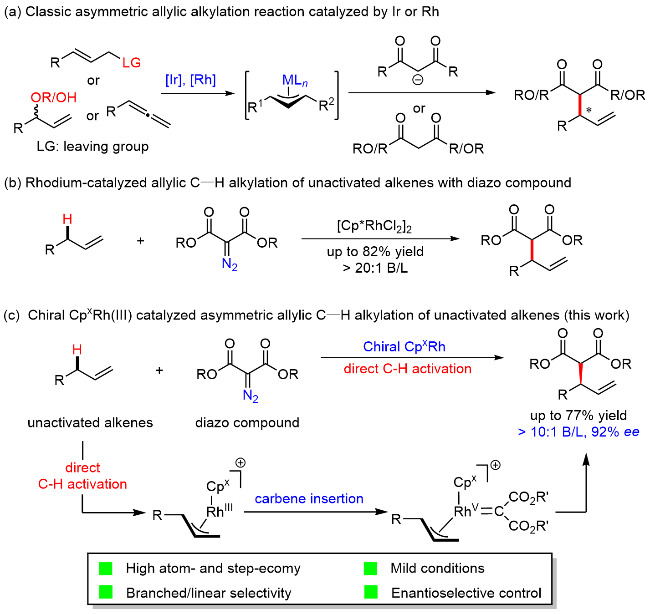
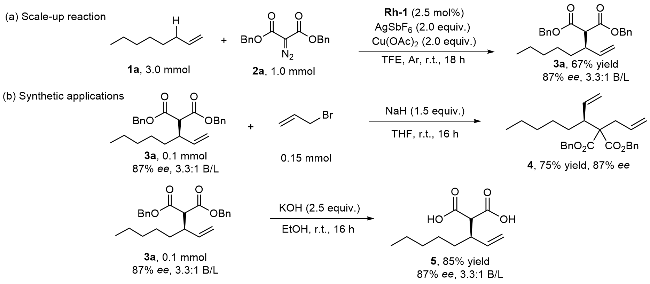
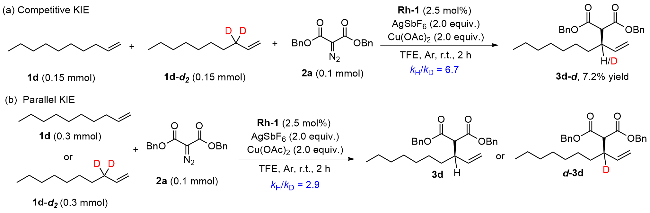
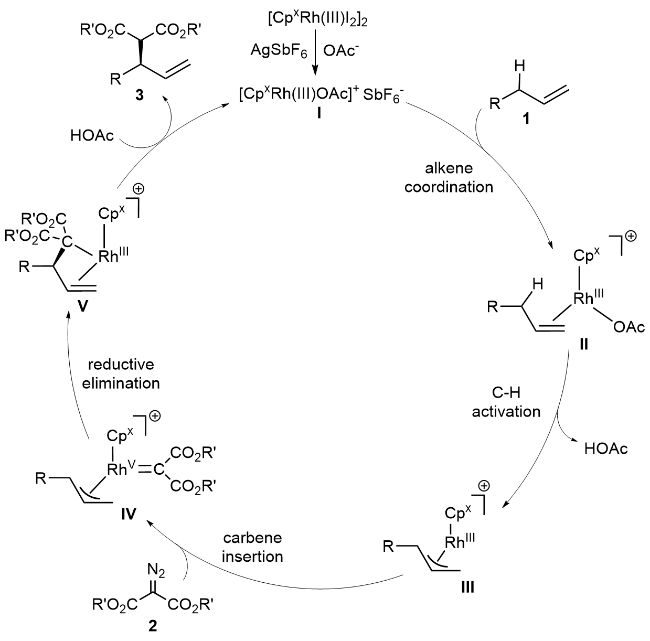
1 Introduction
2 Results and discussion
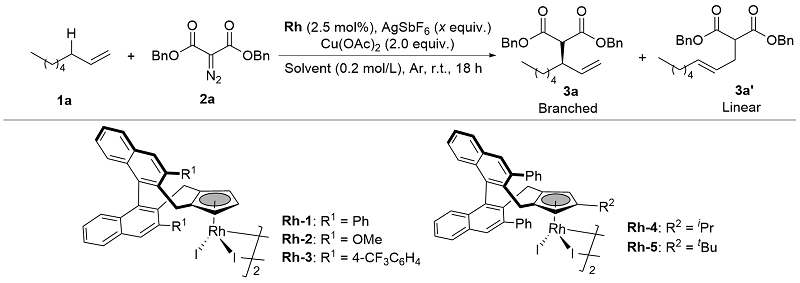
Table 1 Reaction optimization of asymmetric alkylationa
|
| Entry | Rh | Solvent | AgSbF6 (equiv.) | Yield/% | ee/% | B/L |
|---|---|---|---|---|---|---|
| 1 | Rh-1 | TFE | 0.1 | 42 | 56 | 1.5∶1 |
| 2 | Rh-2 | TFE | 0.1 | 25 | 15 | 1.1∶1 |
| 3 | Rh-3 | TFE | 0.1 | 20 | 35 | 1.3∶1 |
| 4 | Rh-4 | TFE | 0.1 | 35 | 49 | 2.4∶1 |
| 5 | Rh-5 | TFE | 0.1 | 32 | 51 | 2.1∶1 |
| 6 | Rh-1 | HFIP | 0.1 | 43 | 40 | 2.8∶1 |
| 7 | Rh-1 | DCE | 0.1 | 48 | 37 | 2.2∶1 |
| 8 | Rh-1 | MeOH | 0.1 | 48 | 54 | 1.2∶1 |
| 9 | Rh-1 | TFE | — | NR | — | — |
| 10 | Rh-1 | TFE | 1.0 | 60 | 70 | 3.2∶1 |
| 11 | Rh-1 | TFE | 2.0 | 71 | 87 | 3.3∶1 |
| 12 | Rh-1 | TFE | 2.5 | 68 | 87 | 3.2∶1 |
a Reaction conditions: 1a (0.3 mmol), 2a (0.1 mmol), Rh cat (2.5 mol%), AgSbF6 (x eqiuv.), Cu(OAc)2 (2.0 equiv.), solvent (0.2 mol/L), r.t., 18 h. Isolated yields (branched and linear). Determined by HPLC analysis on a chiral stationary phase. The radiometric ratio (branched/linear) was obtained by analysis of the 1H NMR spectra of the crude reaction mixtures. |
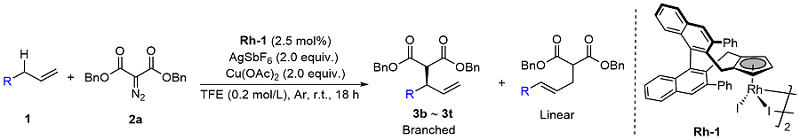
Table 2 Substrate scope of unactivated terminal alkenes
|
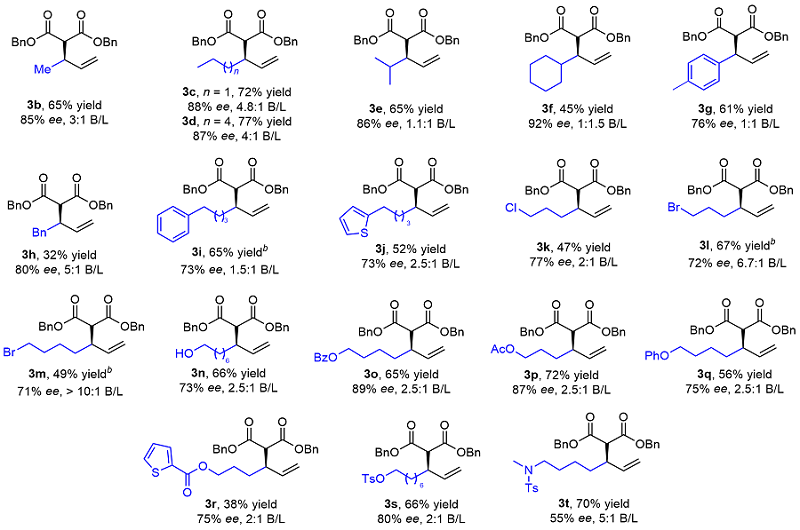 |
a Reaction conditions: 1 (0.3 mmol), 2a (0.1 mmol), Rh-1 (2.5 mol%), AgSbF6 (2.0 equiv.), Cu(OAc)2 (2.0 equiv.), TFE (0.2 mol/L), r.t., 18 h. Isolated yields (branched and linear). Enantiomeric excess was determined by HPLC analysis. Regiomeric ratio (branched/linear) was determined by analysis of the 1H NMR spectra of the crude reaction mixtures. b 40 ℃, 48 h. |
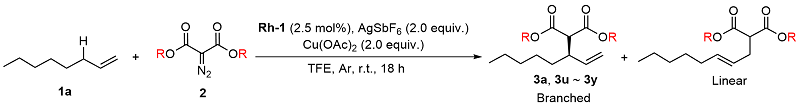
Table 3 Substrate scope of substituted diazo compounds
|
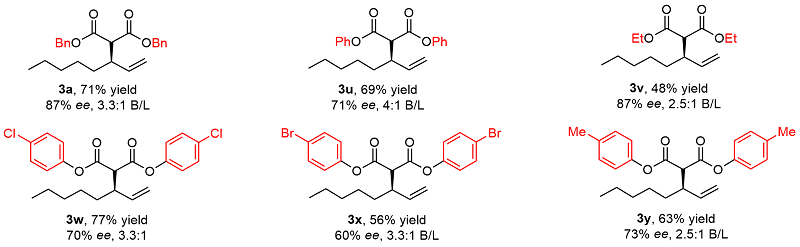 |
a Reaction conditions: 1a (0.3 mmol), 2 (0.1 mmol), Rh-1 (2.5 mol%), AgSbF6 (2.0 equiv.), Cu(OAc)2 (2.0 equiv.), TFE (0.2 mol/L), r.t., 18 h. Isolated yields (branched and linear). Enantiomeric excess was determined by HPLC analysis. Regiomeric ratio (branched/linear) was determined by analysis of the 1H NMR spectra of the crude reaction mixtures. |
3 Conclusions
4 Experimental section
4.1 General information
4.2 General procedure for Rh(III)-catalyzed allylation of olefins with diazo compounds
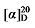 -1.04 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.38~7.26 (m, 13H), 5.68~5.57 (m, 1H), 5.53~5.46 (m, 0.33H), 5.37~5.31 (m, 0.30H), 5.15 (d, J=1.5 Hz, 3H), 5.10 (s, 2H), 5.05~4.98 (m, 2H), 3.52~3.49 (m, 0.36H), 3.47 (d, J=8.8 Hz, 1H), 2.79 (dd J=9.3, 3.7 Hz, 1H), 2.65~2.59 (m, 0.61H), 1.91 (q, J=7.0 Hz, 0.63H), 1.44~1.15 (m, 10H), 0.86 (dt, J=14.1, 7.0 Hz, 4H); 13C NMR (101 MHz, CDCl3) δ: 168.89, 168.25, 168.08, 138.14, 135.55, 135.52, 134.44, 128.66, 128.62, 128.46, 128.44, 128.42, 128.29, 125.02, 117.60, 67.18, 67.16, 67.07, 57.15, 52.46, 44.36, 32.35, 31.64, 26.73, 22.61, 14.15. HRMS (ESI-TOF) calcd for C25H31O4 [M+H]+ 395.2222, found 395.2229. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexa-ne)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=32.02 min, tR(major)=33.29 min.
