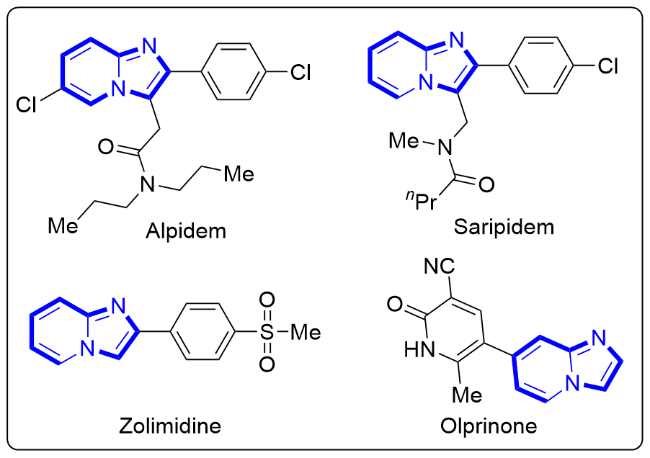
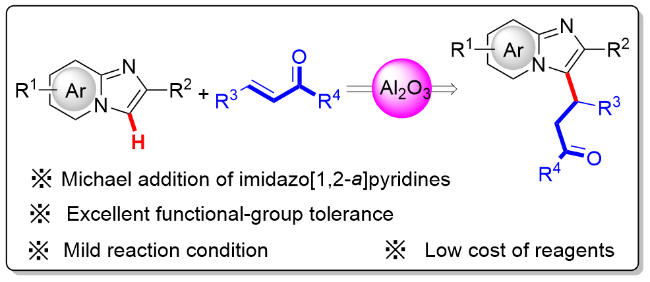
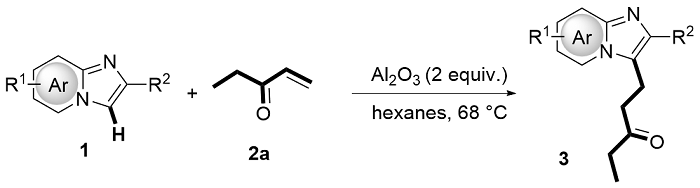
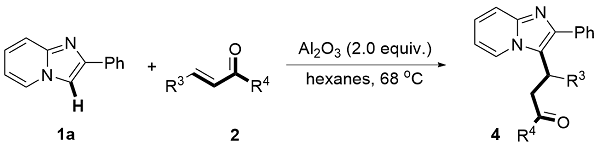
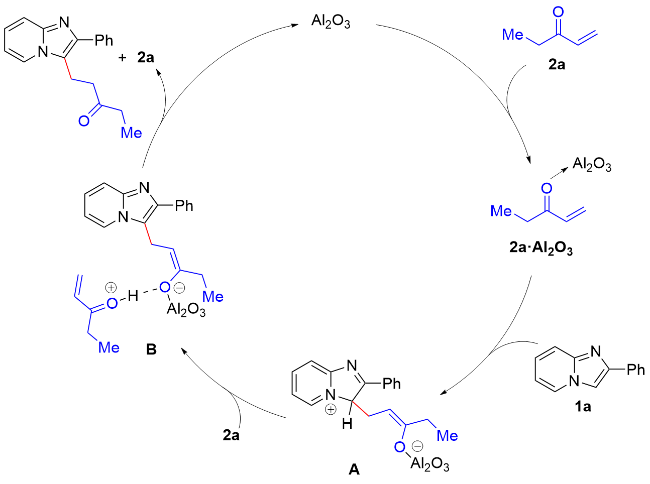
The mixture of 1 (0.2 mmol, 1.0 equiv.), 2 (0.26 mmol, 1.3 equiv.) and Al2O3 (acidic, 0.4 mmol, 2.0 equiv) was stirred in n-hexane (10 mL/mmol) in an oil bath at 68 oC in a sealed tube. After the reaction was completed (monitored by TLC), the resulting mixture was cooled and filtered through filter paper. The solids were rinsed with EtOAc, and the combined filtrate was concentrated in vacuo. The desired products 3 and 4 were obtained in the corresponding yields after purified by column chromatography on silica gel with a mixture of petroleum ether and ethyl acetate.
