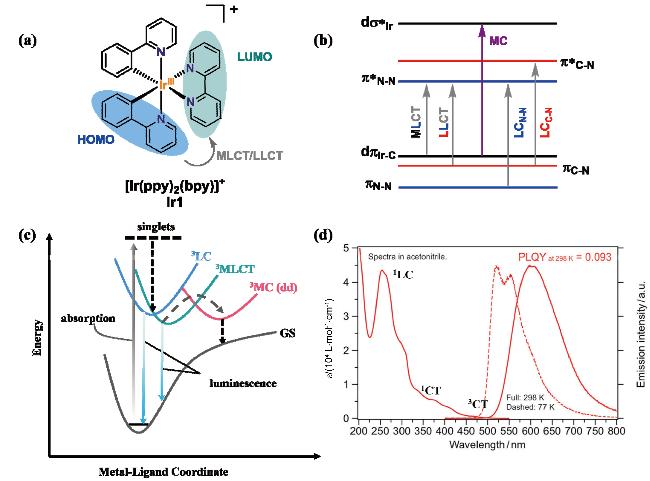
图2 (a)杂环金属铱配合物[Ir(ppy)2(bpy)]+ (Ir1)的结构式; (b) Ir(III)配合物电子构型和最低能跃迁能级; (c) LC、MLCT和MC激发态的电子跃迁(MC电子能级无发光, GS表示基态); (d)金属铱配合物Ir1在乙腈溶液中的吸收和发射光谱[11a]Figure 2 (a) Structure of heteroleptic iridium complex [Ir(ppy)2(bpy)]+ (Ir1); (b) Electronic configuration and the lowest-energy transitions of Ir(III) complexes; (c) Electronic transitions involving the excited states of LC, MLCT, and MC (MC levels are not emissive, GS represents ground state); (d) Absorption and emission spectra of Ir1 complex in acetonitrile[11a] |
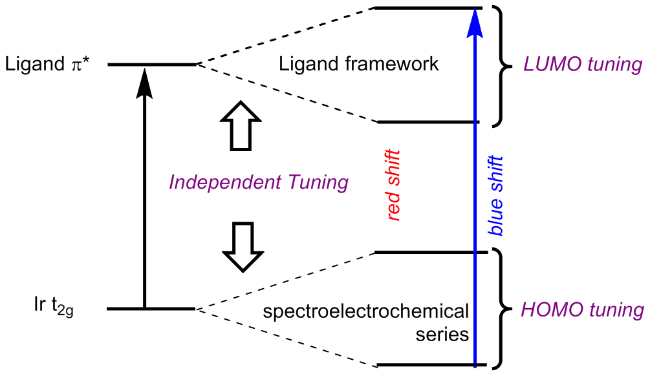
1.2 [Ir(C^N)2(N^N)]+类环金属铱(III)配合物的光物理性质的调控
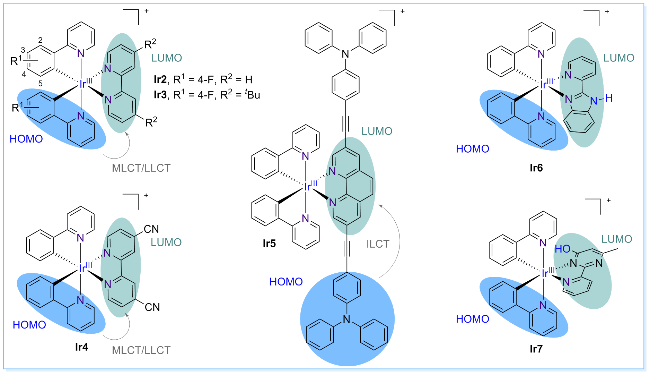
图4 氟取代(F)、氰基(CN)、TPA取代及含N—H、O—H的杂金属铱配合物IrIII(L1L1L2)及HOMO和LUMO的轨道分布Figure 4 Fluorine substitution (F), cyano group (CN) substitution, TPA (triphenylamine) substitution, and the presence of N—H and O—H groups of heterometallic iridium(III) complexes IrIII(L1L1L2), along with the distribution of HOMO and LUMO orbitals |
表1 所选环金属铱(III)光敏剂的氧化还原电位Table 1 Redox potentials of selected cyclometalated iridium(III) photosensitizers |
| Photocatalyst | Eoxa/V | Ereda/V | E0-0b/eV | $E_{\operatorname{ox}}^{*}$c/V | $E_{\operatorname{red}}^{*}$c/V | Ref. |
|---|---|---|---|---|---|---|
| Ir1 | +0.87 | -1.70 | 2.00 | -1.13 | +0.22 | [11a, 11e] |
| Ir2 | +1.51 | -1.37 | 2.40 | -0.89 | +1.03 | [19f] |
| Ir3 | +1.49 | -1.45 | 2.52 | -1.04 | +1.07 | [19f] |
| Ir4 | +0.83 | -1.10 | 1.94 | -1.11 | +0.84 | [19e, 19h] |
| Ir6 | +0.87 | -1.65 | —d | —d | —d | [22a] |
| Ir7 | +0.87 | -1.33 | —d | —d | —d | [22b] |
| Ir8 | +0.91 | -1.80 | 2.05 | -1.14 | +0.25 | [11a] |
