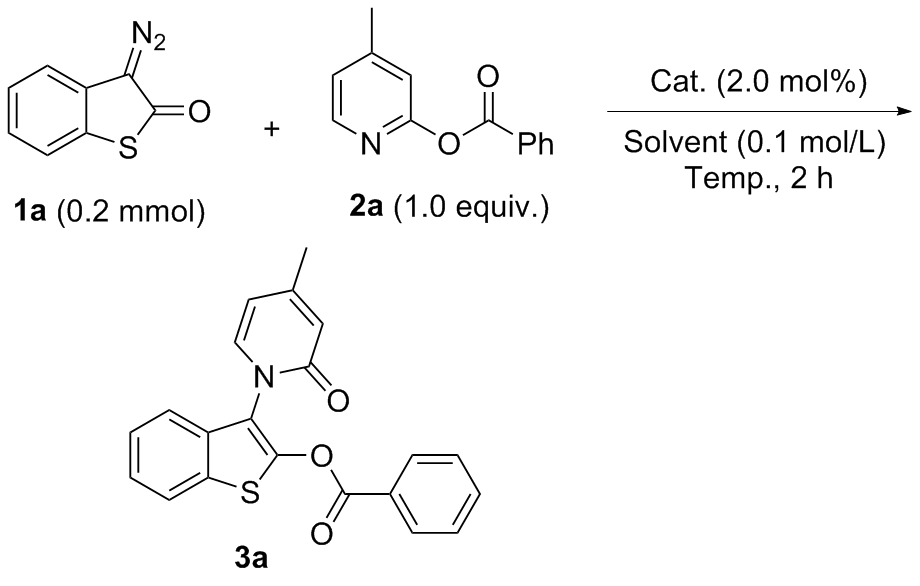
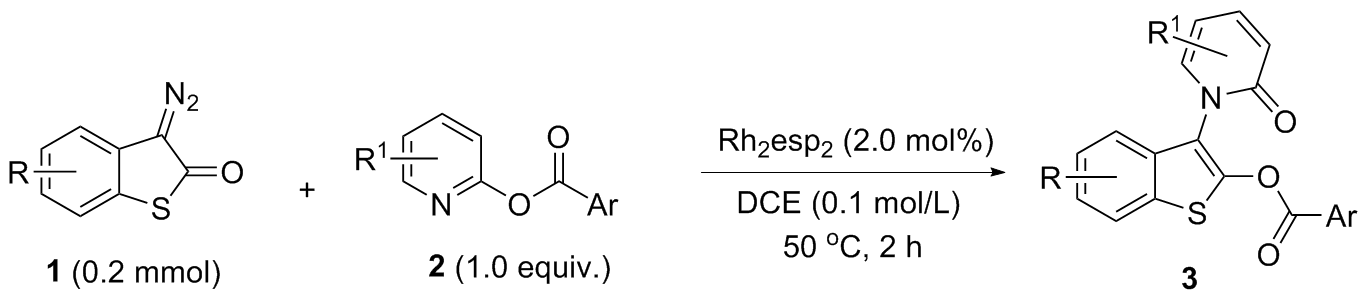
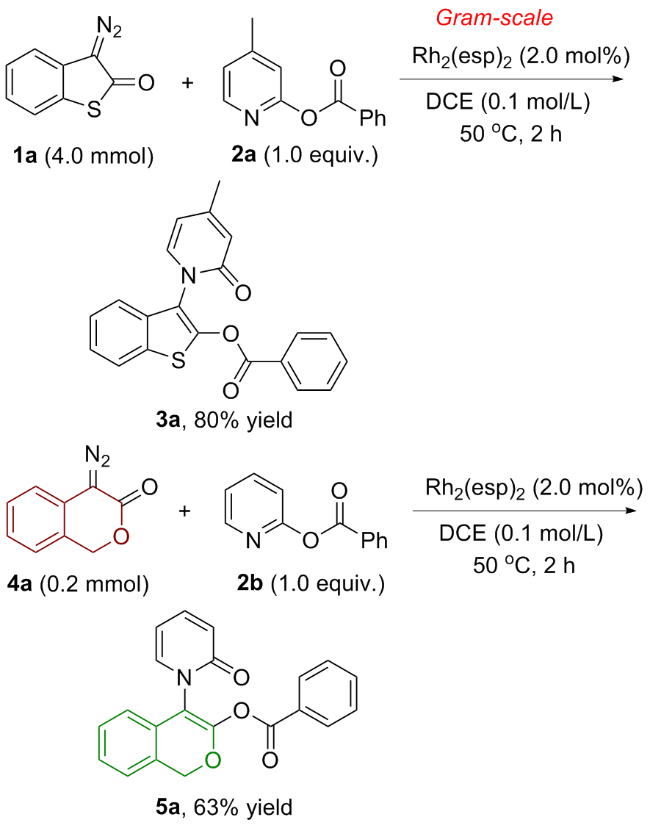
氮气氛围下, 在预先干燥的10 mL Schlenk管中依次加入Rh2(esp)2 (0.004 mmol)、硫代重氮氧化吲哚衍生物1 (0.2 mmol)、吡啶类苯甲酸酯衍生物2 (0.2 mmol)和无水1,2-二氯乙烷(2.0 mL). 50 ℃反应2 h, 用TLC监测原料反应完毕, 柱层析硅胶柱中纯化[淋洗剂: V(石油醚)∶V(乙酸乙酯)=3∶1~1.5∶1], 减压浓缩纯化得到目标产物3.