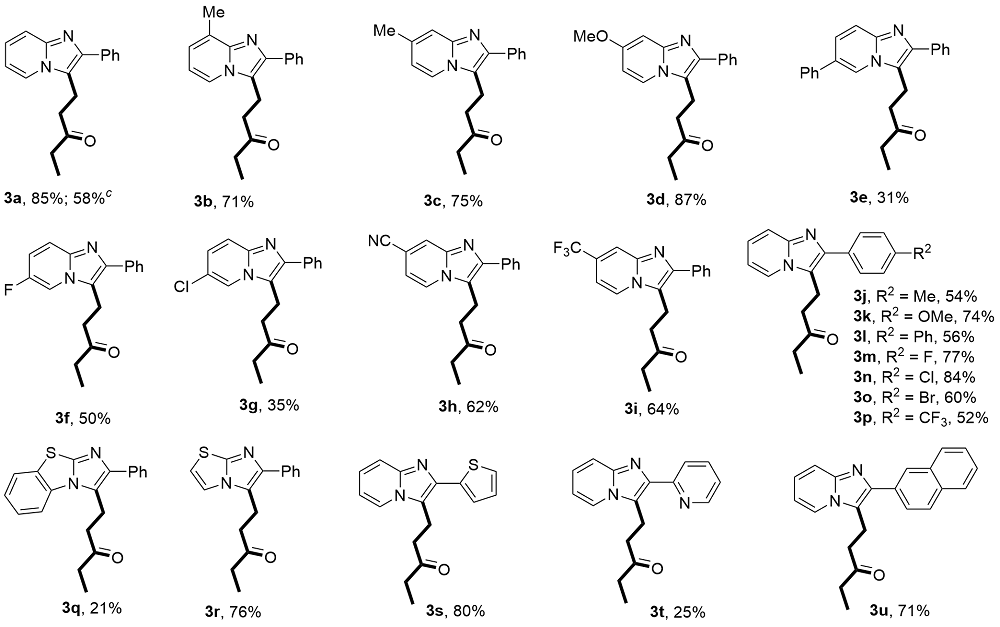
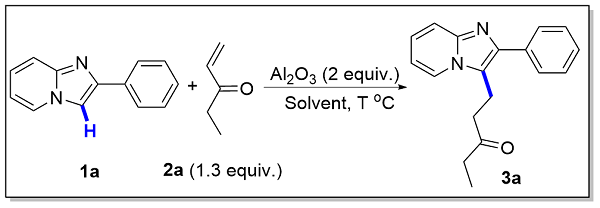
1-(2-Phenylimidazo[1,2-a]pyridin-3-yl)pentan-3-one(3a): 47.3 mg, 85% yield, yellow solid, m.p. 106.3~ 107.3 °C. 1H NMR (400 MHz, CDCl3) δ: 8.04 (d, J=6.9 Hz, 1H), 7.77 (d, J=7.0 Hz, 2H), 7.63 (d, J=9.0 Hz, 1H), 7.46 (t, J=7.6 Hz, 2H), 7.36 (t, J=7.4 Hz, 1H), 7.21~ 7.11 (m, 1H), 6.84 (t, J=6.8 Hz, 1H), 3.40 (t, J=7.6 Hz, 2H), 2.79 (t, J=7.6 Hz, 2H), 2.41 (q, J=7.3 Hz, 2H), 1.04 (t, J=7.3 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 210.0, 144.5, 142.4, 134.7, 128.6, 128.0, 127.6, 123.8, 123.1, 119.2, 117.6, 112.2, 40.0, 36.1, 17.6, 7.7. HRMS (ESI- TOF) calcd for C18H19N2O [M+H]+ 279.1492, found 279.1498.
1-(8-Methyl-2-phenylimidazo[1,2-a]pyridin-3-yl)pen-tan-3-one (3b): 41.5 mg, 71% yield, yellow solid, m.p. 113.4~114.2 °C. 1H NMR (500 MHz, CDCl3) δ: 7.88 (d, J=6.9 Hz, 1H), 7.76 (d, J=7.7 Hz, 2H), 7.45 (t, J=7.6 Hz, 2H), 7.34 (t, J=7.6 Hz, 1H), 6.96 (d, J=6.9 Hz, 1H), 6.74 (t, J=6.8 Hz, 1H), 3.35 (t, J=7.7 Hz, 2H), 2.84-2.72 (m, 2H), 2.65 (s, 3H), 2.40 (q, J=7.3 Hz, 2H), 1.03 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.1, 145.0, 142.0, 135.0, 128.6, 128.3, 127.6, 127.5, 122.7, 120.9, 119.5, 112.3, 40.1, 36.1, 17.8, 17.2, 7.7. HRMS (ESI-TOF) calcd for C19H21N2O [M+H]+ 293.1648, found 293.1656.