| Ir9 | +1.31 | -1.73 | 2.47 | -1.16 | +0.74 | [9, 11a] |
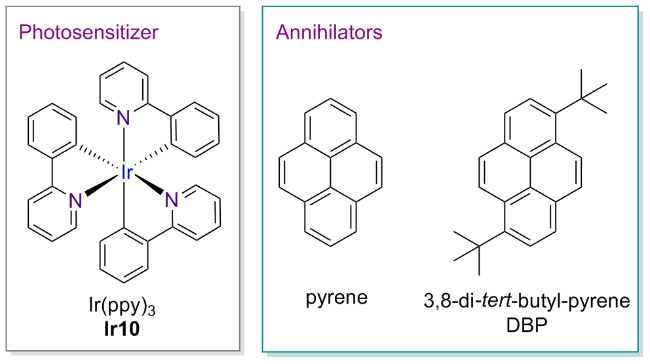
| Ir10 | +0.36 | -2.60 | 2.28 | -1.92 | +0.32 | [2c, 11c] |
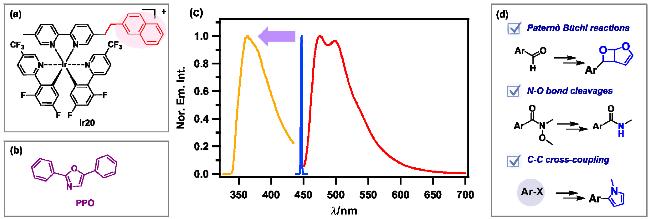
| Ir20 | +1.55 | —d | —d | —d | —d | [8a] |
a The redox potential in acetonitrile solution, expressed as V vs Fc+/0 (where VFc+/Fc=VSCE-0.41 V). b The energy gap between the ground state and the excited state in acetonitrile solution, estimated with an error margin of ±0.1 eV. c Values obtained from Eq. 1 and Eq. 2. d No relevant reports available. |
2 [Ir(C^N)2(N^N)]+类环金属铱(III)配合物的应用
表2 所选环金属铱(III)光敏剂的光物理性质Table 2 Photophysical properties of selected cyclometalated iridium(III) photosensitizers |
| Photo- catalyst | λemmax a/nm | τTb/μs | ET/eV | Ref. |
|---|---|---|---|---|
| Ir1 | 602 | 0.275 | 2.29 | [11e] |