-1.04 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.38~7.26 (m, 13H), 5.68~5.57 (m, 1H), 5.53~5.46 (m, 0.33H), 5.37~5.31 (m, 0.30H), 5.15 (d, J=1.5 Hz, 3H), 5.10 (s, 2H), 5.05~4.98 (m, 2H), 3.52~3.49 (m, 0.36H), 3.47 (d, J=8.8 Hz, 1H), 2.79 (dd J=9.3, 3.7 Hz, 1H), 2.65~2.59 (m, 0.61H), 1.91 (q, J=7.0 Hz, 0.63H), 1.44~1.15 (m, 10H), 0.86 (dt, J=14.1, 7.0 Hz, 4H); 13C NMR (101 MHz, CDCl3) δ: 168.89, 168.25, 168.08, 138.14, 135.55, 135.52, 134.44, 128.66, 128.62, 128.46, 128.44, 128.42, 128.29, 125.02, 117.60, 67.18, 67.16, 67.07, 57.15, 52.46, 44.36, 32.35, 31.64, 26.73, 22.61, 14.15. HRMS (ESI-TOF) calcd for C25H31O4 [M+H]+ 395.2222, found 395.2229. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexa-ne)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=32.02 min, tR(major)=33.29 min.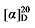 +4.32 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.38~7.27 (m, 13H), 5.83~5.71 (m, 1H), 5.55~5.44 (m, 0.32H), 5.39~5.32 (m, 0.38H), 5.15 (s, 3H), 5.11 (s, 2H), 5.04 (dt, J=17.1, 1.3 Hz, 1H), 4.97 (dt, J=10.4, 1.1 Hz, 1H), 3.49 (t, J=7.6 Hz, 0.32H), 3.41 (d, J=8.8 Hz, 1H), 3.06~2.94 (m, 1H), 2.61 (td, J=7.2, 1.3 Hz, 0.64H), 1.58 (dt, J=2.8, 1.29 Hz, 1H), 1.08 (d, J=6.8 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.1, 168.1, 139.7, 135.5, 135.5, 128.8, 128.7, 128.6, 128.5, 128.4, 128.4, 128.4, 128.3, 126.3, 115.7, 67.2, 67.1, 57.8, 52.4, 38.2, 32.0, 18.1, 18.0. HRMS (ESI-TOF) calcd for C21H23O4Na [M+Na]+ 361.1416, found 361.1415. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=34.19 min, tR(major)=34.10 min.
+4.32 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.38~7.27 (m, 13H), 5.83~5.71 (m, 1H), 5.55~5.44 (m, 0.32H), 5.39~5.32 (m, 0.38H), 5.15 (s, 3H), 5.11 (s, 2H), 5.04 (dt, J=17.1, 1.3 Hz, 1H), 4.97 (dt, J=10.4, 1.1 Hz, 1H), 3.49 (t, J=7.6 Hz, 0.32H), 3.41 (d, J=8.8 Hz, 1H), 3.06~2.94 (m, 1H), 2.61 (td, J=7.2, 1.3 Hz, 0.64H), 1.58 (dt, J=2.8, 1.29 Hz, 1H), 1.08 (d, J=6.8 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.1, 168.1, 139.7, 135.5, 135.5, 128.8, 128.7, 128.6, 128.5, 128.4, 128.4, 128.4, 128.3, 126.3, 115.7, 67.2, 67.1, 57.8, 52.4, 38.2, 32.0, 18.1, 18.0. HRMS (ESI-TOF) calcd for C21H23O4Na [M+Na]+ 361.1416, found 361.1415. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=34.19 min, tR(major)=34.10 min.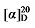 -1.37 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.37~7.27 (m, 12H), 5.69~5.56 (m, 1H), 5.54~5.44 (m, 0.25H), 5.39~5.27 (m, 0.25H), 5.20~5.13 (m, 3H), 5.10 (s, 2H), 5.05~4.98 (m, 2H), 3.50 (t, J=7.6 Hz, 0.22H), 3.46 (d, J=8.8 Hz, 1H), 2.81 (d, J=1.3 Hz, 1H), 2.66~2.59 (m, 0.41H), 1.90 (q, J=7.2, 9.2 Hz, 0.45H), 1.42~1.14 (m, 5H), 0.86 (d, J=7.4 Hz, 0.60H), 0.84~0.79 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.1, 138.1, 135.5, 135.5, 134.2, 128.7, 128.6, 128.5, 128.5, 128.4, 128.4, 128.3, 125.3, 117.6, 67.2, 67.1, 57.1, 52.5, 44.1, 34.6, 34.5, 32.0, 22.5, 20.2, 13.9, 13.7. HRMS (ESI-TOF) calcd for C23H27O4 [M+H]+ 367.1909, found 367.1912. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=13.33 min, tR(major)=14.67 min.
-1.37 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.37~7.27 (m, 12H), 5.69~5.56 (m, 1H), 5.54~5.44 (m, 0.25H), 5.39~5.27 (m, 0.25H), 5.20~5.13 (m, 3H), 5.10 (s, 2H), 5.05~4.98 (m, 2H), 3.50 (t, J=7.6 Hz, 0.22H), 3.46 (d, J=8.8 Hz, 1H), 2.81 (d, J=1.3 Hz, 1H), 2.66~2.59 (m, 0.41H), 1.90 (q, J=7.2, 9.2 Hz, 0.45H), 1.42~1.14 (m, 5H), 0.86 (d, J=7.4 Hz, 0.60H), 0.84~0.79 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.1, 138.1, 135.5, 135.5, 134.2, 128.7, 128.6, 128.5, 128.5, 128.4, 128.4, 128.3, 125.3, 117.6, 67.2, 67.1, 57.1, 52.5, 44.1, 34.6, 34.5, 32.0, 22.5, 20.2, 13.9, 13.7. HRMS (ESI-TOF) calcd for C23H27O4 [M+H]+ 367.1909, found 367.1912. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=13.33 min, tR(major)=14.67 min.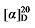 -17.32 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.37~7.27 (m, 12H), 5.69~5.57 (m, 1H), 5.53~5.45 (m, 0.29H), 5.37~5.28 (m, 0.33H), 5.15 (d, J=2.0 Hz, 3H), 5.10 (s, 2H), 5.05~5.01 (m, 1H), 5.00 (s, 1H), 3.51 (d, J=7.6 Hz, 0.18H), 3.47 (d, J=8.7 Hz, 1H), 2.79 (q, J=9.2, 1H), 2.65~2.58 (m, 0.51H), 1.91 (q, J=5.2 Hz, 0.58H), 1.46~1.36 (m, 1H), 1.32~1.14 (m, 14H), 0.87 (t, J=7.0 Hz, 4H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.1, 138.1, 135.5, 135.5, 134.4, 128.7, 128.6, 128.5, 128.5, 128.4, 128.4, 128.3, 125.0, 117.6, 67.2, 67.2, 67.1, 57.1, 52.5, 44.4, 32.6, 32.4, 32.0, 31.9, 29.4, 29.3, 29.2, 27.1, 22.8, 22.8, 14.2. HRMS (ESI-TOF) calcd for C27H33O4 [M-H]+ 421.2386, found 421.2384. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(he-xane)∶V(i-PrOH)=99∶1, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=29.27 min, tR(major)=34.27 min.
-17.32 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.37~7.27 (m, 12H), 5.69~5.57 (m, 1H), 5.53~5.45 (m, 0.29H), 5.37~5.28 (m, 0.33H), 5.15 (d, J=2.0 Hz, 3H), 5.10 (s, 2H), 5.05~5.01 (m, 1H), 5.00 (s, 1H), 3.51 (d, J=7.6 Hz, 0.18H), 3.47 (d, J=8.7 Hz, 1H), 2.79 (q, J=9.2, 1H), 2.65~2.58 (m, 0.51H), 1.91 (q, J=5.2 Hz, 0.58H), 1.46~1.36 (m, 1H), 1.32~1.14 (m, 14H), 0.87 (t, J=7.0 Hz, 4H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.1, 138.1, 135.5, 135.5, 134.4, 128.7, 128.6, 128.5, 128.5, 128.4, 128.4, 128.3, 125.0, 117.6, 67.2, 67.2, 67.1, 57.1, 52.5, 44.4, 32.6, 32.4, 32.0, 31.9, 29.4, 29.3, 29.2, 27.1, 22.8, 22.8, 14.2. HRMS (ESI-TOF) calcd for C27H33O4 [M-H]+ 421.2386, found 421.2384. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(he-xane)∶V(i-PrOH)=99∶1, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=29.27 min, tR(major)=34.27 min.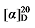 -7.12 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.38~7.26 (m, 21H), 5.69 (dt, J=17.0, 10.1 Hz, 1H), 5.53~5.43 (m, 1H), 5.35~5.25 (m, 1H), 5.15 (s, 6H), 5.10 (s, 2H), 5.08~4.97 (m, 2H), 3.67 (d, J=9.5 Hz, 1H), 3.51 (t, J=7.6 Hz, 1H), 2.70 (td, J=9.7, 5.1 Hz, 1H), 2.62 (td, J=7.2, 1.0 Hz, 2.26H), 2.24~2.13 (m, 1.13H), 1.73 (pd, J=6.8, 5.0 Hz, 1.05H), 0.91 (d, J=6.8 Hz, 7H), 0.88 (d, J=6.7 Hz, 3H), 0.82 (d, J=6.8 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.4, 168.1, 141.3, 135.6, 134.8, 128.7, 128.6, 128.6, 128.5, 128.4, 128.4, 128.4, 128.3, 122.1, 118.9, 67.2, 67.1, 67.1, 55.1, 52.5, 50.5, 31.9, 31.0, 29.1, 22.4, 21.3, 17.7. HRMS (ESI-TOF) calcd for C23H26-NO4 [M+NH4]+ 384.2175, found 384.2170. HPLC analysis: chiral MQ (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=95∶5, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=29.35 min, tR(major)=34.14 min.
