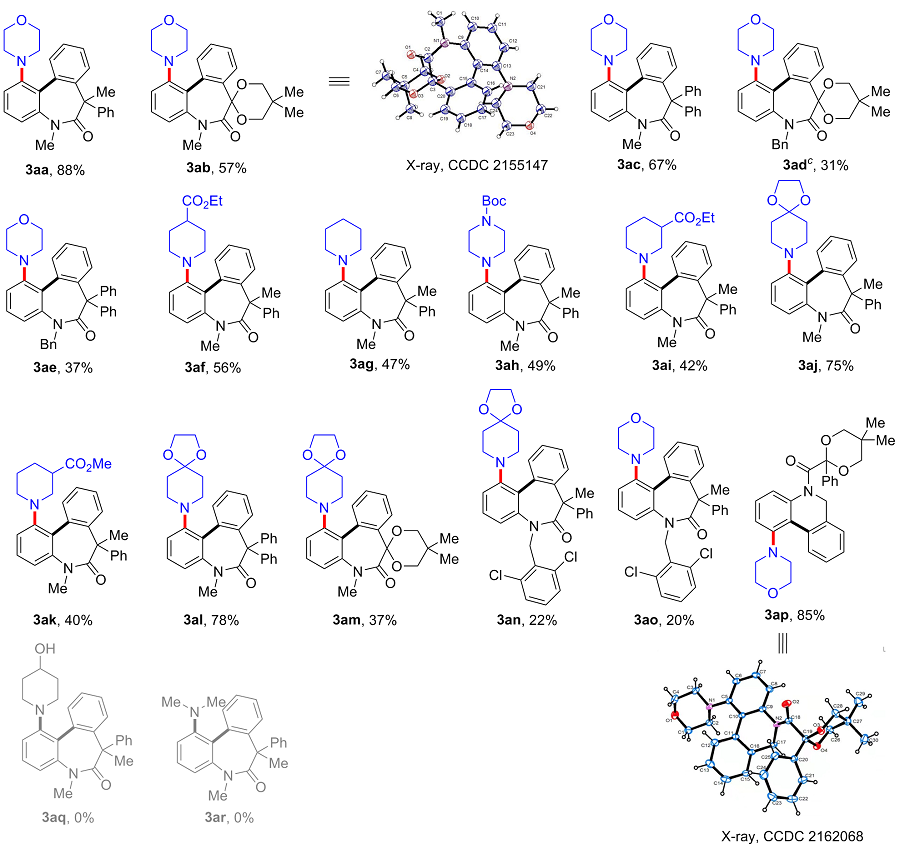
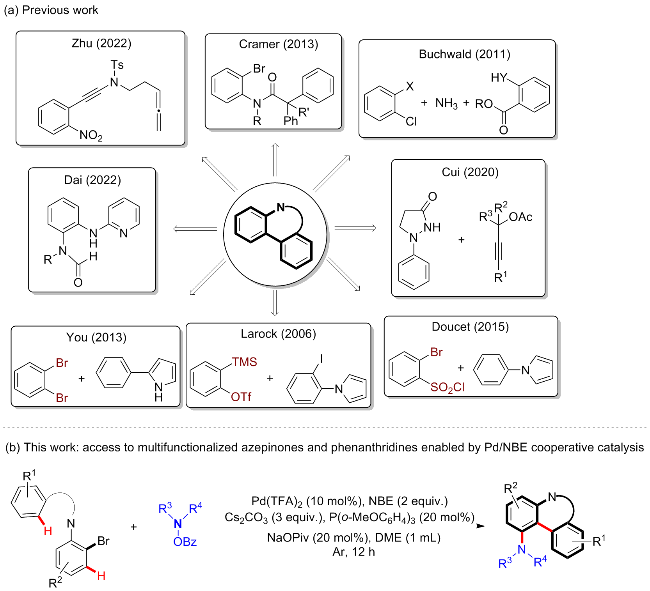
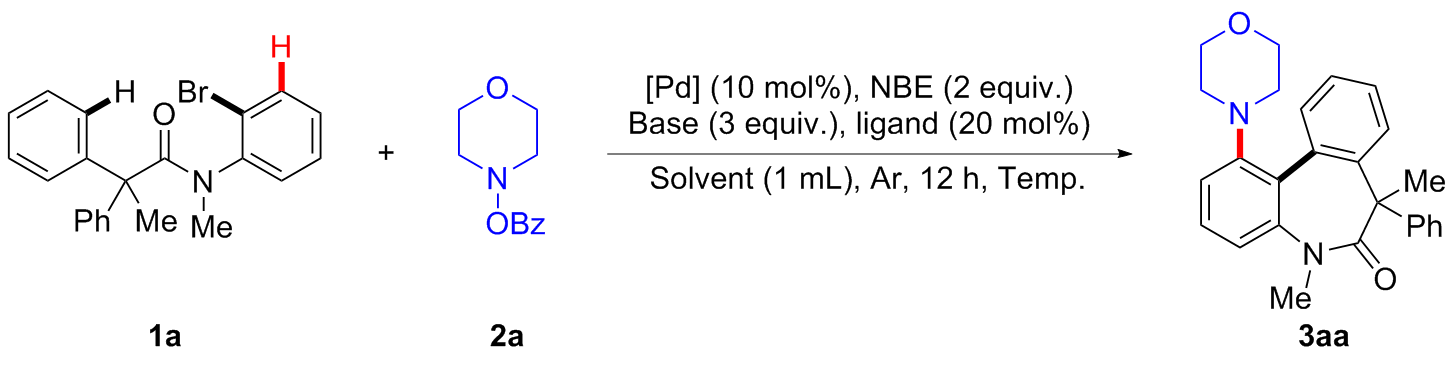
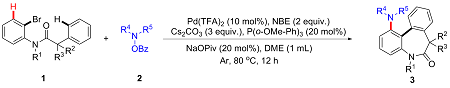5,7-二甲基-1-吗啉基-7-苯基-5,7-二氢-6H-二苯并[b,d]氮杂䓬-6-酮(3aa): 白色固体, 分离收率88%. m.p. 192~193 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.17~8.15 (m, 1H), 7.56 (d, J=8.0 Hz, 1H), 7.47~7.43 (m, 1H), 7.37~7.33 (m, 1H), 7.03 (t, J=8.1 Hz, 1H), 6.88~6.83 (m, 4H), 6.75~6.72 (m, 1H), 6.60 (s, 1H), 6.46 (d, J=8.0 Hz, 1H), 3.56 (t, J=4.6 Hz, 4H), 3.37 (s, 3H), 2.69~2.64 (m, 2H), 2.46~2.43 (m, 2H), 1.19 (s, 3H); 13C NMR (100 MHz, Chloroform-d) δ: 174.0, 149.1, 143.4, 143.0, 142.5, 133.8, 130.9, 128.6, 128.1, 127.5, 126.9, 126.2, 126.1, 125.9, 116.4, 114.7, 66.6, 56.0, 50.8, 38.1, 29.0. HRMS (ESI) calcd for C26H27N2O2 [M+H]+ 399.2067, found 399.2071.
5,5',5'-三甲基-1-吗啉基螺[二苯并[
b,
d]氮杂䓬-7,2'-[
1,
3]二噁烷]-6(5
H)-酮(
3ab): 白色固体, 分离收率57%. m.p. 172~173 ℃;
1H NMR (400 MHz, Chloroform-
d)
δ: 8.04~8.02 (m, 1H), 7.83~7.81 (m, 1H), 7.40~7.33 (m, 2H), 7.29~7.25 (m, 1H), 6.99~6.96 (m, 1H), 6.88~6.86 (m, 1H), 4.30 (d,