1-(7-methyl-2-phenylimidazo[1,2-a]pyridin-3-yl)pentan-3-one (3c): 43.8 mg, 75% yield, white solid, m.p. 89.4~ 90.5 °C. 1H NMR (500 MHz, CDCl3) δ: 7.91 (d, J=7.0 Hz, 1H), 7.79~7.69 (m, 2H), 7.44 (t, J=7.7 Hz, 2H), 7.38 (s, 1H), 7.36~7.31 (m, 1H), 6.66 (dd, J=7.0, 1.7 Hz, 1H), 3.41~3.22 (m, 2H), 2.80~2.67 (m, 3H), 2.51~2.26 (m, 5H), 1.03 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.2, 144.9, 141.9, 134.9, 134.8, 128.6, 128.0, 127.5, 122.4, 118.6, 115.9, 114.9, 40.1, 36.1, 21.3, 17.6, 7.7. HRMS (ESI-TOF) calcd for C19H21N2O [M+H]+ 293.1648, found 293.1658.
1-(7-Methoxy-2-phenylimidazo[1,2-a]pyridin-3-yl)pen-tan-3-one (3d): 53.6 mg, 87% yield, yellow solid, m.p. 126.8~128.0 °C. 1H NMR (500 MHz, CDCl3) δ: 7.86 (d, J=7.5 Hz, 1H), 7.76-7.71 (m, 2H), 7.43 (t, J=7.7 Hz, 2H), 7.32 (td, J=7.3, 1.3 Hz, 1H), 6.89 (d, J=2.6 Hz, 1H), 6.54 (dd, J=7.5, 2.5 Hz, 1H), 3.84 (s, 3H), 3.32 (t, J=7.5 Hz, 2H), 2.75 (t, J=7.5 Hz, 2H), 2.39 (q, J=7.3 Hz, 2H), 1.02 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.2, 157.5, 145.8, 141.4, 134.8, 128.6, 128.6, 127.7, 127.4, 123.7, 118.1, 107.2, 94.6, 55.5, 40.4, 36.1, 17.5, 7.7. HRMS (ESI-TOF) calcd for C19H21N2O2 [M+ H]+ 309.1598, found 309.1603.
1-(2,6-Diphenylimidazo[1,2-a]pyridin-3-yl)pentan-3-one (3e): 21.9 mg, 31% yield, brown solid, m.p. 108.5~ 109.3 °C. 1H NMR (500 MHz, CDCl3) δ: 8.25 (s, 1H), 7.85~7.78 (m, 2H), 7.77 (d, J=9.2 Hz, 1H), 7.68~7.61 (m, 2H), 7.58~7.48 (m, 5H), 7.47~7.36 (m, 2H), 3.51~3.44 (m, 2H), 2.85 (t, J=7.6 Hz, 2H), 2.44 (q, J=7.3 Hz, 2H), 1.07 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.0, 143.6, 142.5, 137.5, 134.2, 129.2, 128.8, 128.1, 128.0, 128.0, 127.1, 125.1, 120.5, 119.8, 117.3, 40.1, 36.2, 17.6, 7.7. HRMS (ESI-TOF) calcd for C24H23- N2O [M+H]+ 355.1805, found 355.1815.
1-(6-Fluoro-2-phenylimidazo[1,2-a]pyridin-3-yl)pentan-3-one (3f): 29.6 mg, 50% yield, brown solid, m.p. 140.0~ 140.6 °C. 1H NMR (500 MHz, CDCl3) δ: 8.07~ 7.99 (m, 1H), 7.76 (d, J=7.7 Hz, 2H), 7.63 (dd, J=9.8, 5.2 Hz, 1H), 7.48 (t, J=7.6 Hz, 2H), 7.42~7.34 (m, 1H), 7.17~ 7.07 (m, 1H), 3.37 (t, J=7.5 Hz, 2H), 2.80 (t, J=7.5 Hz, 2H), 2.43 (q, J=7.2 Hz, 2H), 1.06 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.8, 153.4 (d, JC—F=236 Hz), 143.7, 142.1, 134.3, 128.7, 128.0, 127.9, 120.8 (d, JC—F=2.5 Hz), 118.0 (d, JC—F=10 Hz), 116.0 (d, JC—F=25 Hz), 110.0 (d, JC—F=41.3 Hz), 39.8, 36.1, 17.6, 7.7; 19F NMR (471 MHz, CDCl3) δ: -139.6. HRMS (ESI-TOF) calcd for C18H18FN2O [M+H]+ 297.1398, found 297.1407.