-7.12 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.38~7.26 (m, 21H), 5.69 (dt, J=17.0, 10.1 Hz, 1H), 5.53~5.43 (m, 1H), 5.35~5.25 (m, 1H), 5.15 (s, 6H), 5.10 (s, 2H), 5.08~4.97 (m, 2H), 3.67 (d, J=9.5 Hz, 1H), 3.51 (t, J=7.6 Hz, 1H), 2.70 (td, J=9.7, 5.1 Hz, 1H), 2.62 (td, J=7.2, 1.0 Hz, 2.26H), 2.24~2.13 (m, 1.13H), 1.73 (pd, J=6.8, 5.0 Hz, 1.05H), 0.91 (d, J=6.8 Hz, 7H), 0.88 (d, J=6.7 Hz, 3H), 0.82 (d, J=6.8 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.4, 168.1, 141.3, 135.6, 134.8, 128.7, 128.6, 128.6, 128.5, 128.4, 128.4, 128.4, 128.3, 122.1, 118.9, 67.2, 67.1, 67.1, 55.1, 52.5, 50.5, 31.9, 31.0, 29.1, 22.4, 21.3, 17.7. HRMS (ESI-TOF) calcd for C23H26-NO4 [M+NH4]+ 384.2175, found 384.2170. HPLC analysis: chiral MQ (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=95∶5, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=29.35 min, tR(major)=34.14 min.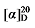 -8.19 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.34~7.28 (m, 25H), 5.72 (dt, J=17.0, 10.1 Hz, 1H), 5.50~5.41 (m, 1.51H), 5.35~5.25 (m, 1.64H), 5.15 (s, 8H), 5.11 (d, J=3.8 Hz, 2H), 5.03~4.93 (m, 2H), 3.71 (d, J=9.2 Hz, 1H), 3.50 (t, J=7.6 Hz, 1.47H), 2.67 (td, J=9.5, 5.5 Hz, 1H), 2.61 (t, J=7.2 Hz, 3H), 1.88~1.79 (m, 1.55H), 1.71~1.61 (m, 11H), 1.28~0.82 (m, 15H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.6, 168.2, 140.2, 135.8, 135.6, 128.7, 128.6, 128.5, 128.5, 128.4, 128.4, 128.3, 122.6, 118.5, 67.2, 67.1, 67.0, 54.5, 52.6, 50.3, 40.6, 39.1, 33.0, 32.1, 31.6, 28.6, 26.4, 26.3, 26.2, 26.1. HRMS (ESI-TOF) calcd for C26H31O4 [M+H]+ 407.2222, found 407.2230. HPLC analysis: chiral MX (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-Pr-OH)=95∶5, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=13.74 min, tR(major)=15.70 min.
-8.19 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.34~7.28 (m, 25H), 5.72 (dt, J=17.0, 10.1 Hz, 1H), 5.50~5.41 (m, 1.51H), 5.35~5.25 (m, 1.64H), 5.15 (s, 8H), 5.11 (d, J=3.8 Hz, 2H), 5.03~4.93 (m, 2H), 3.71 (d, J=9.2 Hz, 1H), 3.50 (t, J=7.6 Hz, 1.47H), 2.67 (td, J=9.5, 5.5 Hz, 1H), 2.61 (t, J=7.2 Hz, 3H), 1.88~1.79 (m, 1.55H), 1.71~1.61 (m, 11H), 1.28~0.82 (m, 15H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.6, 168.2, 140.2, 135.8, 135.6, 128.7, 128.6, 128.5, 128.5, 128.4, 128.4, 128.3, 122.6, 118.5, 67.2, 67.1, 67.0, 54.5, 52.6, 50.3, 40.6, 39.1, 33.0, 32.1, 31.6, 28.6, 26.4, 26.3, 26.2, 26.1. HRMS (ESI-TOF) calcd for C26H31O4 [M+H]+ 407.2222, found 407.2230. HPLC analysis: chiral MX (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-Pr-OH)=95∶5, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=13.74 min, tR(major)=15.70 min.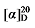 -7.17 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.38~7.20 (m, 20 H), 7.16 (d, J=8.0 Hz, 2H), 7.12~7.02 (m, 8H), 6.41 (d, J=15.8 Hz, 1H), 6.12~6.01 (m, 1H), 6.02~5.93 (m, 1H), 5.16 (d, J=3.5 Hz, 6H), 5.09~4.98 (m, 2H), 4.91 (s, 2H), 4.14~4.05 (m, 1H), 3.94 (d, J=11.0 Hz, 1H), 3.61 (t, J=7.5 Hz, 1H), 2.87~2.77 (m, 2H), 2.32 (d, J=14.2 Hz, 6H); 13C NMR (126 MHz, CDCl3) δ: 168.8, 167.7, 167.4, 138.0, 137.3, 137.0, 136.8, 135.5, 135.4, 135.3, 134.3, 133.0, 129.5, 129.3, 128.7, 128.7, 128.5, 128.5, 128.4, 128.3, 128.2, 127.9, 126.3, 124.3, 116.6, 67.4, 67.3, 67.2, 57.6, 52.3, 49.5, 32.4, 21.3, 21.2. HRMS (ESI-TOF) calcd for C26H25O4 [M+H]+ 401.1753, found 401.1759. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(mi-nor)=40.52 min, tR(major)=42.91 min.
-7.17 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.38~7.20 (m, 20 H), 7.16 (d, J=8.0 Hz, 2H), 7.12~7.02 (m, 8H), 6.41 (d, J=15.8 Hz, 1H), 6.12~6.01 (m, 1H), 6.02~5.93 (m, 1H), 5.16 (d, J=3.5 Hz, 6H), 5.09~4.98 (m, 2H), 4.91 (s, 2H), 4.14~4.05 (m, 1H), 3.94 (d, J=11.0 Hz, 1H), 3.61 (t, J=7.5 Hz, 1H), 2.87~2.77 (m, 2H), 2.32 (d, J=14.2 Hz, 6H); 13C NMR (126 MHz, CDCl3) δ: 168.8, 167.7, 167.4, 138.0, 137.3, 137.0, 136.8, 135.5, 135.4, 135.3, 134.3, 133.0, 129.5, 129.3, 128.7, 128.7, 128.5, 128.5, 128.4, 128.3, 128.2, 127.9, 126.3, 124.3, 116.6, 67.4, 67.3, 67.2, 57.6, 52.3, 49.5, 32.4, 21.3, 21.2. HRMS (ESI-TOF) calcd for C26H25O4 [M+H]+ 401.1753, found 401.1759. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(mi-nor)=40.52 min, tR(major)=42.91 min.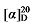 +11.43 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.35~7.28 (m, 12H), 7.25~7.14 (m, 5H), 7.09~7.02 (m, 2H), 5.80~5.69 (m, 1H), 5.67~5.61 (m, 0.21H), 5.47~5.19 (m, 0.19H), 5.15~5.09 (m, 5H), 4.98~4.81 (m, 2H), 3.52 (d, J=7.8 Hz, 1H), 3.17~3.16 (m, 0.07 H) 3.15~3.05 (m, 1H), 2.87~2.79 (m, 1H), 2.70~2.59 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 168.3, 168.0, 139.0, 137.2, 129.5, 128.7, 128.7, 128.7, 128.6, 128.6, 128.5, 128.5, 128.5, 128.3, 128.3, 126.4, 117.8, 67.3, 67.2, 55.9, 45.7, 38.9. HRMS (ESI-TOF) calcd for C27H25O4 [M-H]+ 413.1758, found 413.156. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexane)∶V(i-PrOH)=95∶5, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=10.45 min, tR(major)=11.38 min.
+11.43 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.35~7.28 (m, 12H), 7.25~7.14 (m, 5H), 7.09~7.02 (m, 2H), 5.80~5.69 (m, 1H), 5.67~5.61 (m, 0.21H), 5.47~5.19 (m, 0.19H), 5.15~5.09 (m, 5H), 4.98~4.81 (m, 2H), 3.52 (d, J=7.8 Hz, 1H), 3.17~3.16 (m, 0.07 H) 3.15~3.05 (m, 1H), 2.87~2.79 (m, 1H), 2.70~2.59 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 168.3, 168.0, 139.0, 137.2, 129.5, 128.7, 128.7, 128.7, 128.6, 128.6, 128.5, 128.5, 128.5, 128.3, 128.3, 126.4, 117.8, 67.3, 67.2, 55.9, 45.7, 38.9. HRMS (ESI-TOF) calcd for C27H25O4 [M-H]+ 413.1758, found 413.156. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexane)∶V(i-PrOH)=95∶5, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=10.45 min, tR(major)=11.38 min.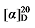 +12.76 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.36~7.22 (m, 20H), 7.21~7.10 (m, 5H), 5.67~5.55 (m, 1H), 5.52~5.42 (m, 0.69H), 5.36~5.29 (m, 0.69H), 5.14 (d, J=1.3 Hz, 2H), 5.12 (d, J=3.0 Hz, 2H), 5.09 (s, 2H), 5.04~4.99 (m, 1H), 4.98 (s, 1H), 3.49 (t, J=7.7 Hz, 0.65H), 3.46 (d, J=8.8 Hz, 1H), 2.78 (q, J=9.2, 1H), 2.59 (dt, J=15.6, 7.8 Hz, 3H), 2.54~2.49 (m, 1.49H), 1.94 (q, J=7.1 Hz, 1.30H), 1.57~1.48 (m, 3H), 1.47~1.40 (m, 1H), 1.39~1.28 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.0, 142.7, 138.0, 135.5, 135.5, 135.5, 134.1, 128.7, 128.6, 128.5, 128.5, 128.5, 128.4, 128.4, 128.4, 128.3, 125.7, 125.3, 117.7, 67.2, 67.2, 67.1, 57.1, 52.4, 44.2, 35.9, 35.9, 32.4, 32.2, 32.0, 31.2, 31.0, 29.0, 26.7. HRMS (ESI-TOF) calcd for C30H31O4 [M+H]+ 455.2227, found 455.2229. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=41.17 min, tR(major)=43.73 min.
