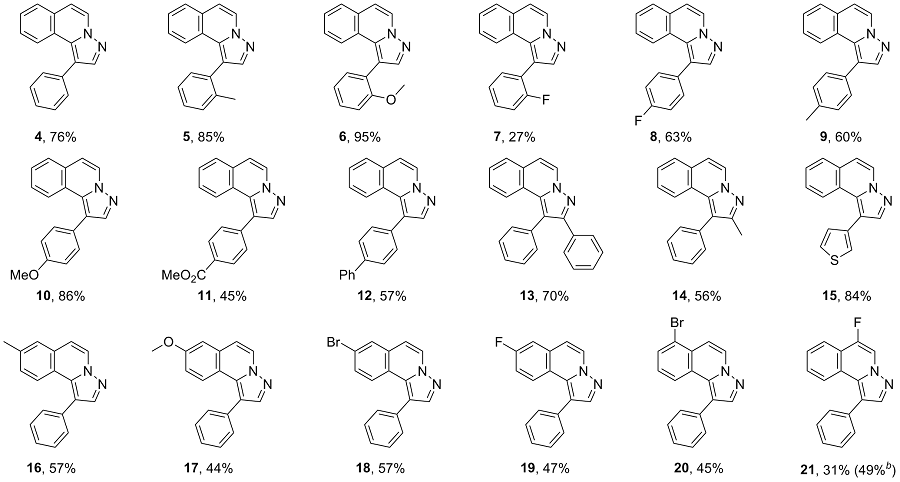
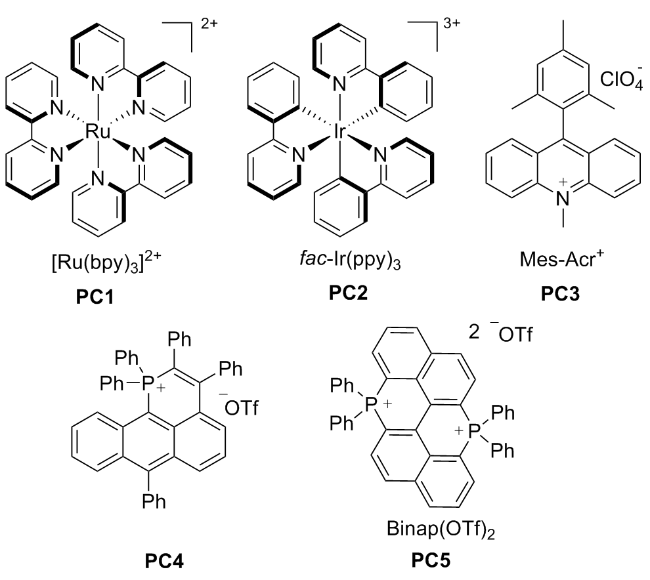
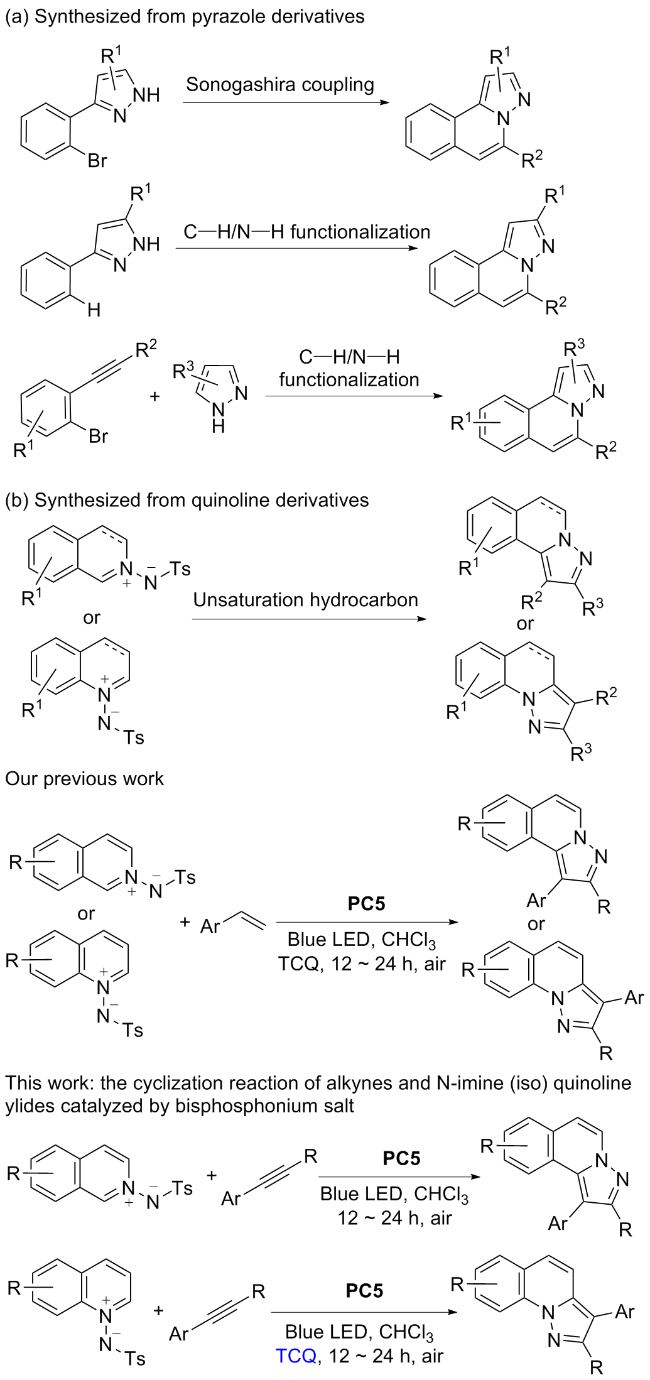
将2a (0.1 mmol), 苯乙炔(0.25 mmol), Binap(OTf)2 (0.5% mmol)和CHCl3 (1 mL)置于10 mL光催管中, 在430 nm, 6 W的光照下常温搅拌12~24 h. 全部产物均通过薄层色谱[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得到.
1-苯基吡唑并[5,1-a]异喹啉(4): 黄色油状物, 产率76%. 1H NMR (CDCl3, 300 MHz) δ: 8.28 (d, J=7.4 Hz, 1H), 8.06 (d, J=8.2 Hz, 1H), 7.94 (s, 1H), 7.71 (d, J=7.8Hz, 1H), 7.59 (d, J=4.2 Hz, 2H), 7.54~7.44 (m, 4H), 7.37~7.32 (m, 1H), 7.01 (d, J=7.4 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 142.0, 134.1, 133.7, 120.0, 129.4, 128.7, 127.8, 127.4, 127.3, 127.2, 126.6, 125.2, 123.4, 116.8, 112.4; HRMS (ESI) calcd for C17H13N2 [M+H]+245.1074, found 245.1085.