1-(6-Chloro-2-phenylimidazo[1,2-a]pyridin-3-yl)pentan-3-one (3g): 21.8 mg, 35% yield, brown solid, m.p. 131.3~132.7 °C. 1H NMR (500 MHz, CDCl3) δ: 8.12 (d, J=1.9 Hz, 1H), 7.79~7.74 (m, 2H), 7.60 (dd, J=9.6, 0.8 Hz, 1H), 7.48 (t, J=7.7 Hz, 2H), 7.43~7.35 (m, 1H), 7.17 (dd, J=9.5, 2.0 Hz, 1H), 3.43~3.34 (m, 2H), 2.81 (t, J=7.6 Hz, 2H), 2.44 (q, J=7.3 Hz, 2H), 1.07 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.7, 143.4, 142.8, 134.1, 128.8, 128.0, 128.0, 125.3, 121.2, 120.6, 120.0, 117.9, 39.8, 36.1, 17.6, 7.7. HRMS (ESI-TOF) calcd for C18H18ClN2O [M+H]+ 313.1102, found 313.1109.
3-(3-Oxopentyl)-2-phenylimidazo[1,2-a]pyridine-7-car-bonitrile (3h): 37.6 mg, 62% yield, yellow solid, m.p. 137.8~139.2 °C. 1H NMR (500 MHz, CDCl3) δ: 8.02 (d, J=7.0 Hz, 1H), 7.74~7.66 (m, 2H), 7.60 (dt, J=9.1, 1.2 Hz, 1H), 7.45~7.37 (m, 2H), 7.17 (ddd, J=9.1, 6.7, 1.2 Hz, 1H), 6.83 (td, J=6.7, 1.2 Hz, 1H), 3.40~3.31 (m, 2H), 2.75 (t, J=7.6 Hz, 2H), 2.40 (q, J=7.3 Hz, 2H), 1.03 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.8, 144.5, 141.2, 133.5, 133.2, 129.2, 128.8, 124.2, 123.2, 119.4, 117.6, 112.4, 39.8, 36.1, 17.5, 7.7. HRMS (ESI- TOF) calcd for C19H18N3O [M+H]+ 304.1444, found 304.1451.
1-(2-Phenyl-7-(trifluoromethyl)imidazo[1,2-a]pyridin-3-yl)pentan-3-one (3i): 44.3 mg, 64% yield, white solid, m.p. 122.6~123.4 °C. 1H NMR (500 MHz, CDCl3) δ: 8.23 (d, J=7.1 Hz, 1H), 7.93 (s, 1H), 7.77 (d, J=8.4 Hz, 2H), 7.47 (t, J=6.9 Hz, 2H), 7.41~7.37 (m, 1H), 7.02 (d, J=7.2 Hz, 1H), 3.42 (t, J=7.4 Hz, 2H), 2.80 (t, J=7.2 Hz, 2H), 2.40 (q, J=7.4 Hz, 2H), 1.03 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.7, 143.6 (d, JC—F=266.3 Hz), 134.0, 128.8, 128.1, 128.0, 126.8, 125.6 (q, JC—F=33.8 Hz), 124.6, 123.5 (q, JC—F=362.5 Hz), 115.4 (q, JC—F=5 Hz), 108.0 (d, JC—F=3.8 Hz), 39.9, 36.1, 17.4, 7.7; 19F NMR (471 MHz, CDCl3) δ: -63.4. HRMS (ESI-TOF) calcd for C19H18F3N2O [M+H]+ 347.1366, found 347.1374.