+12.76 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.36~7.22 (m, 20H), 7.21~7.10 (m, 5H), 5.67~5.55 (m, 1H), 5.52~5.42 (m, 0.69H), 5.36~5.29 (m, 0.69H), 5.14 (d, J=1.3 Hz, 2H), 5.12 (d, J=3.0 Hz, 2H), 5.09 (s, 2H), 5.04~4.99 (m, 1H), 4.98 (s, 1H), 3.49 (t, J=7.7 Hz, 0.65H), 3.46 (d, J=8.8 Hz, 1H), 2.78 (q, J=9.2, 1H), 2.59 (dt, J=15.6, 7.8 Hz, 3H), 2.54~2.49 (m, 1.49H), 1.94 (q, J=7.1 Hz, 1.30H), 1.57~1.48 (m, 3H), 1.47~1.40 (m, 1H), 1.39~1.28 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.0, 142.7, 138.0, 135.5, 135.5, 135.5, 134.1, 128.7, 128.6, 128.5, 128.5, 128.5, 128.4, 128.4, 128.4, 128.3, 125.7, 125.3, 117.7, 67.2, 67.2, 67.1, 57.1, 52.4, 44.2, 35.9, 35.9, 32.4, 32.2, 32.0, 31.2, 31.0, 29.0, 26.7. HRMS (ESI-TOF) calcd for C30H31O4 [M+H]+ 455.2227, found 455.2229. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=41.17 min, tR(major)=43.73 min.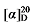 +6.02 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.37~7.26 (m, 17H), 7.14~7.05 (m, 2H), 6.97~6.88 (m, 2H), 6.82~6.71 (m, 2H), 5.69~5.58 (m, 1H), 5.51~5.42 (m, 0.35H), 5.38~5.31 (m, 0.49H), 5.17~5.10 (m, 6H), 5.07~4.99 (m, 2H), 3.51 (t, J=7.6 Hz, 0.38H), 3.47 (d, J=8.7 Hz, 1H), 2.84~2.72 (m, 4H), 2.63 (t, J=7.2 Hz, 1H), 2.00~1.92 (m, 1H), 1.65~1.57 (m, 3H), 1.50~1.20 (m, 8H); 13C NMR (101 MHz, CDCl3) δ: 168.8, 168.2, 168.0, 145.5, 137.9, 135.5, 135.5, 135.5, 133.9, 128.7, 128.7, 128.6, 128.6, 128.5, 128.5, 128.4, 128.3, 128.3, 128.1, 126.8, 126.8, 125.4, 124.1, 124.1, 122.9, 117.8, 67.2, 67.2, 67.1, 57.1, 52.4, 44.2, 32.2, 32.0, 32.0, 31.5, 31.3, 29.9, 29.8, 29.8, 28.7, 26.5. HRMS (ESI-TOF) calcd for C28H31O4S [M+H]+ 463.1943, found 463.1944. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=50.17 min, tR(major)=53.21 min.
+6.02 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.37~7.26 (m, 17H), 7.14~7.05 (m, 2H), 6.97~6.88 (m, 2H), 6.82~6.71 (m, 2H), 5.69~5.58 (m, 1H), 5.51~5.42 (m, 0.35H), 5.38~5.31 (m, 0.49H), 5.17~5.10 (m, 6H), 5.07~4.99 (m, 2H), 3.51 (t, J=7.6 Hz, 0.38H), 3.47 (d, J=8.7 Hz, 1H), 2.84~2.72 (m, 4H), 2.63 (t, J=7.2 Hz, 1H), 2.00~1.92 (m, 1H), 1.65~1.57 (m, 3H), 1.50~1.20 (m, 8H); 13C NMR (101 MHz, CDCl3) δ: 168.8, 168.2, 168.0, 145.5, 137.9, 135.5, 135.5, 135.5, 133.9, 128.7, 128.7, 128.6, 128.6, 128.5, 128.5, 128.4, 128.3, 128.3, 128.1, 126.8, 126.8, 125.4, 124.1, 124.1, 122.9, 117.8, 67.2, 67.2, 67.1, 57.1, 52.4, 44.2, 32.2, 32.0, 32.0, 31.5, 31.3, 29.9, 29.8, 29.8, 28.7, 26.5. HRMS (ESI-TOF) calcd for C28H31O4S [M+H]+ 463.1943, found 463.1944. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=50.17 min, tR(major)=53.21 min.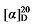 +6.82 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.38~7.27 (m, 14H), 5.62 (dt, J=17.4, 9.5 Hz, 1H), 5.42 (q, J=5.5 Hz, 1H), 5.16 (s, 2H), 5.15 (d, J=1.2 Hz, 2H), 5.11 (s, 2H), 5.08~5.00 (m, 2H), 3.54~3.36 (m, 4.41H), 2.80 (q, J=9.2, 1H), 2.67~2.59 (m, 1H), 2.07 (q, J=6.4, 1H), 1.84~1.72 (m, 2H), 1.70~1.63 (m, 1H), 1.59~1.53 (m, 1H), 1.47~1.37 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 168.7, 168.0, 167.8, 137.4, 135.5, 135.4, 132.1, 128.7, 128.6, 128.5, 128.5, 128.5, 128.3, 126.8, 118.4, 67.3, 67.2, 67.2, 57.0, 52.2, 44.7, 44.3, 43.8, 31.9, 31.9, 30.3, 29.6, 29.5. HRMS (ESI-TOF) calcd for C23H24ClO4 [M-H]+ 399.1368, found 399.1370. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=20.54 min, tR(major)=21.86 min.
+6.82 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.38~7.27 (m, 14H), 5.62 (dt, J=17.4, 9.5 Hz, 1H), 5.42 (q, J=5.5 Hz, 1H), 5.16 (s, 2H), 5.15 (d, J=1.2 Hz, 2H), 5.11 (s, 2H), 5.08~5.00 (m, 2H), 3.54~3.36 (m, 4.41H), 2.80 (q, J=9.2, 1H), 2.67~2.59 (m, 1H), 2.07 (q, J=6.4, 1H), 1.84~1.72 (m, 2H), 1.70~1.63 (m, 1H), 1.59~1.53 (m, 1H), 1.47~1.37 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 168.7, 168.0, 167.8, 137.4, 135.5, 135.4, 132.1, 128.7, 128.6, 128.5, 128.5, 128.5, 128.3, 126.8, 118.4, 67.3, 67.2, 67.2, 57.0, 52.2, 44.7, 44.3, 43.8, 31.9, 31.9, 30.3, 29.6, 29.5. HRMS (ESI-TOF) calcd for C23H24ClO4 [M-H]+ 399.1368, found 399.1370. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=20.54 min, tR(major)=21.86 min.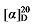 +19.70 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.36~7.30 (m, 15H), 5.62 (dt, J=17.4, 9.6 Hz, 1H), 5.44~5.38 (m, 1H), 5.15 (d, J=4.3 Hz, 3H), 5.11 (s, 2H), 5.04 (q, J=13.6, 2H), 3.51 (d, J=7.6 Hz, 0.25H), 3.49~3.45 (m, 1H), 3.37~3.23 (m, 3H), 3.00 (d, J=5.3 Hz, 0.46H), 2.80 (q, J=9.3, 1H), 2.65~2.60 (m, 0.48H), 2.12~2.03 (m, 1H), 1.91~1.80 (m, 2H), 1.79~1.70 (m, 1H), 1.59~1.52 (m, 1H), 1.48~1.36 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 167.96, 167.81, 137.37, 135.40, 128.71, 128.65, 128.56, 128.51, 128.35, 118.42, 67.35, 67.23, 56.98, 43.73, 33.33, 30.89, 30.52. HRMS (ESI-TOF) calcd for C23H26BrO4 [M+H]+ 443.0863, found 443.0865. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=57.07 min, tR(major)=57.68 min.