J=11.4 Hz, 1H), 3.74 (dd,
J=2.5, 11.3 Hz, 1H), 3.56 (s, 3H), 3.36 (s, 3H), 3.09 (d,
J=11.2 Hz, 1H), 2.99 (dd,
J=2.6 Hz, 11.2 Hz, 1H), 2.86~2.67 (m, 4H), 1.59~1.57 (m, 1H), 1.06 (s, 3H), 0.71 (s, 3H);
13C NMR (100 MHz, Chloroform-
d)
δ: 168.5, 149.9, 141.7, 138.4, 131.6, 130.9, 128.5, 128.1, 127.9, 127.4, 124.1, 115.4, 115.2, 97.9, 73.6, 73.3, 66.8, 37.7, 29.9, 22.8, 21.9. HRMS (ESI) calcd for C
24H
29N
2O
4 [M+H]
+ 409.2122, found 409.2120.
5-甲基-1-吗啉基-7,7-二苯基-5,7-二氢-6H-二苯并[b,d]氮杂䓬-6-酮(3ac): 白色固体, 分离收率67%. m.p. 198~199 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.23 (dd, J=1.3, 7.7 Hz, 1H), 7.37~7.27 (m, 3H), 7.26~7.23 (m, 2H), 7.03~6.96 (m, 4H), 6.90~6.86 (m, 2H), 6.71~6.65 (m, 2H), 6.51~6.49 (m, 1H), 3.58~3.56 (m, 4H), 3.40 (s, 3H), 2.68~2.63 (m, 2H), 2.55~2.50 (m, 2H); 13C NMR (100 MHz, Chloroform-d) δ: 173.5, 148.9, 142.7, 141.5, 139.2, 134.0, 131.4, 129.9, 128.6, 128.5, 128.3, 128.0, 127.5, 127.3, 127.2, 126.8, 126.7, 126.4, 126.0, 116.4, 114.9, 67.2, 66.7, 50.9, 39.0. HRMS (ESI) calcd for C31H29N2O2 [M+H]+: 461.2224, found 461.2221.