1-(2-(p-Tolyl)imidazo[1,2-a]pyridin-3-yl)pentan-3-one (3j): 31.5 mg, 54% yield, brown solid, m.p. 127.7~ 128.8 °C. 1H NMR (500 MHz, CDCl3) δ: 8.02 (d, J=6.5 Hz, 1H), 7.71~7.58 (m, 3H), 7.26 (d, J=7.3 Hz, 2H), 7.16 (t, J=7.9 Hz, 1H), 6.82 (t, J=6.6 Hz, 1H), 3.38 (t, J=7.6 Hz, 2H), 2.77 (t, J=7.6 Hz, 2H), 2.46~2.28 (m, 5H), 1.03 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.1, 144.4, 142.4, 137.4, 131.7, 129.4, 127.9, 123.8, 123.1, 118.9, 117.5, 112.2, 40.0, 36.1, 21.3, 17.6, 7.7. HRMS (ESI-TOF) calcd for C19H21N2O [M+H]+ 293.1648, found 293.1656.
1-(2-(4-Methoxyphenyl)imidazo[1,2-a]pyridin-3-yl)pen-tan-3-one (3k): 45.6 mg, 74% yield, brown solid, m.p. 89.2~90.3 °C. 1H NMR (500 MHz, CDCl3) δ: 7.99 (d, J=6.9 Hz, 1H), 7.71~7.64 (m, 2H), 7.59 (dt, J=9.0, 1.2 Hz, 1H), 7.13 (ddd, J=9.1, 6.7, 1.2 Hz, 1H), 7.01~6.94 (m, 2H), 6.79 (td, J=6.8, 1.2 Hz, 1H), 3.83 (s, 3H), 3.34 (t, J=7.6 Hz, 2H), 2.75 (t, J=7.6 Hz, 2H), 2.39 (q, J=7.3 Hz, 2H), 1.02 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.1, 159.2, 144.4, 142.2, 129.2, 127.2, 123.7, 123.0, 118.5, 117.3, 114.1, 112.1, 55.3, 39.9, 36.1, 17.6, 7.7. HRMS (ESI-TOF) calcd for C19H21N2O2 [M+H]+ 309.1598, found 309.1606.
1-(2-([1'-Biphenyl]-4-yl)imidazo[1,2-a]pyridin-3-yl)pentan-3-one (3l): 39.6 mg, 56% yield, yellow solid, m.p. 131.5~132.7 °C. 1H NMR (500 MHz, CDCl3) δ: 8.06 (dd, J=6.9, 1.2 Hz, 1H), 7.91~7.85 (m, 2H), 7.72 (d, J=8.4 Hz, 2H), 7.69~7.64 (m, 3H), 7.47 (t, J=7.7 Hz, 2H), 7.40~7.35 (m, 1H), 7.20 (ddd, J=9.1, 6.7, 1.3 Hz, 1H), 6.85 (td, J=6.8, 1.2 Hz, 1H), 3.45 (t, J=7.6 Hz, 2H), 2.82 (t, J=7.6 Hz, 2H), 2.44 (q, J=7.3 Hz, 2H), 1.07 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.0, 144.6, 141.9, 140.7, 140.2, 133.7, 128.8, 128.4, 127.4, 127.3, 127.0, 124.0, 123.2, 119.4, 117.6, 112.3, 40.0, 36.2, 17.7, 7.7. HRMS (ESI-TOF) calcd for C24H23N2O [M+H]+ 355.1805, found 355.1814.
1-(2-(4-Fluorophenyl)imidazo[1,2-a]pyridin-3-yl)pen-tan-3-one (3m): 45.6 mg, 77% yield, yellow solid, m.p. 104.7~105.3 °C. 1H NMR (500 MHz, CDCl3) δ: 8.01 (d, J=6.9 Hz, 1H), 7.75~7.66 (m, 2H), 7.59 (dt, J=9.1, 1.2 Hz, 1H), 7.18~7.05 (m, 3H), 6.81 (td, J=6.8, 1.2 Hz, 1H), 3.34 (t, J=7.6 Hz, 2H), 2.74 (t, J=7.6 Hz, 2H), 2.39 (q, J=7.3 Hz, 2H), 1.02 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.8, 162.4 (d, JC—F=246.3 Hz), 144.5, 141.5, 130.8 (d, JC—F=3.8 Hz), 129.7 (d, JC—F=7.5 Hz), 124.0, 123.2, 119.0, 117.5, 115.6 (d, JC—F=21.3 Hz), 112.3, 39.8, 36.1, 17.5, 7.7; 19F NMR (471 MHz, CDCl3) δ: -114.5. HRMS (ESI-TOF) calcd for C18H18FN2O [M+H]+ 297.1398, found 297.1400.