+19.70 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.36~7.30 (m, 15H), 5.62 (dt, J=17.4, 9.6 Hz, 1H), 5.44~5.38 (m, 1H), 5.15 (d, J=4.3 Hz, 3H), 5.11 (s, 2H), 5.04 (q, J=13.6, 2H), 3.51 (d, J=7.6 Hz, 0.25H), 3.49~3.45 (m, 1H), 3.37~3.23 (m, 3H), 3.00 (d, J=5.3 Hz, 0.46H), 2.80 (q, J=9.3, 1H), 2.65~2.60 (m, 0.48H), 2.12~2.03 (m, 1H), 1.91~1.80 (m, 2H), 1.79~1.70 (m, 1H), 1.59~1.52 (m, 1H), 1.48~1.36 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 167.96, 167.81, 137.37, 135.40, 128.71, 128.65, 128.56, 128.51, 128.35, 118.42, 67.35, 67.23, 56.98, 43.73, 33.33, 30.89, 30.52. HRMS (ESI-TOF) calcd for C23H26BrO4 [M+H]+ 443.0863, found 443.0865. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=57.07 min, tR(major)=57.68 min.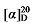 -4.11 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.37~7.25 (m, 11H), 5.68~5.54 (m, 1H), 5.21~5.17 (m, 0.22H), 5.15 (d, J=1.2 Hz, 2H), 5.11 (s, 2H), 5.09~5.02 (m, 1H), 5.02 (s, 1H), 3.51 (d, J=7.3 Hz, 0.06H), 3.46 (d, J=8.7 Hz, 1H), 3.37 (d, J=6.8 Hz, 0.20H), 3.32 (td, J=6.8, 1.3 Hz, 2H), 2.85~2.73 (m, 1H), 2.62 (t,J=7.0 Hz, 0.10H), 1.95 (q, J=7.0 Hz, 0.11H), 1.85~1.67 (m, 2H), 1.48~1.38 (m, 2H), 1.30 (q, J=2.6 Hz, 2H); 13C NMR (101 MHz, CDCl3) δ: 168.1, 167.9, 137.6, 135.4, 135.4, 128.7, 128.7, 128.6, 128.5, 128.5, 128.5, 128.5, 118.1, 67.2, 67.1, 57.0, 44.1, 33.6, 32.4, 31.3, 25.6. HRMS (ESI-TOF) calcd for C24H26BrO4 [M-H]+ 457.1020, found 457.1021. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=32.87 min, tR(major)=35.33 min.
-4.11 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.37~7.25 (m, 11H), 5.68~5.54 (m, 1H), 5.21~5.17 (m, 0.22H), 5.15 (d, J=1.2 Hz, 2H), 5.11 (s, 2H), 5.09~5.02 (m, 1H), 5.02 (s, 1H), 3.51 (d, J=7.3 Hz, 0.06H), 3.46 (d, J=8.7 Hz, 1H), 3.37 (d, J=6.8 Hz, 0.20H), 3.32 (td, J=6.8, 1.3 Hz, 2H), 2.85~2.73 (m, 1H), 2.62 (t,J=7.0 Hz, 0.10H), 1.95 (q, J=7.0 Hz, 0.11H), 1.85~1.67 (m, 2H), 1.48~1.38 (m, 2H), 1.30 (q, J=2.6 Hz, 2H); 13C NMR (101 MHz, CDCl3) δ: 168.1, 167.9, 137.6, 135.4, 135.4, 128.7, 128.7, 128.6, 128.5, 128.5, 128.5, 128.5, 118.1, 67.2, 67.1, 57.0, 44.1, 33.6, 32.4, 31.3, 25.6. HRMS (ESI-TOF) calcd for C24H26BrO4 [M-H]+ 457.1020, found 457.1021. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=32.87 min, tR(major)=35.33 min.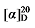 +30.35 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.35~7.27 (m, 14H), 5.67~5.57 (m, 1H), 5.52~5.45 (m, 0.42H), 5.37~5.30 (m, 0.45H), 5.14 (dd, J=3.1, 1.5 Hz, 3H), 5.10 (s, 2H), 5.05~5.01 (m, 1H), 5.00 (s, 1H), 3.62 (td, J=6.6, 2.6 Hz, 2.82H), 3.50 (t, J=7.6 Hz, 0.42H), 3.46 (d, J=8.8 Hz, 1H), 2.78 (q, J=9.3 Hz, 1H), 2.62 (t, J=7.2 Hz, 0.80H), 1.91 (q, J=6.7 Hz, 0.87H), 1.58~1.51 (m, 4H), 1.35~1.21 (m, 12H); 13C NMR (126 MHz, CDCl3) δ: 168.9, 168.2, 168.0, 138.1, 135.5, 135.5, 135.5, 134.3, 128.6, 128.6, 128.5, 128.4, 128.4, 128.3, 125.1, 117.6, 67.2, 67.1, 67.1, 63.1, 63.1, 57.1, 52.4, 44.3, 32.9, 32.8, 32.5, 32.3, 32.0, 29.3, 29.3, 29.2, 29.1, 27.0, 25.8, 25.7. HRMS (ESI-TOF) calcd for C27H34O5Na [M+Na]+ 461.2304, found 461.2308. HPLC analysis: chiral MD (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=90∶10, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=20.29 min, tR(major)=21.57 min.
+30.35 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.35~7.27 (m, 14H), 5.67~5.57 (m, 1H), 5.52~5.45 (m, 0.42H), 5.37~5.30 (m, 0.45H), 5.14 (dd, J=3.1, 1.5 Hz, 3H), 5.10 (s, 2H), 5.05~5.01 (m, 1H), 5.00 (s, 1H), 3.62 (td, J=6.6, 2.6 Hz, 2.82H), 3.50 (t, J=7.6 Hz, 0.42H), 3.46 (d, J=8.8 Hz, 1H), 2.78 (q, J=9.3 Hz, 1H), 2.62 (t, J=7.2 Hz, 0.80H), 1.91 (q, J=6.7 Hz, 0.87H), 1.58~1.51 (m, 4H), 1.35~1.21 (m, 12H); 13C NMR (126 MHz, CDCl3) δ: 168.9, 168.2, 168.0, 138.1, 135.5, 135.5, 135.5, 134.3, 128.6, 128.6, 128.5, 128.4, 128.4, 128.3, 125.1, 117.6, 67.2, 67.1, 67.1, 63.1, 63.1, 57.1, 52.4, 44.3, 32.9, 32.8, 32.5, 32.3, 32.0, 29.3, 29.3, 29.2, 29.1, 27.0, 25.8, 25.7. HRMS (ESI-TOF) calcd for C27H34O5Na [M+Na]+ 461.2304, found 461.2308. HPLC analysis: chiral MD (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=90∶10, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=20.29 min, tR(major)=21.57 min.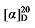 +30.35 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 8.09~8.02 (m, 2.84H), 7.58~7.52 (m, 1.43H), 7.47~7.41 (m, 2.94H), 7.34~7.29 (m, 14H), 5.68~5.57 (m, 1H), 5.52~5.45 (m, 0.44H), 5.37~5.31 (m, 0.43H), 5.15 (dd, J=3.6, 1.7 Hz, 3H), 5.10 (s, 2H), 5.05~4.97 (m, 2H), 4.31 (td, J=6.7, 3.2 Hz, 2.83H), 3.50 (t, J=7.6 Hz, 0.39H), 3.47 (d, J=8.8 Hz, 1H), 2.79 (q, J=9.3 Hz, 1H), 2.65~2.60 (m, 0.80H), 1.92 (q, J=6.7 Hz, 0.85H), 1.78~1.71 (m, 2.80H), 1.45~1.39 (m, 2.97H), 1.24~1.15 (m, 2.33H); 13C NMR (126 MHz, CDCl3) δ: 168.8, 168.2, 168.0, 166.8, 165.2, 138.0, 135.5, 135.5, 135.5, 134.2, 132.9, 130.6, 129.6, 128.8, 128.8, 128.6, 128.6, 128.5, 128.4, 128.4, 128.3, 125.1, 117.7, 68.1, 67.2, 67.1, 67.1, 65.2, 57.1, 52.4, 44.3, 32.3, 29.3, 29.3, 29.2, 29.2, 29.1, 28.8, 28.8, 27.0, 26.1, 26.1. HRMS (ESI-TOF) calcd for C31H32O6Na [M+Na]+ 523.2097, found 523.2099. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=27.81 min, tR(major)=30.70 min.
+30.35 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 8.09~8.02 (m, 2.84H), 7.58~7.52 (m, 1.43H), 7.47~7.41 (m, 2.94H), 7.34~7.29 (m, 14H), 5.68~5.57 (m, 1H), 5.52~5.45 (m, 0.44H), 5.37~5.31 (m, 0.43H), 5.15 (dd, J=3.6, 1.7 Hz, 3H), 5.10 (s, 2H), 5.05~4.97 (m, 2H), 4.31 (td, J=6.7, 3.2 Hz, 2.83H), 3.50 (t, J=7.6 Hz, 0.39H), 3.47 (d, J=8.8 Hz, 1H), 2.79 (q, J=9.3 Hz, 1H), 2.65~2.60 (m, 0.80H), 1.92 (q, J=6.7 Hz, 0.85H), 1.78~1.71 (m, 2.80H), 1.45~1.39 (m, 2.97H), 1.24~1.15 (m, 2.33H); 13C NMR (126 MHz, CDCl3) δ: 168.8, 168.2, 168.0, 166.8, 165.2, 138.0, 135.5, 135.5, 135.5, 134.2, 132.9, 130.6, 129.6, 128.8, 128.8, 128.6, 128.6, 128.5, 128.4, 128.4, 128.3, 125.1, 117.7, 68.1, 67.2, 67.1, 67.1, 65.2, 57.1, 52.4, 44.3, 32.3, 29.3, 29.3, 29.2, 29.2, 29.1, 28.8, 28.8, 27.0, 26.1, 26.1. HRMS (ESI-TOF) calcd for C31H32O6Na [M+Na]+ 523.2097, found 523.2099. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=27.81 min, tR(major)=30.70 min.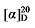 -27.06 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.34~7.27 (m, 14H), 5.68~5.55 (m, 1H), 5.51~5.43 (m, 0.46H), 5.38 (dd, J=14.5, 7.5 Hz, 0.56H), 5.14 (s, 3.44H), 5.10 (s, 2H), 5.04 (dd, J=13.8, 2.4 Hz, 2H), 4.05~3.91 (m, 2.87H), 3.48 (dd, J=14.4, 8.0 Hz, 1.39H), 2.79 (td, J=9.0, 6.2 Hz, 1H), 2.62 (t, J=7.1 Hz, 0.86H), 2.02 (d, J=11.3 Hz, 5.34H), 1.60 (s, 1H), 1.50 (td, J=11.9, 4.1 Hz, 2H), 1.37~1.29 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 171.22, 168.78, 168.01, 167.85, 137.43, 135.52, 135.42, 132.74, 128.75, 128.69, 128.67, 128.64, 128.54, 128.47, 128.44, 128.32, 126.22, 118.34, 67.28, 67.21, 67.18, 64.25, 57.01, 44.04, 28.70, 28.22, 26.37, 21.08. HRMS (ESI-TOF) calcd for C25H27O6 [M-H]+ 423.1713, found 423.1714. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexane)∶V(i-PrOH)=95∶5, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=46.70 min, tR(major)=54.66 min.
