

Chinese Journal of Organic Chemistry >
Studies on the Synthesis and Insecticidal Activities of Novel β-Naphthol Betti Bases and Their Cyclized Derivatives
Received date: 2024-11-14
Revised date: 2024-12-02
Online published: 2024-12-12
Supported by
National Natural Science Foundation of China(22077070); National Natural Science Foundation of China(21772103)
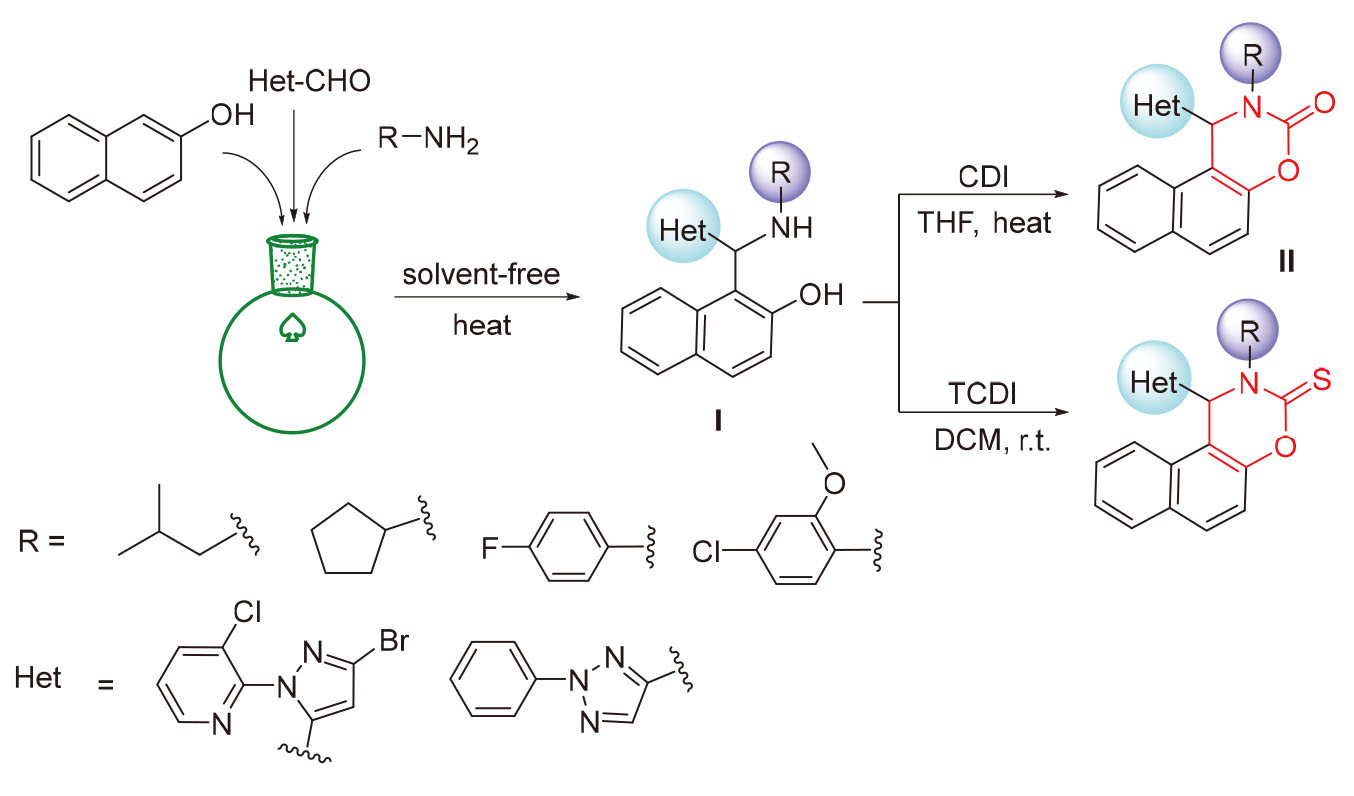
Based on the strategy of scaffold hopping molecular design, eight novel β-naphthol Betti bases Ia~Ih were synthesized through a one-pot Betti reaction of β-naphthol, heterocyclic aldehydes, and different types of amines. Fourteen novel dihydronaphthoxazine (thio) ketone derivatives IIa~IIn were further obtained via the cyclization of the synthesized Betti bases. The bioassay results showed that some of the compounds exhibited good insecticidal activity at 200 mg•L-1 with lethality rates of 80%~100% and 40%~100% against Plutella xylostella L. and Mythimna separata Walker, respectively. Overall, the Betti bases 1-((3-bromo-1-(3-chloropyridin-2-yl)-1H-pyrazol-5-yl)(isobutylamino)methyl)naphthalen-2-ol (Ia) and 1- (((4-fluorophenyl)amino)(2-phenyl-2H-1,2,3-triazol-4-yl)methyl)naphthalen-2-ol (If), and the cyclized derivatives 1-(3-bromo- 1-(3-chloropyridin-2-yl)-1H-pyrazol-5-yl)-2-isobutyl-1,2-dihydro-3H-naphtho[1,2-e][1,3]oxazin-3-one (IIa), 1-(3-bromo-1-(3- chloro-pyridin-2-yl)-1H-pyrazol-5-yl)-2-isobutyl-1,2-dihydro-3H-naphtho[1,2-e][1,3]oxazine-3-thione (IIc) and 1-(3-bromo- 1-(3-chloropyridin-2-yl)-1H-pyrazol-5-yl)-2-(4-fluorophenyl)-1,2-dihydro-3H-naphtho[1,2-e][1,3]oxazine-3-thione (IIk) possess favorable insecticidal potentials and deserve further investigation. The structure-activity relationship of the compounds was analyzed and discussed in detail. The results of this study provide useful reference and guidance for the molecular design of new pesticides based on phenolic derivatives.