5-苄基-5',5'-二甲基-1-吗啉基螺[二苯并[
b,
d]氮杂䓬-7,2'-[
1,
3]二噁烷]-6(5
H)-酮(
3ad): 白色固体, 分离收率31%. m.p. 145~146 ℃;
1H NMR (400 MHz, Chloroform-
d)
δ: 7.83~7.80 (m, 2H), 7.40~7.36 (m, 1H), 7.34~7.30 (m, 1H), 7.18 (t,
J=8.0 Hz, 1H), 7.09~7.00 (m, 4H), 6.82~6.80 (m, 1H), 6.74~6.72 (m, 2H), 5.36 (d,
J=15.1 Hz, 1H), 4.78 (d,
J=15.1 Hz, 1H), 4.31 (d,
J=11.4 Hz, 1H), 3.76 (dd,
J=2.6, 11.4 Hz, 1H), 3.52~3.51 (m, 3H), 3.23 (d,
J=11.2 Hz, 1H), 3.03 (dd,
J=2.5, 11.2 Hz, 1H), 2.82 (s, 2H), 2.62 (s, 2H), 1.08 (s, 3H), 0.74 (s, 3H);
13C NMR (100 MHz, Chloroform-
d)
δ: 168.3, 149.8, 140.3, 137.9, 137.1, 132.0, 130.8, 129.8, 128.1, 127.8, 127.3, 127.1, 126.8, 124.1, 116.2, 115.7, 98.5, 73.8, 73.4, 66.8, 53.4, 29.8, 22.8, 22.0. HRMS (ESI) calcd for C
30H
33- N
2O
4 [M+H]
+ 485.2435, found 485.2429.
5-苄基-1-吗啉基-7,7-二苯基-5,7-二氢-6H-二苯并[b,d]氮杂䓬-6-酮(3ae): 白色固体, 分离收率37%. m.p. 165~166 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.14 (dd, J=1.2 Hz, 7.6 Hz, 1H), 7.44~7.42 (m, 1H), 7.39~7.30 (m, 3H), 7.27~7.24 (m, 3H), 7.15~7.07 (m, 5H), 7.03~6.93 (m, 3H), 6.91~6.84 (m, 3H), 6.77~6.70 (m, 2H), 6.48 (d, J=7.7 Hz, 1H), 5.47 (d, J=15.2 Hz), 4.85 (d, J=15.2 Hz, 1H), 3.62 (t, J=3.8 Hz, 4H), 2.78~2.74 (m, 2H), 2.59~2.56 (m, 2H); 13C NMR (100 MHz, Chloroform-d) δ: 172.3, 148.5, 141.9, 141.5, 140.4, 137.8, 134.3, 131.2, 130.0, 129.1, 128.6, 128.4, 128.1, 128.0, 127.8, 127.5, 127.4, 127.2, 126.6, 126.6, 126.5, 126.1, 118.0, 115.5, 67.1, 66.7, 54.4, 50.8. HRMS (ESI) calcd for C37H33N2O2 [M+H]+ 537.2537, found 537.2540.
1-(5,7-二甲基-6-氧代-7-苯基-6,7-二氢-5H-二苯并[b,d]氮杂䓬-1-基)哌啶-4-羧酸乙酯(3af): 白色固体, 分离收率56%. m.p. 168~169 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.10 (dd, J=1.5 Hz, 7.8 Hz, 1H), 7.76~7.74 (m, 1H), 7.46~7.42 (m, 1H), 7.37~7.33 (m, 1H), 7.00 (t, J=8.1 Hz, 1H), 6.86~6.82 (m, 4H), 6.72~6.70 (m, 1H), 6.23~6.22 (m, 1H), 6.47~6.45 (m, 1H), 4.13 (q, J=7.1 Hz, 2H), 3.36 (s, 3H), 3.04~3.02 (m, 1H), 2.70~2.59 (m, 2H), 2.25~2.18 (m, 1H), 1.91 (s, 3H), 1.77~1.67 (m, 1H), 1.59~1.55 (m, 1H), 1.46~1.36 (m, 1H), 1.25 (t, J=7.0 Hz, 3H); 13C NMR (100 MHz, Chloroform-d) δ: 175.0, 174.0, 149.9, 143.4, 142.8, 142.2, 133.9, 130.7, 128.4, 128.0, 127.4, 127.1, 126.3, 126.0, 125.7, 116.1, 115.2, 60.3, 56.0, 50.9, 49.8, 40.8, 38.1, 29.1, 28.2, 27.8, 14.2. HRMS (ESI) calcd for C30H33N2O3 [M+H]+ 469.2486, found 469.2483.