-27.06 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.34~7.27 (m, 14H), 5.68~5.55 (m, 1H), 5.51~5.43 (m, 0.46H), 5.38 (dd, J=14.5, 7.5 Hz, 0.56H), 5.14 (s, 3.44H), 5.10 (s, 2H), 5.04 (dd, J=13.8, 2.4 Hz, 2H), 4.05~3.91 (m, 2.87H), 3.48 (dd, J=14.4, 8.0 Hz, 1.39H), 2.79 (td, J=9.0, 6.2 Hz, 1H), 2.62 (t, J=7.1 Hz, 0.86H), 2.02 (d, J=11.3 Hz, 5.34H), 1.60 (s, 1H), 1.50 (td, J=11.9, 4.1 Hz, 2H), 1.37~1.29 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 171.22, 168.78, 168.01, 167.85, 137.43, 135.52, 135.42, 132.74, 128.75, 128.69, 128.67, 128.64, 128.54, 128.47, 128.44, 128.32, 126.22, 118.34, 67.28, 67.21, 67.18, 64.25, 57.01, 44.04, 28.70, 28.22, 26.37, 21.08. HRMS (ESI-TOF) calcd for C25H27O6 [M-H]+ 423.1713, found 423.1714. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexane)∶V(i-PrOH)=95∶5, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=46.70 min, tR(major)=54.66 min.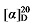 +47.68 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.38~7.24 (m, 18H), 6.95~6.90 (m, 1H), 6.90~6.84 (m, 2H), 5.68~5.58 (m, 1H), 5.53~5.46 (m, 0.42H), 5.40~5.33 (m, 0.45H), 5.14 (d, J=1.3 Hz, 3H), 5.10 (s, 2H), 5.06~5.00 (m, 2H), 3.90 (dt, J=17.4, 6.4 Hz, 2.80H), 3.48 (q, J=8.4 Hz, 1.40H), 2.85~2.76 (m, 1H), 2.65~2.59 (m, 0.76H), 1.99 (q, J=7.1 Hz, 0.80H), 1.77~1.62 (m, 3H), 1.53~1.42 (m, 3H), 1.37~1.30 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.0, 159.1, 137.8, 135.5, 135.5, 133.8, 129.5, 129.5, 128.8, 128.7, 128.6, 128.6, 128.5, 128.5, 128.5, 128.3, 125.7, 120.6, 118.0, 114.6, 114.6, 67.7, 67.6, 67.2, 67.2, 67.1, 57.1, 52.4, 44.3, 32.2, 32.1, 29.1, 28.8, 25.8, 23.7. HRMS (ESI-TOF) calcd for C30H33O5 [M+H]+ 473.2328, found 473.2325. HPLC analysis: chiral MX (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=33.32 min, tR(major)=43.37 min.
+47.68 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.38~7.24 (m, 18H), 6.95~6.90 (m, 1H), 6.90~6.84 (m, 2H), 5.68~5.58 (m, 1H), 5.53~5.46 (m, 0.42H), 5.40~5.33 (m, 0.45H), 5.14 (d, J=1.3 Hz, 3H), 5.10 (s, 2H), 5.06~5.00 (m, 2H), 3.90 (dt, J=17.4, 6.4 Hz, 2.80H), 3.48 (q, J=8.4 Hz, 1.40H), 2.85~2.76 (m, 1H), 2.65~2.59 (m, 0.76H), 1.99 (q, J=7.1 Hz, 0.80H), 1.77~1.62 (m, 3H), 1.53~1.42 (m, 3H), 1.37~1.30 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.0, 159.1, 137.8, 135.5, 135.5, 133.8, 129.5, 129.5, 128.8, 128.7, 128.6, 128.6, 128.5, 128.5, 128.5, 128.3, 125.7, 120.6, 118.0, 114.6, 114.6, 67.7, 67.6, 67.2, 67.2, 67.1, 57.1, 52.4, 44.3, 32.2, 32.1, 29.1, 28.8, 25.8, 23.7. HRMS (ESI-TOF) calcd for C30H33O5 [M+H]+ 473.2328, found 473.2325. HPLC analysis: chiral MX (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=33.32 min, tR(major)=43.37 min.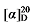 +30.35 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.80 (dd, J=3.7, 1.3 Hz, 0.54H), 7.78 (dd, J=3.7, 1.3 Hz, 2H), 7.55~7.53 (m, 0.55H), 7.53~7.51 (m, 2H), 7.33~7.28 (m, 15H), 7.15~7.03 (m, 2H), 5.71~5.60 (m, 1H), 5.54~5.48 (m, 0.51H), 5.44~5.36 (m, 0.53H), 5.17~5.11 (m, 6H), 5.11~5.03 (m, 2H), 4.28~4.15 (m, 3H), 3.52 (t, J=8.9 Hz, 0.46H), 3.50 (d, J=8.9 Hz, 1H), 2.90~2.82 (m, 1H), 2.67~2.60 (m, 1H), 2.22 (s, 1H), 2.11~2.03 (m, 1H), 1.83~1.70 (m, 3H), 1.66~1.55 (m, 2H), 1.48~1.40 (m, 1H); 13C NMR (126 MHz, CDCl3) δ: 168.7, 167.9, 167.8, 164.3, 162.3, 162.3, 137.4, 133.4, 132.4, 128.7, 128.7, 128.6, 128.6, 128.6, 128.5, 128.4, 128.4, 128.4, 128.3, 127.8, 127.0, 118.3, 68.2, 67.2, 67.1, 67.1, 64.8, 57.0, 44.0, 28.7, 26.4. HRMS (ESI-TOF) calcd for C28H29O6S [M+H]+ 493.1685, found 493.1680. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=90∶10, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=33.32 min, tR(major)=24.32 min.
+30.35 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.80 (dd, J=3.7, 1.3 Hz, 0.54H), 7.78 (dd, J=3.7, 1.3 Hz, 2H), 7.55~7.53 (m, 0.55H), 7.53~7.51 (m, 2H), 7.33~7.28 (m, 15H), 7.15~7.03 (m, 2H), 5.71~5.60 (m, 1H), 5.54~5.48 (m, 0.51H), 5.44~5.36 (m, 0.53H), 5.17~5.11 (m, 6H), 5.11~5.03 (m, 2H), 4.28~4.15 (m, 3H), 3.52 (t, J=8.9 Hz, 0.46H), 3.50 (d, J=8.9 Hz, 1H), 2.90~2.82 (m, 1H), 2.67~2.60 (m, 1H), 2.22 (s, 1H), 2.11~2.03 (m, 1H), 1.83~1.70 (m, 3H), 1.66~1.55 (m, 2H), 1.48~1.40 (m, 1H); 13C NMR (126 MHz, CDCl3) δ: 168.7, 167.9, 167.8, 164.3, 162.3, 162.3, 137.4, 133.4, 132.4, 128.7, 128.7, 128.6, 128.6, 128.6, 128.5, 128.4, 128.4, 128.4, 128.3, 127.8, 127.0, 118.3, 68.2, 67.2, 67.1, 67.1, 64.8, 57.0, 44.0, 28.7, 26.4. HRMS (ESI-TOF) calcd for C28H29O6S [M+H]+ 493.1685, found 493.1680. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=90∶10, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=33.32 min, tR(major)=24.32 min.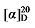 +3.71 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.78 (d, J=8.3 Hz, 3H), 7.53~7.15 (m, 17H), 5.60 (dt, J=17.5, 9.5 Hz, 1H), 5.45 (dt, J=13.9, 6.6 Hz, 1H), 5.36~5.27 (m, 1H), 5.14 (d, J=1.5 Hz, 3H), 5.09 (s, 2H), 5.05~4.99 (m, 1H), 4.98 (d, J=2.0 Hz, 1H), 4.00 (td, J=6.5, 2.9 Hz, 3H), 3.50 (d, J=7.6 Hz, 1H), 3.44 (d, J=8.8 Hz, 1H), 2.75 (q, J=9.3 Hz, 1H), 2.61 (t, J=7.2 Hz, 1H), 2.44 (s, 4H), 1.88 (q, J=6.8 Hz, 1H), 1.62 (d, J=7.0 Hz, 1H), 1.57 (d, J=6.6 Hz, 2H), 1.33~1.05 (m, 14H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.0, 144.8, 138.0, 135.5, 129.9, 128.7, 128.6, 128.5, 128.5, 128.4, 128.3, 128.0, 117.7, 70.8, 67.2, 67.1, 57.1, 44.3, 32.4, 32.3, 29.2, 28.9, 28.9, 26.9, 25.4, 21.8. HRMS (ESI-TOF) calcd for C34H41O7S [M+H]+ 593.2573, found 593.2579. HPLC analysis: chiral MX (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=90∶10, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=64.95 min, tR(major)=68.88 min.