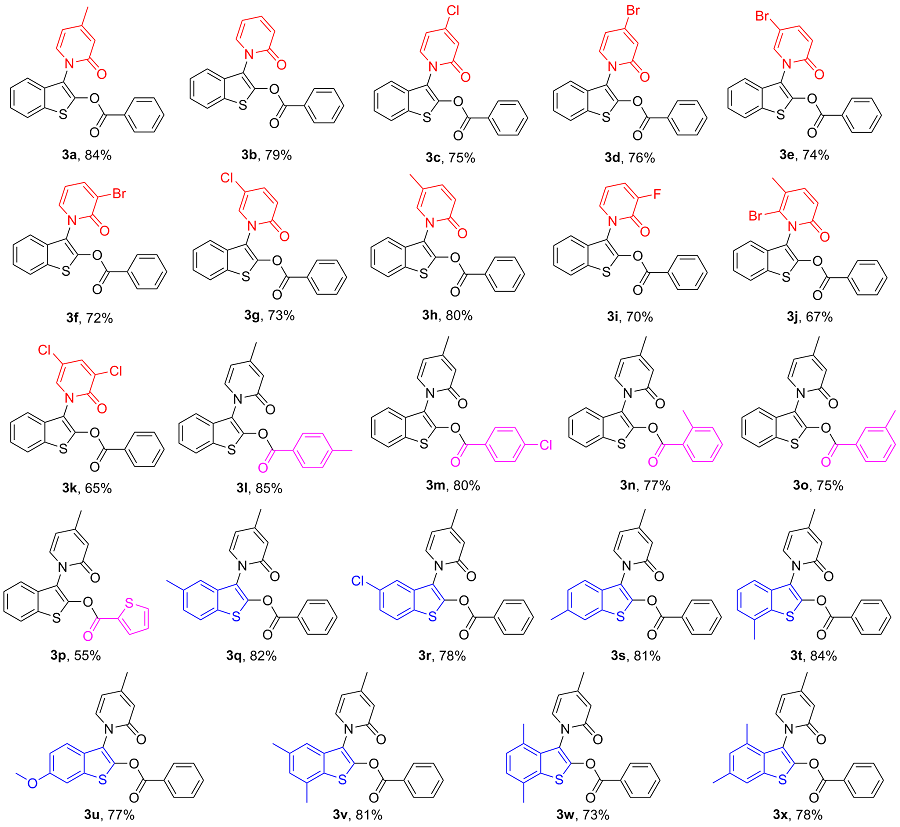
3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3a): 60.6 mg, 黄色固体, 产率84%. m.p. 173~178 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.08~8.06 (m, 2H), 7.80~7.78 (m, 1H), 7.50~7.38 (m, 5H), 7.26~7.24 (m, 1H), 6.58~6.57 (m, 1H), 6.16~6.14 (m, 1H), 2.29 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.5, 161.8, 152.2, 146.3, 137.3, 134.5, 132.6, 131.6, 130.5, 128.9, 127.5, 125.3, 125.1, 122.6, 120.7, 120.2, 119.0, 109.0, 21.6; IR (ATR) ν: 1744, 1373, 1250, 1103, 1049, 856, 756 cm-1; HRMS (ESI) calcd for C21H16NO3S [M+H]+ 362.0851, found 362.0854.