1-(2-甲基苯基)-吡唑并[5,1-a]异喹啉(5): 黄色油状物, 产率85%. 1H NMR (CDCl3, 300 MHz) δ: 8.30 (d, J=7.4 Hz, 1H), 7.88 (s, 1H), 7.71 (d, J=7.8 Hz, 1H), 7.56 (d, J=8.1 Hz, 1H), 7.50~7.45 (m, 1H), 7.41~7.37 (m, 3H), 7.35~7.28 (m, 2H), 7.03 (d, J=7.4 Hz, 1H), 2.17 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 141.6, 138.0, 134.1, 133.4, 131.2, 130.3, 129.3, 128.1, 127.7, 127.5, 127.1, 126.6, 126.1, 125.3, 123.2, 115.3, 112.3, 20.3; HRMS (ESI) calcd for C18H15N2 [M+H]+ 259.1230, found 259.1237.
1-(2-甲氧基苯基)-吡唑并[5,1-a]异喹啉(6): 黄色油状物, 产率95%. 1H NMR (CDCl3, 300 MHz) δ: 8.28 (d, J=7.4 Hz, 1H), 7.96 (s, 1H), 7.78 (d, J=8.2 Hz, 1H), 7.69 (d, J=7.8 Hz, 1H), 7.50~7.43 (m, 3H), 7.36~7.31 (m, 1H), 7.14~7.07 (m, 2H), 7.00 (d, J=7.4 Hz, 1H), 3.73 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 157.5, 142.3, 134.5, 132.1, 129.3, 129.2, 127.5, 127.0, 126.9, 126.7, 125.6, 123.9, 122.9, 120.8, 112.3, 112.1, 111.0, 55.4; HRMS (ESI) calcd for C18H15N2O [M+H]+ 275.1179, found 275.1187.