+3.71 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.78 (d, J=8.3 Hz, 3H), 7.53~7.15 (m, 17H), 5.60 (dt, J=17.5, 9.5 Hz, 1H), 5.45 (dt, J=13.9, 6.6 Hz, 1H), 5.36~5.27 (m, 1H), 5.14 (d, J=1.5 Hz, 3H), 5.09 (s, 2H), 5.05~4.99 (m, 1H), 4.98 (d, J=2.0 Hz, 1H), 4.00 (td, J=6.5, 2.9 Hz, 3H), 3.50 (d, J=7.6 Hz, 1H), 3.44 (d, J=8.8 Hz, 1H), 2.75 (q, J=9.3 Hz, 1H), 2.61 (t, J=7.2 Hz, 1H), 2.44 (s, 4H), 1.88 (q, J=6.8 Hz, 1H), 1.62 (d, J=7.0 Hz, 1H), 1.57 (d, J=6.6 Hz, 2H), 1.33~1.05 (m, 14H); 13C NMR (101 MHz, CDCl3) δ: 168.9, 168.2, 168.0, 144.8, 138.0, 135.5, 129.9, 128.7, 128.6, 128.5, 128.5, 128.4, 128.3, 128.0, 117.7, 70.8, 67.2, 67.1, 57.1, 44.3, 32.4, 32.3, 29.2, 28.9, 28.9, 26.9, 25.4, 21.8. HRMS (ESI-TOF) calcd for C34H41O7S [M+H]+ 593.2573, found 593.2579. HPLC analysis: chiral MX (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=90∶10, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=64.95 min, tR(major)=68.88 min.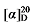 41.27 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.69~7.62 (m, 3H), 7.34~7.28 (m, 16H), 5.67~5.52 (m, 1H), 5.47~5.42 (m, 0.49H), 5.37~5.32 (m, 0.50H), 5.19~5.06 (m, 5H), 5.01 (q, J=13.9 Hz, 2H), 3.47 (d, J=8.2 Hz, 0.41H), 3.45 (d, J=8.8 Hz, 1H), 2.97~2.88 (m, 3H), 2.76 (dd, J=9.2, 3.4 Hz, 1H), 2.66 (d, J=8.2 Hz, 4H), 2.64~2.57 (m, 1H), 2.42 (d, J=2.5 Hz, 5H), 2.11~2.00 (m, 1H), 1.93 (q, J=7.1 Hz, 1H), 1.56~1.25 (m, 9H); 13C NMR (101 MHz, CDCl3) δ: 168.13, 167.96, 143.31, 137.72, 135.47, 134.71, 129.74, 128.68, 128.63, 128.50, 128.48, 128.47, 128.30, 127.55, 118.03, 67.24, 67.14, 57.05, 49.95, 44.15, 34.68, 31.79, 27.33, 24.01, 21.62. HRMS (ESI-TOF) calcd for C32H36NO6S [M-H]+ 564.2268, found 562.2266. HPLC analysis: chiral MX (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=80∶20, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=37.43 min, tR(major)=41.27 min.
41.27 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.69~7.62 (m, 3H), 7.34~7.28 (m, 16H), 5.67~5.52 (m, 1H), 5.47~5.42 (m, 0.49H), 5.37~5.32 (m, 0.50H), 5.19~5.06 (m, 5H), 5.01 (q, J=13.9 Hz, 2H), 3.47 (d, J=8.2 Hz, 0.41H), 3.45 (d, J=8.8 Hz, 1H), 2.97~2.88 (m, 3H), 2.76 (dd, J=9.2, 3.4 Hz, 1H), 2.66 (d, J=8.2 Hz, 4H), 2.64~2.57 (m, 1H), 2.42 (d, J=2.5 Hz, 5H), 2.11~2.00 (m, 1H), 1.93 (q, J=7.1 Hz, 1H), 1.56~1.25 (m, 9H); 13C NMR (101 MHz, CDCl3) δ: 168.13, 167.96, 143.31, 137.72, 135.47, 134.71, 129.74, 128.68, 128.63, 128.50, 128.48, 128.47, 128.30, 127.55, 118.03, 67.24, 67.14, 57.05, 49.95, 44.15, 34.68, 31.79, 27.33, 24.01, 21.62. HRMS (ESI-TOF) calcd for C32H36NO6S [M-H]+ 564.2268, found 562.2266. HPLC analysis: chiral MX (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=80∶20, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=37.43 min, tR(major)=41.27 min.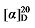 +3.28 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.45~7.34 (m, 5H), 7.29~7.21 (m, 3H), 7.17~7.08 (m, 5H), 5.88 (dt, J=17.1, 9.8 Hz, 1H), 5.75~5.67 (m, 0.27H), 5.60~5.51 (m, 0.22H), 5.31~5.21 (m, 2H), 3.88 (t, J=7.6 Hz, 0.25H), 3.85 (d, J=8.2 Hz, 1H), 3.02 (q, J=9.6 Hz, 1H), 2.88~2.82 (m, 0.47H), 2.05 (q, J=7.0 Hz, 0.56H), 1.75~1.66 (m, 1H), 1.59~1.50 (m, 1H), 1.43~1.27 (m, 7H), 0.93~0.86 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 167.44, 166.88, 166.63, 150.62, 150.58, 137.91, 135.09, 129.69, 129.67, 129.63, 126.37, 126.36, 126.32, 124.69, 121.50, 121.46, 118.31, 56.81, 52.41, 44.48, 32.65, 32.60, 32.08, 31.66, 31.47, 29.12, 26.84, 22.66, 14.15. HRMS (ESI-TOF) calcd for C23H27O4 [M+H]+ 367.1909, found 367.1904. HPLC analysis: chiral MD (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=33.32 min, tR(major)=35.16 min.
+3.28 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.45~7.34 (m, 5H), 7.29~7.21 (m, 3H), 7.17~7.08 (m, 5H), 5.88 (dt, J=17.1, 9.8 Hz, 1H), 5.75~5.67 (m, 0.27H), 5.60~5.51 (m, 0.22H), 5.31~5.21 (m, 2H), 3.88 (t, J=7.6 Hz, 0.25H), 3.85 (d, J=8.2 Hz, 1H), 3.02 (q, J=9.6 Hz, 1H), 2.88~2.82 (m, 0.47H), 2.05 (q, J=7.0 Hz, 0.56H), 1.75~1.66 (m, 1H), 1.59~1.50 (m, 1H), 1.43~1.27 (m, 7H), 0.93~0.86 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 167.44, 166.88, 166.63, 150.62, 150.58, 137.91, 135.09, 129.69, 129.67, 129.63, 126.37, 126.36, 126.32, 124.69, 121.50, 121.46, 118.31, 56.81, 52.41, 44.48, 32.65, 32.60, 32.08, 31.66, 31.47, 29.12, 26.84, 22.66, 14.15. HRMS (ESI-TOF) calcd for C23H27O4 [M+H]+ 367.1909, found 367.1904. HPLC analysis: chiral MD (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=33.32 min, tR(major)=35.16 min.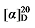 +3.28 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 5.68~5.57 (m, 1H), 5.55~5.47 (m, 0.44H), 5.38~5.29 (m, 0.40H), 5.10~5.02 (m, 2H), 4.21~4.10 (m, 5.64H), 3.39~3.27 (m, 1.36H), 2.74 (q, J=9.3 Hz, 1H), 2.59~2.53 (m, 0.89H), 1.98~1.90 (m, 0.80H), 1.47~1.39 (m, 1H), 1.25 (d, J=6.6 Hz, 16H), 0.85 (td, J=6.9, 2.7 Hz, 4H); 13C NMR (101 MHz, CDCl3) δ: 169.20, 168.56, 168.37, 138.34, 134.12, 125.31, 117.37, 61.39, 61.24, 57.17, 52.44, 44.22, 32.54, 32.37, 31.99, 31.65, 31.39, 29.09, 26.73, 22.61, 14.22, 14.12. HRMS (ESI-TOF) calcd for C15H27O4 [M+H]+ 271.1909, found 271.1905. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=21.50 min, tR(major)=25.15 min.
+3.28 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 5.68~5.57 (m, 1H), 5.55~5.47 (m, 0.44H), 5.38~5.29 (m, 0.40H), 5.10~5.02 (m, 2H), 4.21~4.10 (m, 5.64H), 3.39~3.27 (m, 1.36H), 2.74 (q, J=9.3 Hz, 1H), 2.59~2.53 (m, 0.89H), 1.98~1.90 (m, 0.80H), 1.47~1.39 (m, 1H), 1.25 (d, J=6.6 Hz, 16H), 0.85 (td, J=6.9, 2.7 Hz, 4H); 13C NMR (101 MHz, CDCl3) δ: 169.20, 168.56, 168.37, 138.34, 134.12, 125.31, 117.37, 61.39, 61.24, 57.17, 52.44, 44.22, 32.54, 32.37, 31.99, 31.65, 31.39, 29.09, 26.73, 22.61, 14.22, 14.12. HRMS (ESI-TOF) calcd for C15H27O4 [M+H]+ 271.1909, found 271.1905. HPLC analysis: chiral IG-3 (250 mm×4.6 mm, 3 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=21.50 min, tR(major)=25.15 min.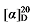 +9.59 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.33~7.27 (m, 5H), 7.22 (dd, J=8.6, 6.8 Hz, 5H), 5.66~5.54 (m, 1H), 5.52~5.43 (m, 0.31H), 5.35~5.27 (m, 0.33H), 5.14~5.07 (m, 3H), 5.05 (s, 2H), 5.04~4.97 (m, 2H), 3.48 (t, J=7.6 Hz, 0.30H), 3.44 (d, J=8.8 Hz, 1H), 2.76 (q, J=9.2 Hz, 1H), 2.63~2.55 (m, 0.60H), 1.90 (q, J=6.8 Hz, 0.63H), 1.36~1.12 (m, 10H), 0.86 (dt, J=9.2, 6.9 Hz, 4H); 13C NMR (101 MHz, CDCl3) δ: 168.66, 168.03, 167.85, 137.95, 134.58, 134.48, 134.40, 133.95, 133.92, 129.87, 129.70, 128.86, 128.80, 124.79, 117.70, 66.32, 66.30, 66.21, 57.01, 52.32, 44.34, 32.50, 32.32, 31.96, 31.61, 31.41, 29.01, 26.69, 22.60, 22.58, 14.17, 14.12. HRMS (ESI-TOF) calcd for C25H29Cl2O4 [M+H]+ 463.1443, found 463.1440. HPLC analysis: chiral MD (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=19.88 min, tR(major)=21.78 min.