3-[2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3b): 58.3 mg, 黄色固体, 产率79%. m.p. 156~158 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.06 (d, J=6.4 Hz, 2H), 7.82~7.80 (m, 1H), 7.66~7.63 (m, 1H), 7.53~7.26 (m, 7H), 6.82 (d, J=7.2 Hz, 1H), 6.33 (t, J=5.2 Hz, 1H); 13C NMR (100 MHz, CDCl3) δ: 162.4, 161.8, 146.3, 140.4, 138.7, 134.6, 132.6, 131.4, 130.5, 128.9, 127.5, 125.4, 125.2, 122.7, 122.2, 120.6, 119.4, 106.3; IR (ATR) ν: 1744, 1389, 1250, 1111, 1018, 910, 756 cm-1; HRMS (ESI) calcd for C20H13NO3SNa [M+Na]+ 370.0514, found 370.0517.
3-[4-氯-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3c): 57.2 mg, 黄色固体, 产率75%. m.p. 174~175 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.08~8.06 (m, 2H), 7.82~7.80 (m, 1H), 7.68~7.64 (m, 1H), 7.53~7.49 (m, 2H), 7.43~7.39 (m, 3H), 7.34~7.32 (m, 1H), 6.84~6.83 (m, 1H), 6.37~6.34 (m, 1H); 13C NMR (100 MHz, CDCl3) δ: 162.4, 160.4, 147.6, 146.7, 138.6, 134.7, 132.6, 131.2, 130.5, 129.0, 127.3, 125.5, 125.3, 122.7, 120.5, 120.4, 118.0, 108.3; IR (ATR) ν: 1744, 1674, 1597, 1304, 1134, 756, 702 cm-1; HRMS (ESI) calcd for C20H13Cl- NO3S [M+H]+ 382.0305, found 382.0296.
3-[4-溴-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3d): 64.6 mg, 黄色固体, 产率76%. m.p. 169~172 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.08~8.05 (m, 2H), 7.81~7.79 (m, 1H), 7.68~7.64 (m, 1H), 7.52~7.49 (m, 2H), 7.42~7.39 (m, 3H), 7.26~7.24 (m, 1H), 7.033~7.028 (m, 1H), 6.48~6.46 (m, 1H); 13C NMR (100 MHz, CDCl3) δ: 162.4, 160.2, 146.7, 138.3, 136.5, 134.7, 132.6, 131.1, 130.5, 129.0, 127.3, 125.5, 124.1, 122.7, 120.4, 118.0, 110.7; IR (ATR) ν: 1744, 1667, 1520, 1196, 1080, 1049, 702 cm-1; HRMS (ESI) calcd for C20H13BrNO3S [M+H]+ 425.9800, found 425.9805.
3-[5-溴-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3e): 62.8 mg, 黄色固体, 产率74%. m.p. 169~171 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.08~8.06 (m, 2H), 7.82~7.79 (m, 1H), 7.68~7.64 (m, 1H), 7.55~7.48 (m, 4H), 7.45~7.40 (m, 3H), 6.72~6.70 (m, 1H); 13C NMR (125 MHz, CDCl3) δ: 162.2, 160.2, 146.6, 143.5, 138.2, 134.7, 132.5, 130.8, 130.5, 129.0, 127.3, 125.6, 125.3, 123.2, 122.7, 120.4, 117.8, 98.3; IR (ATR) ν: 1744, 1674, 1597, 1304, 1134, 756, 702 cm-1; HRMS (ESI) calcd for C20H13BrNO3S [M+H]+ 425.9800, found 425.9793.
3-[3-溴-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3f): 61.1 mg, 黄色固体, 产率72%. m.p. 170~172 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.06~8.03 (m, 2H), 7.92~7.89 (m, 1H), 7.81~7.79 (m, 1H), 7.65~7.63 (m, 1H), 7.51~7.47 (m, 2H), 7.42~7.39 (m, 4H), 6.24~6.21 (m, 1H); 13C NMR (100 MHz, CDCl3) δ: 162.3, 157.9, 146.5, 142.2, 138.2, 134.6, 132.5, 131.0, 130.5, 128.9, 127.3, 125.5, 125.2, 122.7, 120.5, 118.7, 117.8, 106.1; IR (ATR) ν: 1744, 1667, 1543, 1520, 1250, 795, 702 cm-1; HRMS (ESI) calcd for C20H13BrNO3S [M+H]+ 425.9800, found 425.9807.