1-(2-氟苯基)-吡唑并[5,1-a]异喹啉(7): 黄色油状物, 产率27%. 1H NMR (CDCl3, 300 MHz) δ: 8.29 (d, J=7.4 Hz, 1H), 7.96 (s, 1H), 7.85 (d, J=8.3 Hz, 1H), 7.72 (d, J=7.8 Hz, 1H), 7.57~7.49 (m, 2H), 7.48~7.36 (m, 2H), 7.32~7.23 (m, 2H), 7.05 (d, J=7.4 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 160.5 (d, J=247.0 Hz), 142.2, 134.6, 132.4 (d, J=2.8 Hz), 129.6 (d, J=8.0 Hz), 129.5, 127.9, 127.4, 127.2, 126.6, 125.1, 124.4 (d, J=3.8 Hz), 123.4, 121.8 (d, J=16.0 Hz), 116.0 (d, J=22.1 Hz), 112.6, 109.2; 19F NMR (CDCl3, 282 MHz) δ: -111.40; HRMS (ESI) calcd for C17H12FN2 [M+H]+ 263.0980, found 263.0980.
1-(4-氟苯基)-吡唑并[5,1-a]异喹啉(8): 黄色油状物, 产率63%. 1H NMR (CDCl3, 300 MHz) δ: 8.27 (d, J=7.4 Hz, 1H), 7.97 (d, J=8.2 Hz, 1H), 7.90 (s, 1H), 7.71 (d, J=7.8 Hz, 1H), 7.56~7.47 (m, 3H), 7.39~7.33 (m, 1H), 7.24~7.17 (m, 2H), 7.02 (d, J=7.4 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 162.4 (d, J=246.6 Hz), 141.9, 133.8, 131.6 (d, J=7.9 Hz), 130.0 (d, J=3.1 Hz), 129.4, 127.9, 127.4, 127.3, 126.6, 125.0, 123.2, 115.72 (d, J=21.3 Hz), 115.67, 112.5; 19F NMR (CDCl3, 282 MHz) δ: -113.58; HRMS (ESI) calcd for C17H12FN2 [M+H]+ 263.0980, found 263.0980.
1-(4-甲基苯基)-吡唑并[5,1-a]异喹啉(9): 黄色油状物, 产率60%. 1H NMR (CDCl3, 300 MHz) δ: 8.27 (d, J=7.4 Hz, 1H), 8.09 (d, J=8.2 Hz, 1H), 7.91 (s, 1H), 7.70 (d, J=7.8 Hz, 1H), 7.51~7.46 (m, 3H), 7.38~7.31 (m, 3H), 7.00 (d, J=7.4 Hz, 1H), 2.48 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 142.0, 137.2, 133.7, 131.0, 129.8, 129.5, 129.4, 127.7, 127.2, 127.2, 126.6, 125.3, 123.4, 116.8, 112.3, 21.3; HRMS (ESI) calcd for C18H15N2 [M+H]+ 259.1230, found 259.1234.
1-(4-甲氧基苯基)-吡唑并[5,1-a]异喹啉(10): 黄色油状物, 产率86%. 1H NMR (CDCl3, 300 MHz) δ: 8.26 (d, J=7.4 Hz, 1H), 8.05 (d, J=8.2 Hz, 1H), 7.90 (s, 1H), 7.69 (d, J=7.8 Hz, 1H), 7.50~7.45 (m, 3H), 7.37~7.32 (m, 1H), 7.08~7.03 (m, 2H), 6.98 (d, J=7.4 Hz, 1H), 3.91 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 159.1, 142.0, 133.7, 131.1, 129.3, 127.7, 127.2, 127.2, 126.6, 126.2, 125.3, 123.3, 116.4, 114.2, 112.3, 55.4; HRMS (ESI) calcd for C18H15N2 [M+H]+ 275.1179, found 275.1180.
1-(4-乙酰氧基苯基)-吡唑并[5,1-a]异喹啉(11): 黄色固体, 产率45%. m.p. 253.2~254.1 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.26 (d, J=7.4 Hz, 1H), 8.05 (d, J=8.1 Hz, 1H), 7.90 (s, 1H), 7.69 (d, J=7.8 Hz, 1H), 7.57 (d, J=8.5 Hz, 2H), 7.51~7.46 (m, 1H), 7.38~7.33 (m, 1H), 7.24 (d, J=8.5 Hz, 2H), 7.00 (d, J=7.4 Hz, 1H), 2.37 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 169.6, 150.1, 141.9, 133.8, 131.6, 131.0, 129.4, 128.0, 127.4, 127.4, 126.5, 125.0, 123.4, 121.9, 115.9, 112.6, 21.2; HRMS (ESI) calcd for C19H15N2O2 [M+H]+ 303.1129, found 303.1128.