1-(2-(4-Chlorophenyl)imidazo[1,2-a]pyridin-3-yl)pen-tan-3-one (3n): 52.4 mg, 84% yield, yellow solid, m.p. 137.2-138.0 °C. 1H NMR (500 MHz, CDCl3) δ: 8.04 (dd, J=7.0, 1.2 Hz, 1H), 7.71 (d, J=8.6 Hz, 1H), 7.62 (dd, J=9.0, 1.2 Hz, 1H), 7.42 (dd, J=8.5, 1.3 Hz, 2H), 7.19 (ddd, J=9.2, 6.7, 1.3 Hz, 1H), 6.85 (td, J=6.8, 1.2 Hz, 1H), 3.37 (t, J=7.6 Hz, 2H), 2.77 (t, J=7.5 Hz, 2H), 2.42 (q, J=7.3 Hz, 2H), 1.04 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.8, 144.5, 141.1, 133.5, 133.2, 129.2, 128.8, 124.2, 123.2, 119.4, 117.6, 112.4, 39.9, 36.1, 17.5, 7.7. HRMS (ESI-TOF) calcd for C18H18ClN2O [M+H]+ 313.1102, found 313.1108.
1-(2-(4-Bromophenyl)imidazo[1,2-a]pyridin-3-yl)pen-tan-3-one (3o): 42.7 mg, 60% yield, brown solid, m.p. 143.5~144.2 °C. 1H NMR (500 MHz, CDCl3) δ: 8.06 (d, J=6.9 Hz, 1H), 7.71~7.54 (m, 5H), 7.21 (ddd, J=9.1, 6.7, 1.2 Hz, 1H), 6.87 (td, J=6.8, 1.2 Hz, 1H), 3.39 (t, J=7.6 Hz, 2H), 2.78 (t, J=7.5 Hz, 2H), 2.43 (q, J=7.3 Hz, 2H), 1.06 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.8, 144.5, 141.1, 133.6, 131.8, 129.5, 124.3, 123.2, 121.8, 119.5, 117.6, 112.5, 39.9, 36.2, 17.5, 7.7. HRMS (ESI-TOF) calcd for C18H18BrN2O [M+H]+ 357.0597, found 357.0605.
1-(2-(4-(Trifluoromethyl)phenyl)imidazo[1,2-a]pyridin-3-yl)pentan-3-one (3p): 36.0 mg, 52% yield, white solid, m.p. 156.0~156.8 °C. 1H NMR (500 MHz, CDCl3) δ: 8.09 (d, J=6.6 Hz, 1H), 7.92 (d, J=8.1 Hz, 2H), 7.73 (d, J=8.1 Hz, 2H), 7.67 (d, J=9.1 Hz, 1H), 7.24 (ddd, J=9.1, 6.7, 1.2 Hz, 1H), 6.90 (t, J=6.8 Hz, 1H), 3.43 (t, J=7.5 Hz, 2H), 2.81 (t, J=7.5 Hz, 2H), 2.44 (q, J=7.3 Hz, 2H), 1.06 (t, J=7.4 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.7, 144.7, 140.7, 138.2, 129.5 (q, JC—F=32.5 Hz), 128.1, 125.6 (q, JC—F=5.0 Hz), 124.6, 123.0 (q, JC—F=388.8 Hz), 117.8, 112.7, 39.9, 36.2, 17.5, 7.7; 19F NMR (471 MHz, CDCl3) δ: -62.5. HRMS (ESI-TOF) calcd for C19H18F3N2O [M+H]+ 347.1366, found 347.1371.
1-(2-Phenylbenzo[d]imidazo[2,1-b]thiazol-3-yl)pentan-3-one (3q): 14.0 mg, 21% yield, brown solid, m.p. 87.1~87.9 °C. 1H NMR (500 MHz, CDCl3) δ: 7.77~7.60 (m, 4H), 7.47 (t, J=7.7 Hz, 3H), 7.36 (td, J=7.6, 4.6 Hz, 2H), 3.57 (t, J=7.3 Hz, 2H), 2.91 (t, J=7.2 Hz, 2H), 2.51 (q, J=7.3 Hz, 2H), 1.12 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 209.6, 146.8, 133.2, 130.6, 128.7, 127.7, 127.4, 127.4, 126.4, 126.4, 124.6, 124.6, 123.5, 112.7, 112.7, 41.5, 36.3, 19.2, 7.8. HRMS (ESI-TOF) calcd for C20H19SN2O [M+H]+ 335.1213, found 335.1223.