3-[5-氯-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3g) 55.6 mg, 黄色固体, 产率73%. m.p. 131~133 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.08~8.05 (m, 2H), 7.82~7.79 (m, 1H), 7.67~7.63 (m, 1H), 7.52~7.39 (m, 7H), 6.77~6.75 (m, 1H); 13C NMR (100 MHz, CDCl3) δ: 162.2, 160.1, 146.7, 141.5, 135.9, 134.7, 132.6, 130.9, 130.5, 129.0, 127.3, 125.5, 125.3, 122.8, 122.7, 120.3, 117.9, 112.8; IR (ATR) ν: 1744, 1311, 1242, 1018, 957, 795, 702 cm-1; HRMS (ESI) calcd for C20H13ClNO3S [M+H]+ 382.0305, found 382.0298.
3-[5-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3h): 57.8 mg, 黄色固体, 产率80%. m.p. 162~164 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.08~8.06 (m, 2H), 7.81~7.78 (m, 1H), 7.66~7.62 (m, 1H), 7.45~7.34 (m, 6H), 7.14~7.13 (m, 1H), 6.73 (d, J=9.2 Hz, 1H), 2.11 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.4, 161.2, 146.2, 143.2, 135.6, 134.5, 132.7, 131.5, 130.5, 128.9, 127.6, 125.3, 125.1, 122.6, 121.7, 120.6, 119.1, 115.2, 17.0; IR (ATR) ν: 1744, 1389, 1204, 1080, 825, 756, 702 cm-1; HRMS (ESI) calcd for C21H16NO3S [M+H]+ 362.0851, found 362.0852.
3-[3-氟-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3i): 51.1 mg, 黄色固体, 产率70%. m.p. 168~171 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.07~8.04 (m, 2H), 7.82~7.80 (m, 1H), 7.67~7.63 (m, 1H), 7.51~7.40 (m, 5H), 7.28~7.20 (m, 2H), 6.27~6.23 (m, 1H); 13C NMR (100 MHz, CDCl3) δ: 162.3, 155.6 (d, JC-F=21.0 Hz), 153.0 (d, JC-F=200.0 Hz), 146.7, 134.7, 134.0 (d, JC-F=4.0 Hz), 132.6, 131.0, 130.5, 128.9, 127.3, 125.5, 125.3, 122.7, 120.6 (d, JC-F=13.0 Hz), 120.4, 117.8 (d, JC-F=1.0 Hz), 104.1 (d, JC-F=5.0 Hz); 19F NMR (376 MHz, CDCl3): δ: -128.27; IR (ATR) ν: 1744, 1620, 1242, 1180, 1080, 748, 702 cm-1; HRMS (ESI) calcd for C20H13FNO3S [M+H]+ 366.0600, found 366.0604.
3-[5-甲基-6-溴-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3j): 58.7 mg, 黄色固体, 产率67%. m.p. 181~183 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.10~8.07 (m, 2H), 7.81~7.78 (m, 1H), 7.67~7.63 (m, 1H), 7.57 (s, 1H), 7.52~7.48 (m, 2H), 7.45~7.39 (m, 3H), 6.70~6.69 (m, 1H), 2.36 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.3, 160.5, 151.6, 146.6, 137.6, 134.6, 132.5, 131.1, 130.5, 128.9, 127.4, 125.5, 125.2, 122.7, 121.1, 120.5, 117.7, 102.6, 22.9; IR (ATR) ν: 1744, 1597, 1204, 1080, 856, 756, 702 cm-1; HRMS (ESI) calcd for C21H15BrNO3S [M+H]+ 439.9956, found 439.9958.
3-[3,5-二氯-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3k): 54.0 mg, 黄色固体, 产率65%. m.p. 136~139 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.09~8.05 (m, 2H), 7.82~7.79 (m, 1H), 7.68~7.64 (m, 1H), 7.55~7.39 (m, 6H), 6.73~6.70 (m, 1H); 13C NMR (100 MHz, CDCl3) δ: 162.2, 160.1, 146.6, 143.4, 138.2, 134.7, 132.5, 130.9, 130.5, 129.0, 127.3, 125.5, 125.3, 123.2, 122.7, 120.4, 117.8, 98.2; IR (ATR) ν: 1744, 1674, 1520, 1203, 1080, 1049, 702 cm-1; HRMS (ESI) calcd for C20H12Cl2- NO3S [M+H]+ 415.9915, found 415.9919.