1-(1,1'-二苯基)-4-基-吡唑并[5,1-a]异喹啉(12): 黄色固体, 产率57%. m.p. 103.3~103.9 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.31~8.29 (m, 1H), 8.18 (d, J=8.2 Hz, 1H), 7.98 (d, J=1.0 Hz, 1H), 7.78~7.72 (m, 5H), 7.68~7.66 (m, 2H), 7.54~7.49 (m, 3H), 7.44~7.36 (m, 2H), 7.03 (d, J=7.4 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 142.0, 140.7, 140.2, 133.8, 133.1, 130.3, 129.5, 128.9, 127.9, 127.5, 127.4, 127.3, 127.3, 127.1, 126.7, 125.2, 123.5, 116.5, 112.5; HRMS (ESI) calcd for C23H17N2 [M+H]+ 321.1387, found 321.1393.
1,2-二苯基-吡唑并[5,1-a]异喹啉(13): 黄色固体, 产率70%. m.p. 135.3~134.7 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.32 (d, J=7.4 Hz, 1H), 7.71~7.63 (m, 2H), 7.58~7.55 (m, 2H), 7.53~7.47 (m, 6H), 7.44~7.40 (m, 1H), 7.29 (d, J=4.4 Hz, 3H), 7.02 (d, J=7.4 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 150.9, 135.6, 134.6, 133.1, 131.3, 129.5, 129.2, 128.4, 128.3, 127.8, 127.6, 127.3, 127.2, 126.3, 125.2, 123.4, 114.1, 112.4; HRMS (ESI) calcd for C23H17N2 [M+H]+ 321.1387, found 321.1390.
1-苯基-2-甲基-吡唑并[5,1-a]异喹啉(14): 黄色油状物, 产率56%. 1H NMR (CDCl3, 300 MHz) δ: 8.19 (d, J=7.4 Hz, 1H), 7.75 (d, J=8.2 Hz, 1H), 7.67 (d, J=7.9 Hz, 1H), 7.56~7.51 (m, 2H), 7.49~7.42 (m, 4H), 7.30~7.24 (m, 1H), 6.93 (d, J=7.4 Hz, 1H), 2.37 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 149.7, 134.7, 134.3, 130.8, 129.5, 128.8, 127.5, 127.5, 127.1, 127.0, 126.1, 124.8, 123.3, 114.9, 111.3, 12.3; HRMS (ESI) calcd for C18H15N2 [M+H]+259.1230, found 259.1232.
1-(3-噻吩基)-吡唑并[5,1-a]异喹啉(15): 深褐色固体, 产率84%. m.p. 75.5~76.3 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.28 (d, J=7.4 Hz, 1H), 8.09 (d, J=8.0 Hz, 1H), 7.94 (s, 1H), 7.72 (d, J=7.9 Hz, 1H), 7.54~7.48 (m, 2H), 7.43~7.41 (m, 2H), 7.31~7.29 (m, 1H), 7.02 (d, J=7.3 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 142.0, 134.2, 133.8, 129.4, 127.9, 127.4, 127.3, 126.6, 126.0, 125.2, 123.5, 123.5, 116.2, 112.5, 111.2; HRMS (ESI) calcd for C15H11N2S [M+H]+ 251.0638, found 251.0639.
1-苯基-8-甲基-吡唑并[5,1-a]异喹啉(16): 黄色固体, 产率57%. m.p. 75.8~76.2 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.25 (d, J=7.4 Hz, 1H), 7.96 (d, J=8.4 Hz, 1H), 7.91 (s, 1H), 7.60~7.57 (m, 2H), 7.54~7.41 (m, 4H), 7.19~7.16 (m, 1H), 6.95 (d, J=7.4 Hz, 1H), 2.47 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 141.9, 137.8, 134.3, 133.8, 130.0, 129.6, 128.7, 128.7, 127.3, 127.1, 126.6, 123.3, 122.9, 116.3, 112.2, 21.5; HRMS (ESI) calcd for C18H15N2 [M+H]+ 259.1230, found 259.1234.