1-(6-Phenylimidazo[2,1-b]thiazol-5-yl)pentan-3-one (3r): 43.2 mg, 76% yield, brown liquid. 1H NMR (500 MHz, CDCl3) δ: 7.69 (d, J=6.8 Hz, 2H), 7.52 (d, J=4.5 Hz, 1H), 7.42 (t, J=7.7 Hz, 2H), 7.30 (t, J=7.6 Hz, 1H), 6.80 (d, J=4.6 Hz, 1H), 3.28 (t, J=7.0 Hz, 2H), 2.79 (t, J=7.1 Hz, 2H), 2.38 (q, J=7.3 Hz, 2H), 1.02 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.2, 147.9, 143.1, 134.9, 128.6, 127.1, 127.0, 121.4, 117.8, 112.1, 41.1, 36.1, 18.8, 7.7. HRMS (ESI-TOF) calcd for C16H17- SN2O [M+H]+ 285.1056, found 285.1060.
1-(2-(Thiophen-2-yl)imidazo[1,2-a]pyridin-3-yl)pentan-3-one (3s): 45.4 mg, 80% yield, brown solid, m.p. 88.7~ 89.3 °C. 1H NMR (500 MHz, CDCl3) δ: 8.07 (d, J=6.9 Hz, 1H), 7.57 (d, J=9.1 Hz, 1H), 7.43 (d, J=3.7 Hz, 1H), 7.33 (d, J=5.1 Hz, 1H), 7.20-7.07 (m, 2H), 6.81 (t, J=6.8 Hz, 1H), 3.40 (t, J=7.4 Hz, 2H), 2.81 (t, J=7.4 Hz, 2H), 2.41 (q, J=7.3 Hz, 2H), 1.02 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.1, 144.4, 137.7, 137.0, 127.8, 125.2, 124.2, 124.1, 123.3, 118.7, 117.2, 112.3, 39.7, 36.1, 17.5, 7.7. HRMS (ESI-TOF) calcd for C16H17SN2O [M+H]+ 285.1056, found 285.1062.
1-(2-(Pyridin-2-yl)imidazo[1,2-a]pyridin-3-yl)pentan-3-one (3t): 14.0 mg, 25% yield, brown liquid. 1H NMR (500 MHz, CDCl3) δ: 8.63 (ddd, J=4.8, 1.9, 1.0 Hz, 1H), 8.27 (dt, J=8.1, 1.1 Hz, 1H), 8.21 (d, J=6.9 Hz, 1H), 7.77 (td, J=7.7, 1.9 Hz, 1H), 7.62 (dt, J=9.1, 1.2 Hz, 1H), 7.25-7.14 (m, 2H), 6.86 (td, J=6.8, 1.2 Hz, 1H), 3.69 (t, J=7.3 Hz, 2H), 2.97 (t, J=7.2 Hz, 2H), 2.45 (q, J=7.3 Hz, 2H), 1.04 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 211.5, 154.5, 149.0, 144.2, 140.2, 136.4, 124.3, 123.7, 123.1, 121.7, 121.7, 117.6, 112.3, 40.6, 36.0, 18.4, 7.8. HRMS (ESI-TOF) calcd for C17H18N3O [M+H]+ 280.1444, found 280.1452.
1-(2-(Naphthalen-2-yl)imidazo[1,2-a]pyridin-3-yl)pen-tan-3-one (3u): 46.6 mg, 71% yield, brown solid, m.p. 79.5~79.9 °C. 1H NMR (500 MHz, CDCl3) δ: 8.24 (s, 1H), 8.06 (d, J=7.2 Hz, 1H), 7.96~7.88 (m, 3H), 7.89~ 7.83 (m, 1H), 7.68 (d, J=9.0 Hz, 1H), 7.55~7.42 (m, 2H), 7.19 (ddd, J=9.1, 6.7, 1.2 Hz, 1H), 6.84 (td, J=6.8, 1.2 Hz, 1H), 3.46 (t, J=7.5 Hz, 2H), 2.79 (t, J=7.6 Hz, 2H), 2.39 (q, J=7.3 Hz, 2H), 1.03 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 210.0, 144.6, 142.2, 133.5, 132.8, 132.1, 128.3, 128.3, 127.7, 126.9, 126.3, 126.1, 126.0, 124.1, 123.2, 119.7, 117.6, 112.3, 40.0, 36.1, 17.7, 7.7. HRMS (ESI-TOF) calcd for C22H21N2O [M+H]+ 329.1648, found 329.1646.