| Ir2 | 557 | 0.224 | 2.23 | [19g] |
| Ir3 | 540 | —c | 2.30 | [19f] |
| Ir4 | 750 | 0.027 | —c | [19h] |
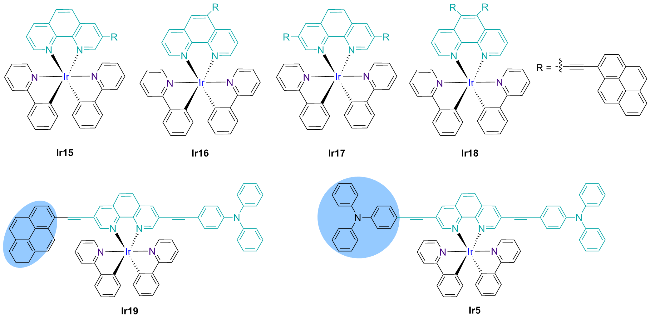
| Ir5 | 666d | 2.3d | 2.07 | [23c] |
| Ir6 | 588e | 0.1895e | —c | [22a] |
| Ir7 | 588f | τ1=0.0092f τ2=0.2156f | —c | [22b] |
| 611g | τ1=0.0354g τ2=0.1g | |||
| Ir8 | 581 | 0.557 | 2.13 | [11c] |
| Ir9 | 470 | 2.3 | 2.61 | [11a, 11c, 19f] |
| Ir10 | 518 | 1.9 | 2.39 | [11c, 19f,] |
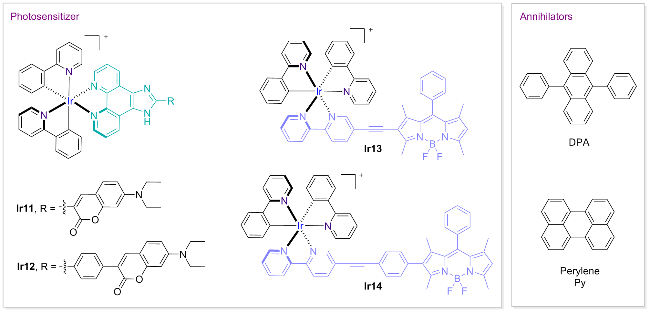
| Ir11 | 603h | 75.5h | 2.06 | [25] |
| Ir12 | 608h | 73.6h | 2.06 | [25] |
| Ir13 | 553/742h | 87.2h | 2.10 | [24a] |
| Ir14 | 514h | 23.7h | 2.43 | [24a] |
| Ir15 | 672/747d,h | 157.2d,h | 1.86 | [23b] |
| Ir16 | 678/751d,h | 363.7d,h | 1.85 | [23b] |
| Ir17 | 682/757d,h | 85.8d,h | 1.85 | [23b] |
| Ir18 | 600/738d,h | 30.7d,h | 2.15 | [23b] |