1-苯基-8-甲氧基-吡唑并[5,1-a]异喹啉(17): 黄色油状物, 产率44%. 1H NMR (CDCl3, 300 MHz) δ: 8.26 (d, J=7.4 Hz, 1H), 7.98 (d, J=9.0 Hz, 1H), 7.90 (s, 1H), 7.59~7.45 (m, 5H), 7.11 (d, J=2.6 Hz, 1H), 6.99~6.93 (m, 2H), 3.90 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 142.0, 134.2, 133.9, 131.2, 123.0, 128.7, 127.3, 126.9, 125.0, 119.2, 116.6, 115.6, 112.1, 108.6, 55.4; HRMS (ESI) calcd for C18H15N2 [M+H]+ 275.1179, found 275.1179.
1-苯基-8-溴-吡唑并[5,1-a]异喹啉(18): 黄色固体, 产率57%. m.p. 80.8~82.1 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.29 (d, J=7.4 Hz, 1H), 7.94 (s, 1H), 7.90 (d, J=8.8 Hz, 1H), 7.84 (d, J=1.9 Hz, 1H), 7.55~7.40 (m, 6H), 6.91 (d, J=7.4 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 142.2, 133.6, 133.3, 131.0, 130.4, 129.8, 129.6, 128.9, 127.7, 127.6, 124.9, 123.8, 121.8, 117.1, 111.3; HRMS (ESI) calcd for C17H12BrN2 [M+H]+ 323.0179, found 323.0177.
1-苯基-8-氟-吡唑并[5,1-a]异喹啉(19): 黄色固体, 产率47%. m.p. 130.3~130.9 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.31 (d, J=7.4 Hz, 1H), 8.06~8.02 (m, 1H), 7.94 (s, 1H), 7.55~7.48 (m, 4H), 7.39~7.33 (m, 2H), 7.11~7.04 (m, 1H), 6.95 (d, J=7.4 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 161.8 (d, J=248.3 Hz), 142.1, 133.8, 133.5, 131.3 (d, J=9.0 Hz), 129.9, 128.9, 127.6, 127.5, 125.7 (d, J=8.7 Hz), 121.7 (d, J=1.9 Hz), 116.5, 115.8 (d, J=23.3 Hz), 112.2 (d, J=21.8 Hz), 111.7 (d, J=3.3 Hz); 19F NMR (CDCl3, 282 MHz) δ: -110.99; HRMS (ESI) calcd for C17H12FN2 [M+H]+ 263.0980, found 263.0980.
1-苯基-7-溴-吡唑并[5,1-a]异喹啉(20): 黄色油状物, 产率45%. 1H NMR (CDCl3, 300 MHz) δ: 8.34 (d, J=7.7 Hz, 1H), 8.01 (d, J=8.2 Hz, 1H), 7.96 (s, 1H), 7.75~7.73 (m, 1H), 7.56~7.49 (m, 4H), 7.48~7.45 (m, 2H), 7.19~7.13 (m, 1H); 13C NMR (CDCl3, 75 MHz) δ: 142.6, 133.7, 133.0, 131.8, 130.0, 128.9, 128.6, 127.8, 127.8, 127.7, 126.7, 122.8, 122.2, 117.4, 111.0; HRMS (ESI) calcd for C17H12BrN2 [M+H]+ 323.0179, found 323.0179.
1-苯基-6-氟-吡唑并[5,1-a]异喹啉(21): 黄色固体, 产率31% (49%). m.p. 105.3~106.1 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.30 (d, J=5.2 Hz, 1H), 8.07 (d, J=8.3 Hz, 1H), 7.97 (d, J=8.0 Hz, 1H), 7.91 (s, 1H), 7.61~7.54 (m, 3H), 7.52~7.45 (m, 3H), 7.43~7.40 (m, 1H); 13C NMR (CDCl3, 75 MHz) δ: 149.9 (d, J=241.2 Hz), 142.0 (d, J=2.6 Hz), 133.7, 131.9, 129.9, 128.8, 128.5, 127.9 (d, J=0.9 Hz), 127.6, 124.6 (d, J=6.0 Hz), 123.4 (d, J=2.8 Hz), 122.9 (d, J=18.7 Hz), 120.7 (d, J=5.0 Hz), 117.2, 112.9 (d, J=40.0 Hz); 19F NMR (CDCl3, 282 MHz) δ: -150.98; HRMS (ESI) calcd for C17H12FN2 [M+H]+ 263.0980, found 263.0978.