5,7-二甲基-7-苯基-1-哌啶基-5,7-二氢-6H-二苯并[b,d]氮杂䓬-6-酮(3ag): 白色固体, 分离收率47%. m.p. 139~140 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.16 (dd, J=1.5 Hz, 7.8 Hz, 1H), 7.75~7.73 (m, 1H), 7.44~7.41 (m, 1H), 7.35~7.32 (m, 1H), 6.99 (t, J=8.1 Hz, 1H), 6.88~6.83 (m, 4H), 6.68~6.64 (m, 2H), 6.47~6.46 (m, 1H), 3.36 (s, 3H), 2.61~2.57 (m, 2H), 2.42 (s, 2H), 1.91 (s, 3H), 1.40 (s, 6H); 13C NMR (125 MHz, Chloroform-d) δ: 174.1, 150.7, 143.4, 142.7, 142.1, 134.2, 130.9, 128.4, 127.8, 127.4, 127.0, 126.2, 125.9, 125.7, 125.5, 115.7, 115.3, 56.0, 51.9, 38.2, 38.5, 29.1, 25.7, 24.0. HRMS (ESI) calcd for C27H29N2O [M+H]+ 397.2274, found 397.2276.
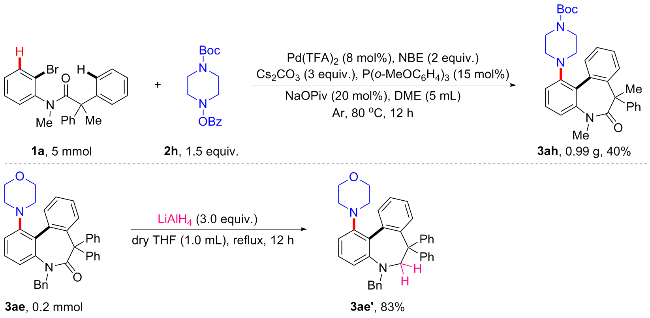
4-(5,7-二甲基-6-氧代-7-苯基-6,7-二氢-5H-二苯并[b,d]氮杂䓬-1-基)哌嗪-1-羧酸叔丁酯(3ah): 白色固体, 分离收率49%. m.p. 132~133 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.12 (dd, J=1.4 Hz, 7.8 Hz, 1H), 7.78~7.76 (m, 1H), 7.47~7.42 (m, 2H), 7.35~7.31 (m, 2H), 7.02 (t, J=8.1 Hz, 1H), 6.87~6.82 (m, 4H), 6.75~6.73 (m, 1H), 6.62 (s, 1H), 6.45~6.43 (m, 1H), 3.37 (s, 3H), 3.27 (s, 3H), 2.65 (m, 3H), 2.42 (s, 2H), 1.92 (s, 3H), 1.45 (s, 9H); 13C NMR (100 MHz, Chloroform-d) δ: 174.0, 154.8, 149.1, 143.3, 143.3, 142.5, 133.7, 130.9, 128.5, 128.1, 127.5, 127.1, 126.2, 125.8, 116.6, 115.1, 79.7, 56.0, 50.3, 38.1, 29.4, 29.1, 28.4. HRMS (ESI) calcd for C31H36- N3O3 [M+H]+ 498.2751, found 498.2758.
1-(5,7-二甲基-6-氧代-7-苯基-6,7-二氢-5H-二苯并[b,d]氮杂䓬-1-基)哌啶-3-羧酸乙酯(3ai): 白色固体, 分离收率42%. m.p. 138~139 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.05~8.01 (m, 1H), 7.77~7.74 (m, 1H), 7.46~7.41 (m, 1H), 7.34~7.30 (m, 1H), 7.04~6.99 (m, 1H), 6.87~6.83 (m, 4H), 6.74~6.71 (m, 1H), 6.64~6.63 (m, 1H), 6.53~6.46 (m, 1H), 4.15~4.06 (m, 2H), 3.38~3.36 (m, 3H), 3.26~3.23 (m, 1H), 2.72~2.51 (m, 3H), 2.05~1.94 (m, 1H), 1.91 (s, 3H), 1.87~1.84 (m, 1H), 1.40~1.34 (m, 2H), 1.26~1.22 (m, 4H); 13C NMR (100 MHz, Chloroform-d) δ: 174.0, 173.5, 149.9, 143.4, 142.7, 142.2, 133.8, 131.1, 128.4, 127.9, 127.6, 127.4, 126.1, 126.0, 125.8, 116.4, 116.0, 60.4, 55.9, 52.6, 51.9, 42.3, 38.1, 29.1, 26.8, 24.0, 14.2. HRMS (ESI) calcd for C30H33- N2O3 [M+H]+ 469.2486, found 469.2482.