| Ir19 | 677d | 53.3d | 2.57 | [23c] |
| Ir20 | 474h | 72.1h | 2.86 | [8a] |
a The maximum emission peak, unless otherwise specified, was measured in deoxygenated acetonitrile with 298 K. b The triplet excited state lifetime at room temperature, unless otherwise specified, was measured in deoxygenated acetonitrile with 298 K. c No relevant reports available. d The solvent used was deoxygenated dichloromethane (DCM). e 296 K. f Measured in acetonitrile solution with [Ir7]=0.1 mmol/L. g Measured in acetonitrile solution with [Ir7]=1.0 mmol/L. h 293 K. |
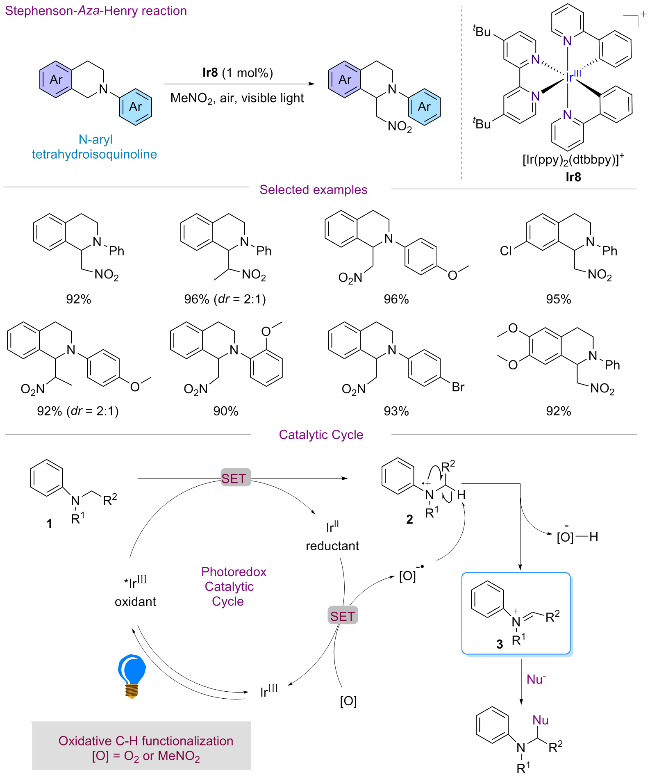
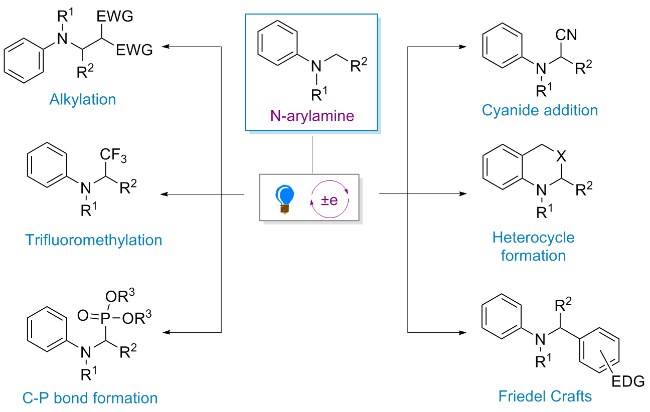
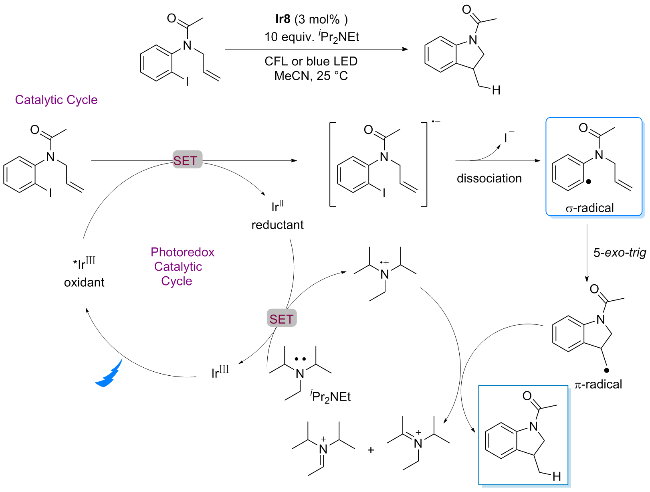
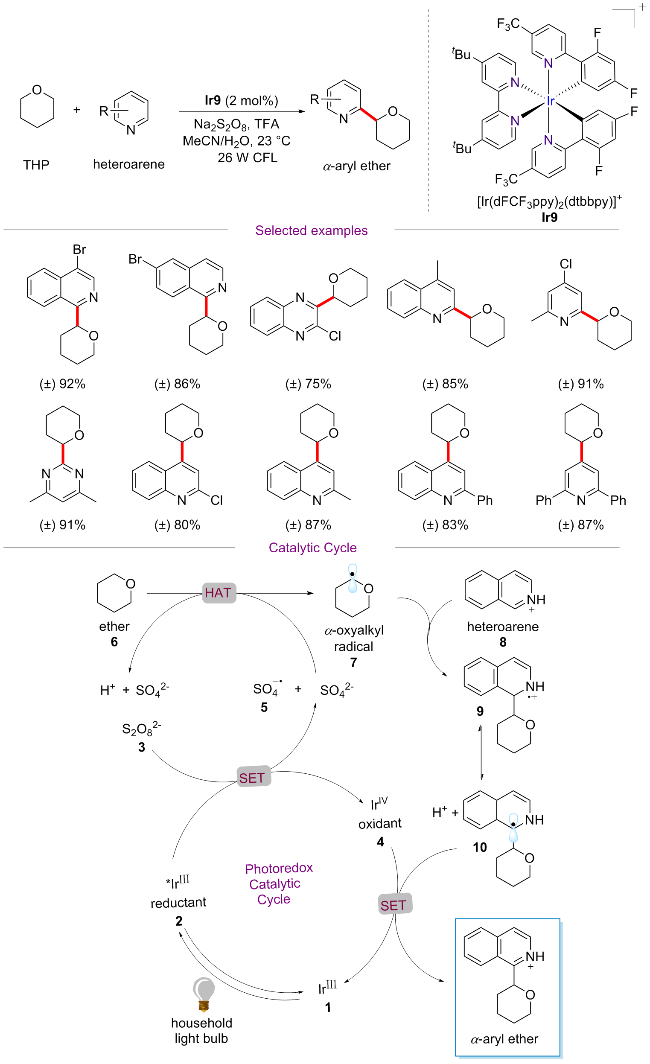
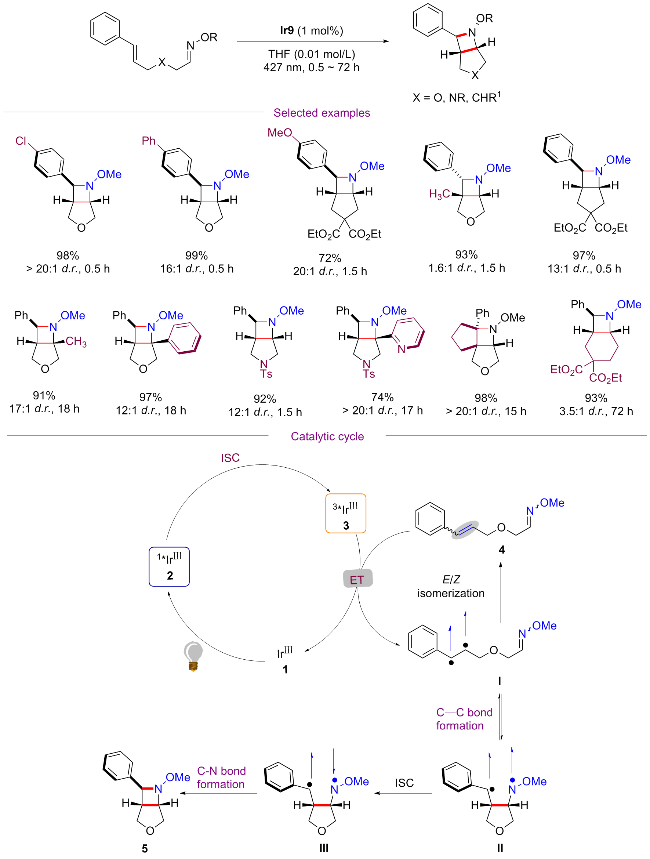
2.1 环金属铱配合物在光催化合成领域的应用
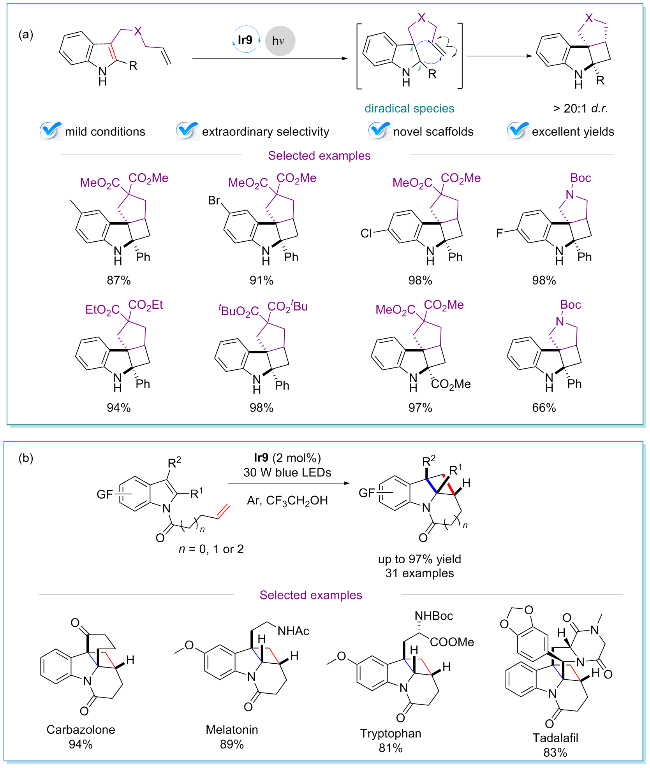