3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-4-甲基苯甲酸酯(3l): 63.8 mg, 黄色固体, 产率85%. m.p. 142~144 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.96~7.94 (m, 2H), 7.79~7.77 (m, 1H), 7.44~7.37 (m, 3H), 7.29~7.23 (m, 3H), 6.56~6.55 (m, 1H), 6.15~6.13 (m, 1H), 2.42 (s, 3H), 2.28 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.6, 161.8, 152.1, 146.3, 138.7, 137.4, 135.3, 132.6, 131.6, 131.1, 128.7, 127.6, 127.4, 125.3, 125.1, 122.6, 120.7, 120.2, 119.2, 108.8, 21.6, 21.3; IR (ATR) ν: 1744, 1674, 1373, 1172, 1049, 1018, 740 cm-1; HRMS (ESI) calcd for C22H18NO3S [M+H]+ 376.1007, found 376.1015.
3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-4-氯苯甲酸酯(3m): 63.2 mg, 黄色固体, 产率80%. m.p. 204~205 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.01~7.98 (m, 2H), 7.80~7.77 (m, 1H), 7.48~7.38 (m, 5H), 7.24~7.22 (m, 1H), 6.56~6.55 (m, 1H), 6.16~6.14 (m, 1H), 2.28 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 161.7, 161.6, 152.2, 146.0, 141.2, 137.2, 132.6, 131.8, 131.5, 129.3, 126.0, 125.4, 125.2, 122.6, 120.7, 120.2, 119.1, 109.0, 21.6; IR (ATR) ν: 1744, 1667, 1589, 1373, 1080, 725, 679 cm-1; HRMS (ESI) calcd for C21H15ClNO3S [M+H]+ 396.0461, found 396.0471.
3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-2-甲基苯甲酸酯(3n): 57.8 mg, 黄色固体, 产率77%. m.p. 165~167 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.02~7.99 (m, 1H), 7.81~7.78 (m, 1H), 7.50~7.46 (m, 1H), 7.44~7.381 (m, 3H), 7.374~7.23 (m, 3H), 6.57~6.56 (m, 1H), 6.16~6.14 (m, 1H), 2.61 (s, 3H), 2.28 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 162.8, 161.9, 152.2, 146.4, 142.1, 137.4, 133.7, 132.6, 132.1, 131.7, 131.6, 126.4, 126.2, 125.3, 125.0, 122.6, 120.5, 120.2, 118.7, 109.0, 22.0, 21.6; IR (ATR) ν: 1744, 1674, 1597, 1304, 1134, 756, 702 cm-1; HRMS (ESI) calcd for C22H18NO3S [M+H]+ 376.1007, found 376.1017.
3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-3-甲基苯甲酸酯(3o): 56.3 mg, 黄色固体, 产率75%. m.p. 131~133 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.88~7.84 (m, 2H), 7.80~7.77 (m, 1H), 7.45~7.34 (m, 5H), 7.26~7.24 (m, 1H), 6.57~6.56 (m, 1H), 6.16~6.14 (m, 2H), 2.40 (s, 3H), 2.28 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.3, 161.8, 152.1, 146.3, 138.7, 137.4, 135.3, 132.6, 131.6, 131.1, 128.8, 127.6, 127.4, 125.3, 125.1, 122.6, 120.7, 120.2, 119.2, 108.9, 21.6, 21.3; IR (ATR) ν: 1744, 1674, 1265, 1180, 1003, 887, 756 cm-1; HRMS (ESI) calcd for C22H18NO3S [M+H]+ 376.1007, found 376.1008.
3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-1-噻吩基甲酸酯(3p): 40.4 mg, 黄色固体, 产率 55%. m.p. 197~199 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.91~7.90 (m, 1H), 7.79~7.77 (m, 1H), 7.71~7.69 (m, 1H), 7.45~7.36 (m, 3H), 7.25~7.23 (m, 1H), 7.17~7.14 (m, 1H), 6.56~6.55 (m, 1H), 6.17~6.15 (m, 1H), 2.29 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 161.8, 157.8, 152.1, 145.9, 137.4 136.1, 135.2, 132.5, 131.5, 130.4, 128.4 125.3, 125.1, 122.6, 120.8, 120.2, 119.1, 108.9, 21.6; IR (ATR) ν: 1736, 1667, 1520, 1412, 1150, 725, 656 cm-1; HRMS (ESI) calcd for C19H14NO3S2 [M+H]+ 368.0415, found 368.0421.