5,7-二甲基-7-苯基-1-(1,4-二氧杂-8-氮杂螺[4.5]癸烷-8-基)-5,7-二氢-6H-二苯并[b,d]氮杂䓬-6-酮(3aj): 白色固体, 分离收率75%. m.p. 175~176 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.14 (dd, J=1.3, 7.8 Hz, 1H), 7.75 (d, J=7.6 Hz, 1H), 7.45~7.40 (m, 1H), 7.35~7.31 (m, 1H), 6.99 (t, J=8.1 Hz, 1H), 6.85~6.82 (m, 4H), 6.71~6.68 (m, 1H), 6.61 (s, 1H), 6.47 (d, J=8.0 Hz, 1H), 3.93 (s, 4H), 3.36 (s, 3H), 2.76~2.74 (m, 2H), 2.59 (s, 2H), 1.91 (s, 3H), 1.64~1.58 (m, 4H); 13C NMR (100 MHz, Chloroform-d) δ: 174.0, 149.5, 143.3, 142.8, 142.2, 134.0, 130.5, 128.4, 127.9, 127.5, 127.5, 126.8, 126.3, 126.1, 125.8, 116.0, 115.4, 106.9, 64.2, 55.9, 48.5, 38.1, 34.7, 29.1. HRMS (ESI) calcd for C29H31N2O3 [M+H]+ 455.2329, found 455.2333.
1-(5,7-二甲基-6-氧代-7-苯基-6,7-二氢-5H-二苯并[b,d]氮杂䓬-1-基)哌啶-3-羧酸甲基酯(3ak): 白色固体, 分离收率40%. m.p. 124~125 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 7.99 (d, J=7.2 Hz, 1H), 7.75 (d, J=7.7 Hz, 1H), 7.46~7.42 (m, 1H), 7.34~7.30 (m, 1H), 7.01 (t, J=8.1 Hz, 1H), 6.87~6.83 (m, 4H), 6.74~6.72 (m, 1H), 6.64~6.63 (m, 1H), 6.53 (d, J=7.8 Hz, 1H), 3.64 (s, 3H), 3.36 (s, 3H), 3.24~3.19 (m, 1H), 2.71 (t, J=10.6 Hz, 1H), 2.63~2.57 (m, 2H), 1.97~1.94 (m, 1H), 1.91 (s, 3H), 1.42~1.33 (m, 2H), 1.30~1.24 (m, 2H); 13C NMR (100 MHz, Chloroform-d) δ: 174.4, 174.0, 149.9, 143.5, 142.8, 142.3, 133.7, 131.1, 128.4, 127.9, 127.5, 126.0, 126.0, 125.8, 116.6, 116.2, 56.0, 52.7, 52.0, 51.7, 42.1, 38.1, 29.1, 26.8, 24.0. HRMS (ESI) calcd for C29H31N2O3 [M+H]+ 455.2329, found 455.2325.
甲基-7,7-二苯基-1-(1,4-二氧杂-8-氮杂螺[4.5]癸烷- 8-基)-5,7-二氢-6H-二苯并[b,d]氮杂䓬-6-酮(3al): 白色固体, 分离收率78%. m.p. 210~211 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.21 (dd, J=1.3 Hz, 7.8 Hz, 1H), 7.35~7.21 (m, 7H), 6.99~6.87 (m, 6H), 6.72~6.70 (m, 1H), 6.64~6.62 (m, 1H), 6.53~6.51 (m, 1H), 3.94 (s, 4H), 3.39 (s, 3H), 2.77~2.65 (m, 4H), 1.63~1.59 (m, 4H); 13C NMR (100 MHz, Chloroform-d) δ: 173.5, 149.4, 142.5, 142.4, 141.6, 139.2, 134.2, 131.0, 129.8, 128.4, 128.4, 128.2, 127.9, 127.4, 127.3, 127.0, 126.6, 126.3, 126.1, 116.0, 115.7, 106.9, 67.2, 64.2, 48.6, 39.0, 34.7. HRMS (ESI) calcd for C34H33N2O3 [M+H]+ 517.2486, found 517.2484.