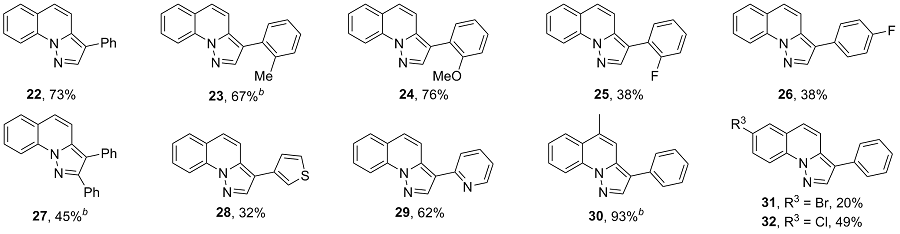
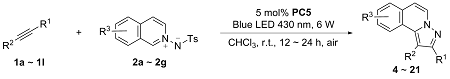
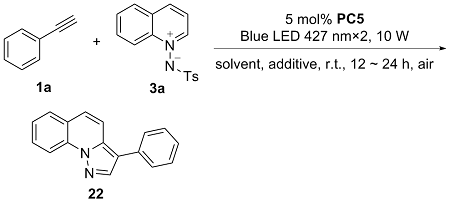
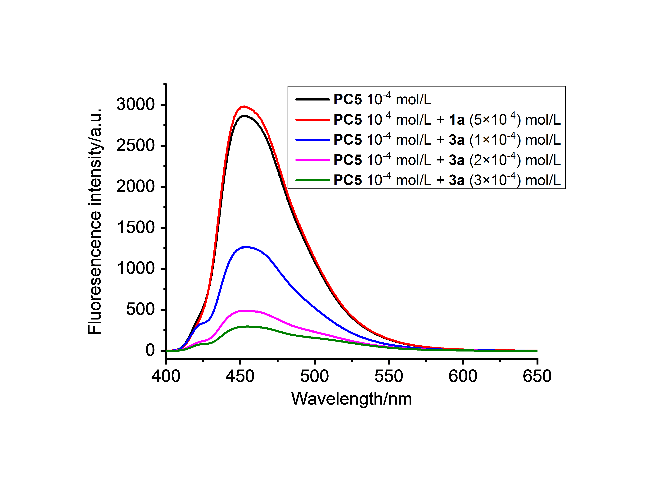
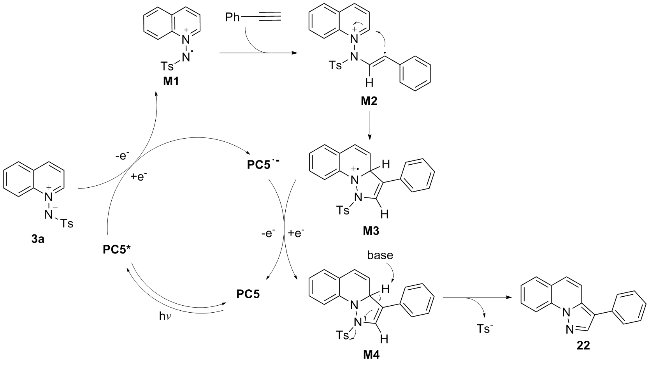
将3a (0.1 mmol), 苯乙炔(0.25 mmol), Binap(OTf)2 (0.5% mmol), TCQ (0.05 mmol)和CHCl3 (1 mL)置于10 mL Schlenk反应管中, 在427 nm, 10 W×2的光照下常温搅拌12~24 h. 全部产物均通过薄层色谱[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得到.
3-苯基-吡唑并[1,5-a]喹啉(22): 白色固体, 产率73%. m.p. 96.9~97.3 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.62 (d, J=8.4 Hz, 1H), 8.22 (s, 1H), 7.78 (d, J=8.0 Hz, 1H), 7.73~7.63 (m, 4H), 7.53~7.45 (m, 4H), 7.38~7.33 (m, 1H); 13C NMR (CDCl3, 75 MHz) δ: 139.9, 135.1, 134.7, 133.0, 129.6, 129.0, 128.3, 127.5, 126.6, 125.1, 124.9, 123.4, 115.9, 115.9, 115.5; HRMS (ESI) calcd for C17H13N2 [M+H]+ 245.1074, found 245.1079.