5-甲基-3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩- 2-基-苯甲酸酯(3q): 61.5 mg, 黄色固体, 产率82%. m.p. 165~167 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.07~8.05 (m, 2H), 7.76~7.61 (m, 2H), 7.50~7.45 (m, 2H), 7.24~7.19 (m, 3H), 6.57~6.56 (m, 1H), 6.16~6.14 (m, 1H), 2.42 (s, 3H), 2.29 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.5, 161.8, 152.1, 146.4, 137.4, 135.3, 134.4, 131.8, 130.5, 129.8, 128.8, 127.6, 126.8, 122.3, 120.6, 120.2, 118.9, 108.9, 21.61, 21.59; IR (ATR) ν: 1744, 1535, 1250, 1173, 1018, 795, 702 cm-1; HRMS (ESI) calcd for C22H18- NO3S [M+H]+ 376.1007, found 376.1003.
5-氯-3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3r): 61.6 mg, 黄色固体, 产率78%. m.p. 179~181 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.07~8.04 (m, 2H), 7.72~7.70 (m, 1H), 7.67~7.63 (m, 1H), 7.51~7.47 (m, 2H), 7.413~7.408 (m, 1H), 7.36~7.33 (m, 1H), 7.23~7.22 (m, 1H), 6.60~6.59 (m, 1H), 6.20~6.17 (m, 1H), 2.31 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.3, 161.6, 152.3, 147.6, 137.1, 134.7, 132.8, 131.8, 130.6, 130.5, 128.9, 127.3, 125.6, 123.8, 120.4, 120.3, 118.3, 109.1, 21.7; IR (ATR) ν: 1744, 1581, 1242, 1049, 964, 802, 702 cm-1; HRMS (ESI) calcd for C21H15ClNO3S [M+H]+ 396.0461, found 396.0464.
6-甲基-3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩- 2-基-苯甲酸酯(3s): 60.8 mg, 黄色固体, 产率81%. m.p. 178~180 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.08~8.05 (m, 2H), 7.63~7.62 (m, 1H), 7.59~7.58 (m, 1H), 7.50~7.46 (m, 2H), 7.33~7.31 (m, 1H), 7.25~7.19 (m, 2H), 6.56~6.55 (m, 1H), 6.15~6.13 (m, 1H), 2.47 (s, 3H), 2.28 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.5, 161.8, 152.2, 146.3, 137.3, 134.5, 132.6, 131.6, 130.5, 128.9, 127.5, 125.3, 125.1, 122.6, 120.7, 120.2, 119.0, 109.0, 21.67, 21.59; IR (ATR) ν: 1744, 1605, 1219, 1103, 1018, 810, 702 cm-1; HRMS (ESI) calcd for C22H18NO3S [M+H]+ 376.1007, found 376.1009.
7-甲基-3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩- 2-基-苯甲酸酯(3t): 63.0 mg, 黄色固体, 产率84%. m.p. 155~157 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.09~8.06 (m, 2H), 7.66~7.62 (m, 1H), 7.50~7.46 (m, 2H), 7.34~7.18 (m, 4H), 6.56~6.55 (m, 1H), 6.15~6.13 (m, 2H), 2.56 (s, 3H), 2.28 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.6, 161.7, 152.1, 146.1, 137.3, 134.5, 132.5, 132.1, 131.5, 130.5 128.8, 127.6, 125.6, 125.5, 120.2, 119.8, 118.4, 108.3, 21.6, 19.5; IR (ATR) ν: 1744, 1597, 1242, 1118, 972, 779, 702 cm-1; HRMS (ESI) calcd for C22H18- NO3S [M+H]+ 376.1007 found 376.1009.
6-甲氧基-3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻 吩-2-基-苯甲酸酯(3u): 60.2 mg, 黄色固体, 产率77%. m.p. 149~151 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.07~8.04 (m, 2H), 7.65~7.61 (m, 1H), 7.50~7.45 (m, 2H), 7.33~7.31 (m, 1H), 7.26~7.23 (m, 2H), 7.02~6.99 (m, 1H), 6.55~6.54 (m, 1H), 6.14~6.12 (m, 1H), 3.87 (s, 3H), 2.27 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.5, 161.8, 152.1, 146.4, 145.6, 137.4, 132.6, 131.6, 130.6, 129.6, 125.2, 125.0, 124.7, 122.6, 120.7, 120.2, 118.9, 108.9, 21.9, 21.6; IR (ATR) ν: 1744, 1667, 1366, 1103, 1018, 810, 702 cm-1; HRMS (ESI) calcd for C22H18NO4S [M+H]+ 392.0957, found 392.0964.