5,5',5'-三甲基-1-(1,4-二氧杂-8-氮杂螺[4.5]癸-8-基)螺[二苯并[
b,
d]氮杂䓬-7,2'-[
1,
3]二氧杂环己烷]-6(5
H)-酮(
3am): 白色固体, 分离收率37%. m.p. 135~136 ℃;
1H NMR (400 MHz, Chloroform-
d)
δ: 8.03~8.00 (m, 1H), 7.82~7.80 (m, 1H), 7.38~7.30 (m, 2H), 7.23 (t,
J=8.1 Hz, 1H), 6.91 (dd,
J=7.6, 19.8 Hz, 2H), 4.29 (d,
J=11.3 Hz, 1H), 3.91 (s, 4H), 3.74 (dd,
J=2.3, 11.2 Hz, 1H), 3.35 (s, 3H), 3.09~3.06 (m, 1H), 2.99~2.95 (m, 2H), 1.32~1.25 (m, 3H), 1.04 (s, 3H), 0.70 (s, 3H);
13C NMR (100 MHz, Chloroform-
d)
δ: 168.5, 150.5, 141.5, 138.1, 132.0, 130.6, 128.6, 127.9, 127.7, 127.5, 124.0, 115.8, 115.0, 107.1, 97.9, 73.5, 73.3, 64.2, 37.7, 34.9, 29.9, 22.8, 21.9. HRMS (ESI) calcd for C
27H
33N
2O
5 [M+H]
+ 465.2384, found 465.2383.
5-(2,6-二氯苄基)-7-甲基-7-苯基-1-(1,4-二氧杂-8-氮杂螺[4.5]癸-8-基)-5,7-二氢-6H-二苯并[b,d]氮杂䓬-6-酮(3an): 白色固体, 分离收率22%. m.p. 185~186 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 7.90 (dd, J=1.4 Hz, 7.8 Hz, 1H), 7.70 (d, J=7.3 Hz, 1H), 7.41~7.37 (m, 1H), 7.32~7.28 (m, 2H), 7.24~7.22 (m, 1H), 6.99~6.97 (m, 2H), 6.93~6.89 (m, 3H), 6.87~6.81 (m, 3H), 6.59~6.58 (m, 1H), 6.35~6.33 (m, 1H), 5.86 (d, J=14.7 Hz, 1H), 5.07 (d, J=14.7 Hz, 1H), 3.91 (s, 4H), 2.71~2.66 (m, 2H), 2.48 (s, 2H), 1.93 (s, 3H), 1.55~1.54 (m, 3H), 1.25 (s, 3H); 13C NMR (100 MHz, Chloroform-d) δ: 174.0, 149.2, 143.6, 141.6, 139.4, 136.4, 134.4, 132.4, 130.6, 128.8, 128.4, 128.2, 128.1, 128.0, 127.7, 127.6, 126.9, 126.2, 126.1, 125.6, 117.6, 116.0, 107.0, 64.2, 56.4, 48.5, 46.9, 34.7, 29.6, 29.4. HRMS (ESI) calcd for C32H29Cl2- N2O2 [M+H]+ 599.1863, found 599.1857.
5-(2,6-二氯苄基)-7-甲基-1-吗啉基-7-苯基-5,7-二氢-6H-二苯并[b,d]氮杂䓬-6-酮(3ao): 白色固体, 分离收率20%. m.p. 191~192 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 7.92 (dd, J=1.4 Hz, 7.8 Hz, 1H), 7.71 (d, J=7.5 Hz, 1H), 7.44~7.40 (m, 1H), 7.34~7.29 (m, 1H), 7.00~6.78 (m, 9H), 6.58~6.57 (m, 1H), 6.34~6.31 (m, 1H), 5.87 (d, J=14.7 Hz, 1H), 5.08 (d, J=14.7 Hz, 1H), 3.53~3.51 (m, 4H), 2.63~2.58 (m, 2H), 2.38~2.35 (m, 2H), 1.93 (s, 3H), 1.58 (s, 2H); 13C NMR (100 MHz, Chloroform-d) δ: 174.0, 148.6, 143.6, 142.0, 139.7, 136.4, 134.2, 132.3, 130.9, 128.8, 128.2, 128.0, 127.9, 127.8, 127.5, 126.2, 126.1, 125.7, 118.0, 115.2, 66.7, 56.4, 50.7, 46.9, 29.3. HRMS (ESI) calcd for C35H33Cl2N2O3 [M+H]+ 543.1601, found 543.1607.
