

Chinese Journal of Organic Chemistry >
Recent Progress on Radical-Mediated Activation of Nitroarenes
Received date: 2025-02-17
Revised date: 2025-03-17
Online published: 2025-04-17
Copyright
Nitroarene is one of the cheapest and most readily available raw materials in the chemical industry and is often used as multifunctional building blocks. In the past, the synthesis of nitrogen-containing compounds mainly depended on aniline. However, the utilization of nitroarenes can save one or more steps as compared to anilines. Therefore, the construction of nitrogen-containing compounds using nitroarenes as potential amino sources has become one of the research hotspots in recent years. Due to the advantages of radicals, such as high activity, mild reaction conditions, and good functional group compatibility, good progress has been made on radical-mediated activation of nitroarenes in recent years, and a variety of reaction types have been implemented. In this review, the recent progress in radical-mediated activation of nitroarenes under different catalytic conditions is summarized. The reaction substrate compatibility, mechanism, applications, advantages, and limitations in this field are also discussed in detail.
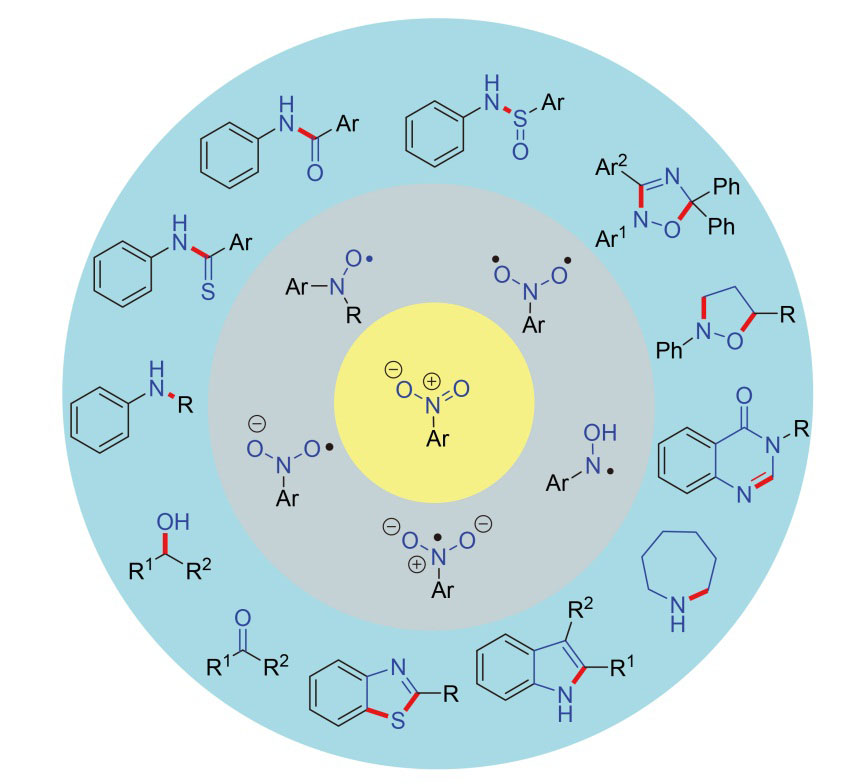
Key words: nitroarenes; radical; activation reaction; nitrogen-containing compounds
Binbin Zhang , Lu'na Liu , Yangbin Zhu , Dong Zou . Recent Progress on Radical-Mediated Activation of Nitroarenes[J]. Chinese Journal of Organic Chemistry, 2025 , 45(8) : 2698 -2725 . DOI: 10.6023/cjoc202502017
| [1] |
(a)
(b)
|
| [2] |
(a)
(b)
(c)
(d)
(e)
|
| [3] |
(a)
(b)
(c)
|
| [4] |
|
| [5] |
|
| [6] |
(a)
(b)
|
| [7] |
(a)
(b)
|
| [8] |
(a)
(b)
(c)
|
| [9] |
(a)
(b)
(c)
(d)
|
| [10] |
|
| [11] |
(a)
(b)
(c)
|
| [12] |
|
| [13] |
(a)
(b)
(c)
|
| [14] |
(a)
(b)
(c)
|
| [15] |
|
| [16] |
(a)
(b)
(c)
|
| [17] |
(a)
(b)
|
| [18] |
|
| [19] |
(a)
(b)
|
| [20] |
(a)
(b)
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
(a)
(b)
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
(a)
(b)
|
| [42] |
|
| [43] |
(a)
(b)
|
| [44] |
|
| [45] |
|
| [46] |
(a)
(b)
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
(a)
(b)
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
(a)
(b)
(c)
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
(a)
(b)
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
(a)
(b)
|
| [82] |
(a)
(b)
|
| [83] |
(a)
(b)
|
/
| 〈 |
|
〉 |