+9.59 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.33~7.27 (m, 5H), 7.22 (dd, J=8.6, 6.8 Hz, 5H), 5.66~5.54 (m, 1H), 5.52~5.43 (m, 0.31H), 5.35~5.27 (m, 0.33H), 5.14~5.07 (m, 3H), 5.05 (s, 2H), 5.04~4.97 (m, 2H), 3.48 (t, J=7.6 Hz, 0.30H), 3.44 (d, J=8.8 Hz, 1H), 2.76 (q, J=9.2 Hz, 1H), 2.63~2.55 (m, 0.60H), 1.90 (q, J=6.8 Hz, 0.63H), 1.36~1.12 (m, 10H), 0.86 (dt, J=9.2, 6.9 Hz, 4H); 13C NMR (101 MHz, CDCl3) δ: 168.66, 168.03, 167.85, 137.95, 134.58, 134.48, 134.40, 133.95, 133.92, 129.87, 129.70, 128.86, 128.80, 124.79, 117.70, 66.32, 66.30, 66.21, 57.01, 52.32, 44.34, 32.50, 32.32, 31.96, 31.61, 31.41, 29.01, 26.69, 22.60, 22.58, 14.17, 14.12. HRMS (ESI-TOF) calcd for C25H29Cl2O4 [M+H]+ 463.1443, found 463.1440. HPLC analysis: chiral MD (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=98∶2, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=19.88 min, tR(major)=21.78 min.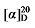 -10.88 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.48~7.43 (m, 5H), 7.15 (t, J=8.5 Hz, 5H), 5.60 (dt, J=17.5, 9.5 Hz, 1H), 5.50~5.44 (m, 0.36H), 5.33~5.28 (m, 0.37H), 5.12~5.06 (m, 3H), 5.03 (s, 2H), 5.03~4.98 (m, 2H), 3.48 (d, J=7.6 Hz, 0.30H), 3.44 (d, J=8.8 Hz, 1H), 2.75 (q, J=9.3 Hz, 1H), 2.62~2.57 (m, 0.65H), 1.90 (q, J=6.9 Hz, 0.68H), 1.40~1.16 (m, 10H), 0.86 (dt, J=11.0, 7.0 Hz, 4H); 13C NMR (126 MHz, CDCl3) δ: 168.65, 168.02, 167.84, 137.96, 134.61, 134.46, 134.43, 131.84, 131.79, 130.16, 129.99, 124.78, 122.65, 122.59, 122.57, 117.73, 66.37, 66.34, 66.25, 57.02, 52.32, 44.35, 32.52, 32.34, 31.97, 31.62, 31.43, 29.03, 26.71, 22.63, 22.60, 14.19, 14.15. HRMS (ESI-TOF) calcd for C25H29Br2O4 [M+H]+ 551.0433, found 551.0430. HPLC analysis: chiral MD (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=95∶5, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=50.71 min, tR(major)=53.21 min.
-10.88 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.48~7.43 (m, 5H), 7.15 (t, J=8.5 Hz, 5H), 5.60 (dt, J=17.5, 9.5 Hz, 1H), 5.50~5.44 (m, 0.36H), 5.33~5.28 (m, 0.37H), 5.12~5.06 (m, 3H), 5.03 (s, 2H), 5.03~4.98 (m, 2H), 3.48 (d, J=7.6 Hz, 0.30H), 3.44 (d, J=8.8 Hz, 1H), 2.75 (q, J=9.3 Hz, 1H), 2.62~2.57 (m, 0.65H), 1.90 (q, J=6.9 Hz, 0.68H), 1.40~1.16 (m, 10H), 0.86 (dt, J=11.0, 7.0 Hz, 4H); 13C NMR (126 MHz, CDCl3) δ: 168.65, 168.02, 167.84, 137.96, 134.61, 134.46, 134.43, 131.84, 131.79, 130.16, 129.99, 124.78, 122.65, 122.59, 122.57, 117.73, 66.37, 66.34, 66.25, 57.02, 52.32, 44.35, 32.52, 32.34, 31.97, 31.62, 31.43, 29.03, 26.71, 22.63, 22.60, 14.19, 14.15. HRMS (ESI-TOF) calcd for C25H29Br2O4 [M+H]+ 551.0433, found 551.0430. HPLC analysis: chiral MD (2) (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=95∶5, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=50.71 min, tR(major)=53.21 min.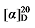 -17.22 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.23~7.17 (m, 5.66H), 7.14 (d, J=7.6 Hz, 5.68H), 5.68~5.57 (m, 1H), 5.52~5.45 (m, 0.41H), 5.36~5.29 (m, 0.45H), 5.13~5.10 (m, 3H), 5.06 (s, 2H), 5.05~4.98 (m, 2H), 3.50~3.42 (m, 1.31H), 2.82~2.74 (m, 1H), 2.61 (t, J=7.2 Hz, 0.73H), 2.36 (s, 8.54H), 1.91 (q, J=5.5 Hz, 0.86H), 1.42~1.36 (m, 1H), 1.30~1.15 (m, 9H), 0.86 (q, J=7.9 Hz, 4H); 13C NMR (101 MHz, CDCl3). HRMS (ESI-TOF) calcd for C27H35O4 [M+H]+ 423.2535, found 423.2538. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=20.54 min, tR(major)=21.56 min.
-17.22 (c 0.3, acetone); 1H NMR (500 MHz, CDCl3) δ: 7.23~7.17 (m, 5.66H), 7.14 (d, J=7.6 Hz, 5.68H), 5.68~5.57 (m, 1H), 5.52~5.45 (m, 0.41H), 5.36~5.29 (m, 0.45H), 5.13~5.10 (m, 3H), 5.06 (s, 2H), 5.05~4.98 (m, 2H), 3.50~3.42 (m, 1.31H), 2.82~2.74 (m, 1H), 2.61 (t, J=7.2 Hz, 0.73H), 2.36 (s, 8.54H), 1.91 (q, J=5.5 Hz, 0.86H), 1.42~1.36 (m, 1H), 1.30~1.15 (m, 9H), 0.86 (q, J=7.9 Hz, 4H); 13C NMR (101 MHz, CDCl3). HRMS (ESI-TOF) calcd for C27H35O4 [M+H]+ 423.2535, found 423.2538. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 0.5 mL/min, 25 ℃, 210 nm), tR(minor)=20.54 min, tR(major)=21.56 min.4.3 Synthesis of 4
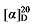 -24.52 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.37~7.26 (m, 10H), 5.82~5.68 (m, 1H), 5.51 (dt, J=17.0, 10.1 Hz, 1H), 5.20~5.10 (m, 4H), 5.10~5.06 (m, 1H), 5.06~4.94 (m, 3H), 2.66 (d, J=7.3 Hz, 2H), 2.59 (td, J=10.2, 2.3 Hz, 1H), 1.60 (dt, J=10.4, 3.9 Hz, 2H), 1.32~1.18 (m, 4H), 1.14~1.07 (m, 2H), 0.84 (t, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.17, 169.91, 136.81, 135.29, 135.25, 132.91, 128.28, 128.19, 128.05, 118.40, 118.33, 66.67, 61.36, 48.56, 38.70, 31.34, 30.28, 27.28, 22.35, 13.86. HRMS (ESI-TOF) calcd for C28H33O4 [M-H]+ 433.2384, found 433.2380. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=15.28 min, tR(major)=16.07 min.
-24.52 (c 0.3, acetone); 1H NMR (400 MHz, CDCl3) δ: 7.37~7.26 (m, 10H), 5.82~5.68 (m, 1H), 5.51 (dt, J=17.0, 10.1 Hz, 1H), 5.20~5.10 (m, 4H), 5.10~5.06 (m, 1H), 5.06~4.94 (m, 3H), 2.66 (d, J=7.3 Hz, 2H), 2.59 (td, J=10.2, 2.3 Hz, 1H), 1.60 (dt, J=10.4, 3.9 Hz, 2H), 1.32~1.18 (m, 4H), 1.14~1.07 (m, 2H), 0.84 (t, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.17, 169.91, 136.81, 135.29, 135.25, 132.91, 128.28, 128.19, 128.05, 118.40, 118.33, 66.67, 61.36, 48.56, 38.70, 31.34, 30.28, 27.28, 22.35, 13.86. HRMS (ESI-TOF) calcd for C28H33O4 [M-H]+ 433.2384, found 433.2380. HPLC analysis: chiral IC (250 mm×4.6 mm, 5 µm, V(hexane)∶V(i-PrOH)=99∶1, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=15.28 min, tR(major)=16.07 min.4.4 Synthesis of 5
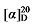 +4.32 (c 0.3, acetone; 1H NMR (400 MHz, CDCl3) δ: 7.90 (s, 3H), 5.64 (dt, J=17.1, 8.4 Hz, 1H), 5.59~5.50 (m, 0.21H), 5.36 (dt, J=9.2, 5.8 Hz, 0.28H), 5.18~5.06 (m, 2H), 3.43 (d, J=8.0 Hz, 1H), 2.80~2.68 (m, 1H), 2.60 (s, 0.34H), 1.98 (p, J=7.0 Hz, 0.60H), 1.43~1.18 (m, 10H), 0.87 (t, J=6.6 Hz, 4H); 13C NMR (101 MHz, CDCl3) 13C NMR (101 MHz, CDCl3) δ: 173.84, 137.49, 118.23, 56.89, 44.58, 32.28, 31.61, 26.82, 22.65, 14.16, 14.14. HRMS (ESI-TOF) calcd for C11H17O4 [M-H]+ 213.1132, found 213.1128. HPLC analysis: chiral IB N-5 (250 mm×4.6 mm, 5 µm, V(he-xane)∶V(i-PrOH)=80∶20, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=65.97 min, tR(major)=72.67 min).
+4.32 (c 0.3, acetone; 1H NMR (400 MHz, CDCl3) δ: 7.90 (s, 3H), 5.64 (dt, J=17.1, 8.4 Hz, 1H), 5.59~5.50 (m, 0.21H), 5.36 (dt, J=9.2, 5.8 Hz, 0.28H), 5.18~5.06 (m, 2H), 3.43 (d, J=8.0 Hz, 1H), 2.80~2.68 (m, 1H), 2.60 (s, 0.34H), 1.98 (p, J=7.0 Hz, 0.60H), 1.43~1.18 (m, 10H), 0.87 (t, J=6.6 Hz, 4H); 13C NMR (101 MHz, CDCl3) 13C NMR (101 MHz, CDCl3) δ: 173.84, 137.49, 118.23, 56.89, 44.58, 32.28, 31.61, 26.82, 22.65, 14.16, 14.14. HRMS (ESI-TOF) calcd for C11H17O4 [M-H]+ 213.1132, found 213.1128. HPLC analysis: chiral IB N-5 (250 mm×4.6 mm, 5 µm, V(he-xane)∶V(i-PrOH)=80∶20, 1.0 mL/min, 25 ℃, 210 nm), tR(minor)=65.97 min, tR(major)=72.67 min).