(5,5-二甲基-2-苯基-1,3-二氧杂环己烷-2-基)(1-吗啉基菲啶-5(6H)-基)甲酮(3ap): 黄色油状, 分离收率85%. 1H NMR (400 MHz, Chloroform-d) δ: 8.81 (d, J=8.0 Hz, 1H), 7.62~7.56 (m, 3H), 7.35~7.34 (m, 3H), 7.25~7.21 (m, 1H), 7.04 (t, J=7.3 Hz, 1H), 6.99~6.96 (m, 1H), 6.66 (s, 1H), 4.43 (s, 2H), 3.96 (s, 4H), 3.76 (s, 2H), 3.53~3.51 (m, 2H), 3.14 (s, 2H), 2.82 (s, 2H), 1.94 (s, 2H), 1.68 (s, 2H), 1.28 (s, 2H), 0.66 (s, 3H); 13C NMR (100 MHz, Chloroform-d) δ: 167.0, 150.7, 140.0, 139.2, 131.9, 129.1, 128.6, 127.6, 127.1, 126.8, 125.8, 125.2, 124.9, 119.8, 116.5, 107.0, 99.3, 73.7, 64.3, 48.8, 35.0, 29.6, 22.9, 21.8. HRMS (ESI) calcd for C30H33N2O4 [M+H]+ 485.2435, found 485.2431.
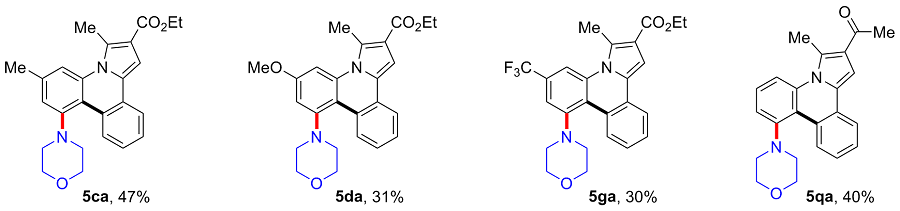
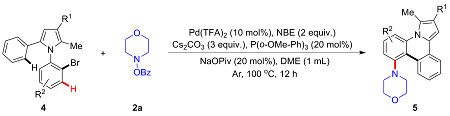
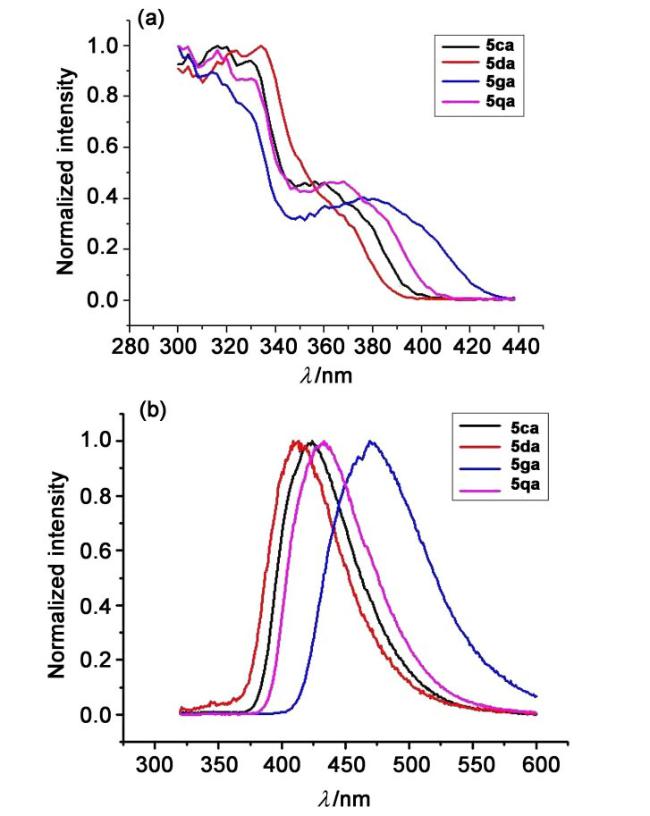
3,6-二甲基-8-吗啉基吡咯并[1,2-f]菲啶-2-羧酸乙酯(5ca): 白色固体, 分离收率47%. m.p. 186~187 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.81~9.79 (m, 1H), 7.99~7.97 (m, 1H), 7.68 (s, 1H), 7.43~7.32 (m, 3H), 6.93 (s, 1H), 4.40 (q, J=7.1 Hz, 2H), 4.02~3.94 (m, 4H), 3.26~3.22 (m, 2H), 3.15 (s, 3H), 3.02~2.93 (m, 2H), 2.49 (s, 3H), 1.45 (t, J=7.2, 3H); 13C NMR (100 MHz, CDCl3) δ: 165.7, 151.6, 137.3, 136.3, 132.9, 129.4, 127.4, 125.9, 125.5, 125.5, 122.4, 116.5, 115.7, 114.7, 114.3, 102.3, 66.8, 59.8, 52.6, 21.9, 16.7, 14.5. HRMS (ESI) calcd for C25H27N2O3 [M+H]+ 403.2016, found 403.2019.