Key words: naphthol derivatives; Betti base; scaffold hopping; synthesis; insecticidal activity
Haoqin Huang , Na Yang , Lixia Xiong , Baolei Wang . Studies on the Synthesis and Insecticidal Activities of Novel β-Naphthol Betti Bases and Their Cyclized Derivatives[J]. Chinese Journal of Organic Chemistry, 2025 , 45(4) : 1386 -1394 . DOI: 10.6023/cjoc202408040
| [1] | Youm, J.; Lee, H.; Choi, Y.; Yoon, J. J. Cell. Mol. Med. 2018, 22, 2680. |
| [2] | Rathod, A. S.; Reddy, P. V.; Biradar, J. S. Russ. J. Org. Chem. 2020, 56, 662. |
| [3] | Jenner, A. M.; Rafter, J.; Halliwell, B. Free Radical Biol. Med. 2005, 38, 763. |
| [4] | Das, B.; Reddy, C. R.; Kashanna, J.; Mamidyala, S. K.; Kumar, C. G. Med. Chem. Res. 2012, 21, 3321. |
| [5] | Hallock, Y. F.; Manfredi, K. P.; Blunt, J. W.; Cardellina II, J. H.; Sch?ffer, M.; Gulden, K.-P.; Bringmann, G.; Lee, A. Y.; Clardy, J.; Fran?ois, G.; Boyd, M. R. J. Org. Chem. 1994, 59, 6349. |
| [6] | Heusinkveld, H. J.; van Vliet, A. C.; Nijssen, P. C.; Westerink, R. H. Toxicol. Lett. 2016, 252, 62. |
| [7] | Khan, S. N.; Khan, S.; Misba, L.; Sharief, M.; Hashmi, A.; Khan, A. U. Biochem. Biophys. Res. Commun. 2019, 518, 459. |
| [8] | Ma, W.; Fan, Y.; Liu, Z.; Hao, Y.; Mou, Y.; Liu, Y.; Zhang, W.; Song, X. Vet. Parasitol. 2019, 266, 56. |
| [9] | Betti, M. Gazz. Chim. Ital. 1900, 30, 310. |
| [10] | Gangopadhyay, A.; Mahapatra, A. K. New J. Chem. 2019, 43, 11743. |
| [11] | Kciuk, M.; Malinowska, M.; Gielecińska, A.; Sundaraj, R.; Mujwar, S.; Zawisza, A.; Kontek, R. Molecules 2023, 28, 7230. |
| [12] | Nishtala, V. B.; Mahesh, C.; Bhargavi, G.; Pasala, V. K.; Basavoju, S. Mol. Diversity 2020, 24, 1139. |
| [13] | Shang, J.; Li, Y.; Yang, N.; Xiong, L.; Wang, B. Enzyme Inhib. Med. Chem. 2022, 37, 641. |
| [14] | Ashcroft, C. P.; Challenger, S.; Clifford, D.; Derrick, A. M.; Hajikarimian, Y.; Slucock, K.; Silk, T. V.; Thomson, N. M.; Williams, J. R. Org. Process Res. Dev. 2005, 9, 663. |
| [15] | Zhang, S.; Liu, H.; Yang, N.; Xiong, L.; Wang, B. Pest Manage. Sci. 2022, 78, 2086. |
| [16] | Wang, L.; Fan, W.; Cui, L.; Yang, N.; Zhang, X.; Yu, S.; Li, Y.; Wang, B. J. Agric. Food Chem. 2023, 71, 19343. |
| [17] | Zhang, Y.; Li, Y.; Li, H.; Shang, J.; Li, Z.; Wang, B. Chin. Chem. Lett. 2022, 33, 501. |
/
| 〈 |
|
〉 |