3-(2-甲基苯基)-吡唑并[1,5-a]喹啉(23): 白色固体, 产率67%. m.p. 76.1~77.2 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.64 (d, J=8.4 Hz, 1H), 8.09 (s, 1H), 7.79 (d, J=7.9 Hz, 1H), 7.74~7.69 (m, 1H), 7.50~7.45 (m, 1H), 7.42~7.34 (m, 4H), 7.33~7.30 (m, 2H), 2.37 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 141.1, 136.9, 135.6, 135.0, 131.8, 130.6, 129.5, 128.4, 127.4, 126.0, 124.8, 124.7, 123.4, 116.1, 115.4, 115.0, 20.6; HRMS (ESI) calcd for C18H15N2 [M+H]+ 259.1230, found 259.1223.
3-(2-甲氧基苯基)-吡唑并[1,5-a]喹啉(24): 黄色油状物, 产率76%. 1H NMR (CDCl3, 300 MHz) δ: 8.62 (d, J=8.4 Hz, 1H), 8.26 (s, 1H), 7.77 (d, J=7.7 Hz, 1H), 7.72~7.66 (m, 1H), 7.56 (d, J=9.4 Hz, 1H), 7.53~7.48 (m, 1H), 7.45~7.42 (m, J=3.2 Hz, 2H), 7.38~7.33 (m, 1H), 7.12~7.05 (m, 2H), 3.89 (s, 3H); 13C NMR (CDCl3, 75 MHz) δ: 156.6, 141.7, 135.5, 135.1, 130.3, 129.4, 128.2, 128.2, 124.6, 124.4, 123.4, 121.8, 120.9, 117.0, 115.4, 111.7, 111.3, 55.5; HRMS (ESI) calcd for C18H15- N2O [M+H]+ 275.1179, found 275.1186.
3-(2-氟苯基)-吡唑并[1,5-a]喹啉(25): 深褐色固体, 产率38%. m.p. 83.8~84.7 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.63 (d, J=8.4 Hz, 1H), 8.24 (d, J=1.3 Hz, 1H), 7.80 (d, J=8.0 Hz, 1H), 7.74~7.69 (m, 1H), 7.62~7.58 (m, 2H), 7.50 (d, J=9.3 Hz, 2H), 7.35~7.29 (m, 2H), 7.22 (d, J=9.5 Hz, 1H); 13C NMR (CDCl3, 75 MHz) δ: 159.7 (d, J=247.0 Hz), 141.2 (d, J=3.8 Hz), 135.4, 135.0, 130.2 (d, J=3.8 Hz), 129.6, 128.4, 128.3, 125.1 (d, J=21.8 Hz), 124.5 (d, J=3.5 Hz), 123.4, 120.8, 120.6, 116.3, 116.1 (d, J=16.0 Hz), 115.5, 109.3; 19F NMR (CDCl3, 282 MHz) δ: -113.55; HRMS (ESI) calcd for C17H12FN2 [M+H]+ 263.0980, found 263.0987.
3-(4-氟苯基)-吡唑并[1,5-a]喹啉(26): 深褐色固体, 产率38%. m.p. 96.2~97.1 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.61 (d, J=8.4 Hz, 1H), 8.16 (s, 1H), 7.79 (d, J=7.9 Hz, 1H), 7.74~7.69 (m, 1H), 7.65 (d, J=9.4 Hz, 1H), 7.60~7.56 (m, 2H), 7.51~7.46 (m, 2H), 7.22~7.16 (m, 2H); 13C NMR (CDCl3, 75 MHz) δ: 161.8 (d, J=249.6 Hz), 139.8, 135.0, 134.7, 129.7, 129.1 (d, J=2.7 Hz), 129.0 (d, J=7.7 Hz), 128.3, 125.3, 125.0, 123.4, 116.0 (d, J=21.5 Hz), 115.6, 115.5, 114.9; 19F NMR (CDCl3, 282 MHz) δ: -114.62; HRMS (ESI) calcd for C17H12FN2 [M+H]+ 263.0980, found 263.0976.
2,3-二苯基-吡唑并[1,5-a]喹啉(27): 黄色固体, 产率45%. m.p. 120.3~121.0 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.72 (d, J=8.4 Hz, 1H), 7.77~7.67 (m, 4H), 7.48~7.41 (m, 7H), 7.41~7.35 (m, 4H); 13C NMR (CDCl3, 75 MHz) δ: 150.6, 137.2, 134.8, 133.3, 133.0, 130.1, 129.4, 128.9, 128.7, 128.4, 128.3, 128.0, 126.9, 124.9, 124.7, 123.6, 115.8, 115.6, 113.3; HRMS (ESI) calcd for C23H17N2 [M+H]+ 321.1387, found 321.1389.