6-甲氧基-3-甲基-8-吗啉基吡咯并[1,2-f]菲啶-2-羧酸乙酯(5da): 白色固体, 分离收率31%. m.p. 190~191 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.69~9.67 (m, 1H), 7.98~7.95 (m, 1H), 7.41~7.31 (m, 4H), 6.69 (d, J=2.5 Hz, 1H), 4.40 (q, J=7.1 Hz), 4.01~3.93 (m, 7H), 3.26~3.23 (m, 2H), 3.17 (s, 3H), 2.99~2.91 (m, 2H),1.45 (t, J=7.1Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 165.7, 158.6, 153.0, 137.4, 132.5, 129.6, 126.9, 125.9, 125.6, 125.0, 124.8, 122.4, 116.7, 110.6, 102.2, 102.2, 98.6, 66.7, 59.9, 55.3, 52.5, 16.7, 14.5. HRMS (ESI) calcd for C25H27- N2O4 [M+H]+ 419.1965, found 419.1959.
3-甲基-8-吗啉基-6-(三氟甲基)吡咯并[1,2-f]菲啶-2-羧酸乙酯(5ga): 白色固体, 分离收率30%. m.p. 175~177 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.80~9.78 (m, 1H), 8.10 (s, 1H), 8.01~7.99 (m, 1H), 7.52~7.48 (m, 1H), 7.41~7.36 (m, 2H), 7.30 (s, 1H), 4.40 (q, J=7.1 Hz, 2H), 4.01~3.94 (m, 4H), 3.32~3.29 (m, 2H), 3.15 (s, 3H), 3.06~2.98 (m, 2H), 1.45 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 165.5, 152.1, 136.2, 133.2, 129.2, 129.1, 128.7 (q, J=32.6 Hz), 126.4, 125.9, 125.8, 124.8, 123.9 (q, J=270.7 Hz), 122.8, 120.0, 117.3, 110.6 (q, J=3.5 Hz), 110.3 (q, J=4.3 Hz), 103.1, 66.6, 60.1, 52.4, 16.5, 14.5. HRMS (ESI) calcd for C25H24F3N2O3 [M+H]+ 457.1734, found 457.1732.
1-(3-甲基-8-吗啉基吡咯并[1,2-f]菲啶-2-基)乙酮(5qa): 白色固体, 分离收率40%. m.p. 169~170 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.88 (d, J=8.4 Hz, 1H), 8.00~7.98 (m, 1H), 7.86~7.83 (m, 1H), 7.48~7.44 (m, 1H), 7.42~7.36 (m, 2H), 7.26 (s, 1H), 7.14~7.12 (m, 1H), 4.02~3.95 (m, 4H), 3.28~3.24 (m, 2H), 3.13 (s, 3H), 3.02~2.95 (m, 2H), 2.63 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 196.2, 151.7, 136.1, 132.4, 129.2, 128.0, 127.3, 125.9, 125.8, 125.7, 124.4, 122.3, 117.4, 114.8, 114.1, 102.6, 66.8, 52.5, 29.4, 17.1. HRMS (ESI) calcd for C23H23N2O2 [M+H]+ 359.1754, found 359.1750.
4-(5-苄基-7,7-二苯基-6,7-二氢-5H-二苯并[b,d]氮杂䓬-1-基)吗啉(3ae'): 白色固体, 分离收率83%. m.p. 179~180 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 7.90 (d, J=7.8 Hz, 1H), 7.44~7.42 (m, 1H), 7.33~7.28 (m, 2H), 7.24~7.20 (m, 4H), 7.19~7.13 (m, 5H), 7.10~7.06 (m, 1H), 7.04~7.02 (m, 3H), 6.97~6.95 (m, 4H), 6.73 (d, J=7.9 Hz, 1H), 6.42 (d, J=8.0 Hz, 1H), 4.50 (d, J=14.5 Hz, 1H), 4.21~4.16 (m, 2H), 4.00 (d, J=13.0 Hz, 1H), 3.49~3.42 (m, 4H), 2.39~2.26 (m, 4H); 13C NMR (100 MHz, Chloroform-d) δ: 150.2, 149.8, 148.1, 144.3, 143.3, 139.4, 138.6, 131.3, 130.5, 129.6, 128.9, 128.3, 128.2, 127.7, 127.6, 127.4, 127.1, 126.7, 126.1, 126.0, 125.5, 125.4, 113.3, 112.4. 68.6, 66.7, 58.0, 57.9, 50.7. HRMS (ESI) calcd for C37H35N2O [M+H]+ 523.2744, found 523.2740.
辅助材料(Supporting Information) 所有产物
3aa~
3ap,
3ae',
5ca,
5da,
5ga,
5qa的
1H NMR和
13C NMR图谱. 这些材料可以免费从本刊网站(
http://sioc-journal.cn/)上下载.