5,7-二甲基-3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3v): 63.0 mg, 黄色固体, 产率81%. m.p. 155~158 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.07~8.05 (m, 2H), 7.64~7.61 (m, 1H), 7.49~7.45 (m, 2H), 7.24~7.22 (m, 1H), 7.06~7.03 (m, 2H), 6.57~6.56 (m, 1H), 6.15~6.13 (m, 1H), 2.51 (s, 3H), 2.39 (s, 3H), 2.28 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.7, 161.8, 152.1, 146.2, 137.4, 135.6, 134.4, 131.8, 131.6, 130.5, 129.7, 128.8, 127.7, 127.4, 120.2, 119.7, 118.2, 108.8, 21.6, 21.5, 19.3; IR (ATR) ν: 1744, 1535, 1450, 1172, 1018, 848, 748 cm-1; HRMS (ESI) calcd for C23H19NO3S [M+H]+ 390.1164, found 390.1165.
4,7-二甲基-3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯3w): 56.8 mg, 黄色固体, 产率73%. m.p. 168~170 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.04~8.01 (m, 2H), 7.65~7.62 (m, 1H), 7.49~7.45 (m, 2H), 7.23~7.21 (m, 1H), 7.09~7.04 (m, 2H), 6.54~6.53 (m, 1H), 6.15~6.12 (m, 1H), 2.52 (s, 3H), 2.29 (s, 3H), 2.23 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 163.0, 162.8, 152.4, 146.7, 137.8, 134.4, 133.1, 130.4, 129.7, 129.6, 128.8, 127.9, 127.6, 125.2, 121.0, 120.0, 108.6, 21.7, 19.2, 18.1; IR (ATR) ν: 1744, 1674, 1597, 1304, 1134, 756, 702 cm-1; HRMS (ESI) calcd for C23H20NO3S [M+H]+ 390.1164, found 390.1178.
5,6-二甲基-3-[4-甲基-2-氧代吡啶-1(2H)-基]苯并噻吩-2-基-苯甲酸酯(3x): 62.7 mg, 黄色固体, 产率78%. m.p. 215~216 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.01~7.99 (m, 2H), 7.64~7.60 (m, 1H), 7.48~7.42 (m, 3H), 7.23~7.21 (m, 1H), 6.95 (s, 1H), 6.55 (s, 1H), 6.15~6.13 (m, 1H), 2.42 (s, 3H), 2.22 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 163.1, 162.7, 152.6, 145.8, 137.9, 134.9, 134.4, 133.5, 131.7, 130.4, 130.0, 129.4, 128.8, 128.3, 127.6, 120.2, 120.0, 108.9, 21.7, 21.4, 18.4; IR (ATR) ν: 1744, 1674, 1597, 1304, 1134, 756, 702 cm-1; HRMS (ESI) calcd for C23H20NO3S [M+H]+ 404.0124, found 404.0150.
4-[2-氧代吡啶-1(2H)-基]-1H-异色烯-3-基苯甲酸酯(5a): 43.5 mg, 白色固体, 产率63%. m.p. 104~105 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.86~7.83 (m, 2H), 7.50~7.32 (m, 4H), 7.28~7.26 (m, 4H), 7.05 (d, J=7.6 Hz, 1H), 6.64 (d, J=9.2 Hz, 1H), 6.36~6.32 (m, 1H), 5.38~5.28 (m, 2H); 13C NMR (100 MHz, CDCl3) δ: 183.7, 165.8, 163.4, 140.6, 136.6, 135.4, 133.0, 132.7, 130.4, 129.3, 129.0, 128.9, 128.2, 127.0, 125.1, 120.8, 107.4, 70.4; IR (ATR) ν: 1744, 1311, 1242, 1018, 957, 795, 702 cm-1; HRMS (ESI) calcd for C21H15NO4Na [M+Na]+ 368.0899, found 368.0903.
辅助材料(Supporting Information) 化合物
3a~
3x以及
5a的
1H NMR、
13C NMR谱图, 和产物
3i的
19F NMR谱图. 这些材料可以免费从本刊网站(
http://sioc-journal.cn/)上下载.