3-(3-噻吩基)-吡唑并[1,5-a]喹啉(28): 深褐色固体, 产率32%. m.p. 95.4~96.1 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.60 (d, J=8.4 Hz, 1H), 8.19 (s, 1H), 7.79 (d, J=7.9 Hz, 1H), 7.73~7.68 (m, 2H), 7.50~7.45 (m, 3H), 7.44~7.41 (m, 2H); 13C NMR (CDCl3, 75 MHz) δ: 139.8, 135.0, 134.6, 133.2, 129.6, 128.3, 127.0, 126.2, 125.0, 124.9, 123.4, 119.5, 115.9, 115.5, 111.1; HRMS (ESI) calcd for C15H11N2S [M+H]+ 251.0638, found 251.0641.
3-(2-吡啶基)-吡唑并[1,5-a]喹啉(29): 深褐色固体, 产率62%. m.p. 126.1~127 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.70~8.68 (m, 1H), 8.63 (d, J=8.3 Hz, 1H), 8.48 (d, J=9.4 Hz, 1H), 8.45 (s, 1H), 7.81 (d, J=7.8 Hz, 1H), 7.74~7.67 (m, 3H), 7.58 (d, J=9.4 Hz, 1H), 7.47~7.52 (m, 1H), 7.12~7.17 (m, 1H); 13C NMR (CDCl3, 75 MHz) δ: 153.1, 149.7, 140.0, 136.5, 136.1, 134.7, 129.5, 128.3, 126.0, 125.1, 123.7, 120.6, 120.3, 118.1, 115.7, 114.8; HRMS (ESI) calcd for C16H12N3 [M+H]+ 246.1026, found 246.1034.
3-苯基-5-甲基-吡唑并[1,5-a]喹啉(30): 黄色固体, 产率93%. m.p. 94.9~95.7 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.66~8.63 (m, 1H), 8.17 (s, 1H), 7.88 (d, J=7.8 Hz, 1H), 7.73~7.67 (m, 1H), 7.66~7.63 (m, 2H), 7.54~7.47 (m, 4H), 7.37~7.32 (m, 1H), 2.62 (d, J=0.7 Hz, 3H); 13C NMR (CDCl3, 75 MHz) δ: 139.9, 134.7, 134.5, 133.3, 132.1, 129.3, 129.0, 127.4, 126.4, 125.1, 124.7, 123.8, 115.7, 115.1, 114.9, 19.4; HRMS (ESI) calcd for C18H15N2 [M+H]+ 259.1230, found 259.1231.
3-苯基-7-溴-吡唑并[1,5-a]喹啉(31): 深褐色固体, 产率20%. m.p. 84.7~85.5 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.49 (d, J=8.9 Hz, 1H), 8.21 (s, 1H), 7.92 (s, 1H), 7.79~7.75 (m, 1H), 7.73 (d, J=9.5 Hz, 1H), 7.62 (d, J=7.8 Hz, 2H), 7.52~7.45 (m, 2H), 7.38 (d, J=9.6 Hz, 2H); 13C NMR (CDCl3, 75 MHz) δ: 140.2, 134.5, 132.7, 132.5, 130.5, 129.1, 128.7, 127.5, 126.8, 124.9, 123.9, 118.1, 117.3, 117.3, 116.4; HRMS (ESI) calcd for C17H12- BrN2 [M+H]+ 323.0179, found 323.0179.
3-苯基-7-氯-吡唑并[1,5-a]喹啉(32): 黄色固体, 产率49%. m.p. 147.0~147.9 ℃; 1H NMR (CDCl3, 300 MHz) δ: 8.53 (d, J=8.9 Hz, 1H), 8.20 (s, 1H), 7.71 (d, J=9.8 Hz, 2H), 7.62 (d, J=8.3 Hz, 3H), 7.52~7.47 (m, 2H), 7.39~7.33 (m, 2H); 13C NMR (CDCl3, 75 MHz) δ: 140.2, 134.5, 133.5, 132.7, 130.4, 129.7, 129.2, 127.5, 127.3, 126.8, 124.5, 123.9, 117.3, 117.1, 116.4; HRMS (ESI) calcd for C17H12ClN2 [M+H]+ 279.0684, found 279.0684.