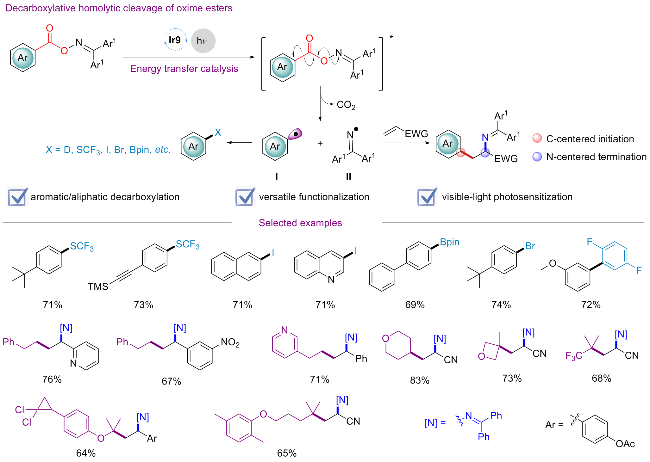
图11 光催化介导的能量转移催化吲哚衍生物的去芳香分子内[2+2]环加成反应Figure 11 Photocatalytic intramolecular dearomative [2+2] cycloaddition of indole derivatives induced by the energy transfer process (a) Indole derivatives tethered with olefins at the C3 position; (b) Indole derivatives tethered with olefins at the N1 position |
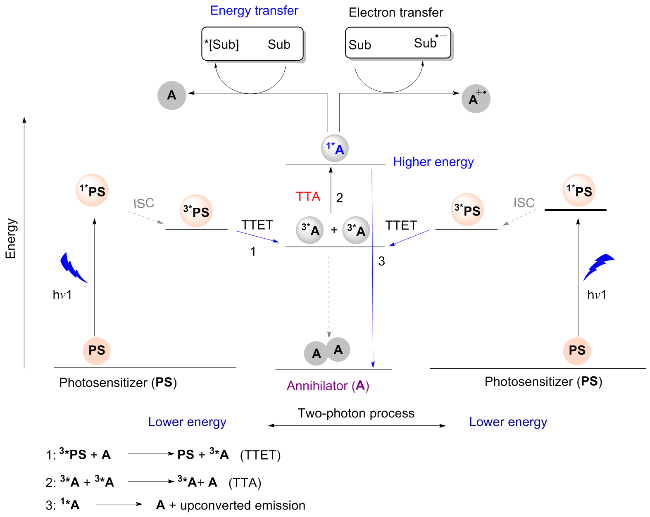
2.2 环金属铱配合物在TTA-UC领域中的应用
图18 IrIII络合物Ir20 (a)和三重态湮灭剂PPO (b)的分子结构; (c) Ir20的荧光谱图(红色)和Ir20/PPO上转换体系在447 nm(蓝色)激发下的上转换发光光谱(黄色); (d) Ir20/PPO上转换体系驱动的光反应Figure 18 Molecular structures of IrIII complex Ir20 (a) and triplet annihilator PPO (b); (c) Fluorescence spectrum of Ir20 (red) and upconversion luminescence spectrum of the Ir20/PPO system excited at 447 nm (yellow) upon upconversion; (d) Photochemical reaction driven via the Ir20/PPO upconversion system |