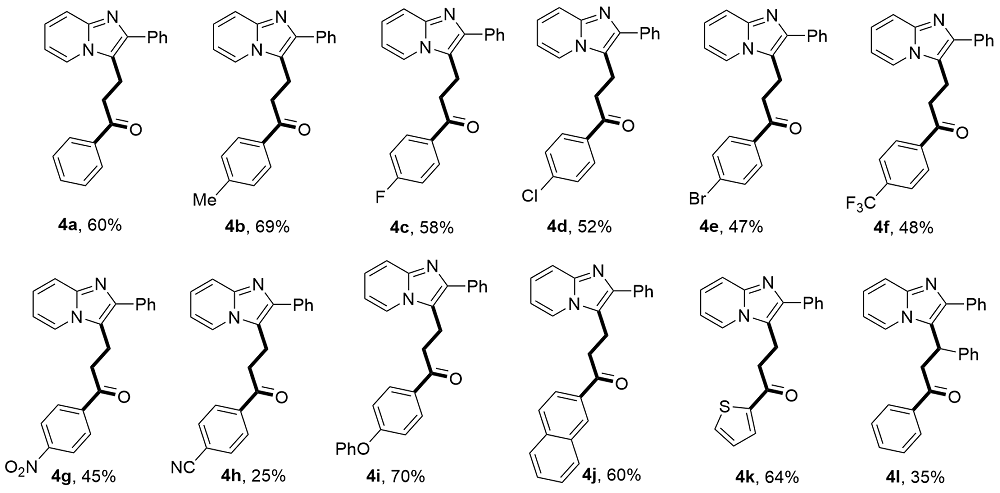
1-Phenyl-3-(2-phenylimidazo[1,2-a]pyridin-3-yl)pro-pan-1-one (4a): 39.1 mg, 60% yield, yellow solid, m.p. 167.3~168.1 °C. 1H NMR (500 MHz, CDCl3) δ: 8.09 (dd, J=6.9, 1.2 Hz, 1H), 7.92 (dd, J=8.4, 1.4 Hz, 2H), 7.82 (dd, J=8.2, 1.4 Hz, 2H), 7.68 (dd, J=9.1, 1.2 Hz, 1H), 7.61-7.54 (m, 1H), 7.46 (dt, J=15.5, 7.7 Hz, 4H), 7.38 (td, J=7.2, 1.3 Hz, 1H), 7.21 (ddd, J=9.1, 6.7, 1.3 Hz, 1H), 6.87 (td, J=6.8, 1.2 Hz, 1H), 3.62-3.55 (m, 2H), 3.40-3.34 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 198.7, 144.6, 142.5, 136.3, 134.6, 133.5, 128.7, 128.7, 128.1, 128.0, 127.7, 124.0, 123.1, 119.2, 117.7, 112.4, 36.5, 18.0. HR- MS (ESI-TOF) calcd for C22H19N2O [M+H]+ 327.1492, found 327.1497.
3-(2-Phenylimidazo[1,2-a]pyridin-3-yl)-1-(p-tolyl)pro-pan-1-one (4b): 46.9 mg, 69% yield, yellow solid, m.p. 82.1~82.9 °C. 1H NMR (500 MHz, CDCl3) δ: 8.09 (d, J=6.8 Hz, 1H), 7.85~7.78 (m, 4H), 7.67 (d, J=9.0 Hz, 1H), 7.47 (t, J=7.8 Hz, 2H), 7.37 (t, J=7.4 Hz, 1H), 7.24 (d, J=7.9 Hz, 2H), 7.23~7.16 (m, 1H), 6.86 (t, J=6.8 Hz, 1H), 3.58 (t, J=7.7 Hz, 2H), 3.33 (t, J=7.7 Hz, 2H), 2.40 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.3, 144.6, 144.4, 142.5, 134.6, 133.9, 129.4, 128.7, 128.2, 128.1, 127.7, 124.0, 123.2, 119.3, 117.7, 112.4, 36.3, 21.7, 18.1. HRMS (ESI-TOF) calcd for C23H21N2O [M+H]+ 341.1648, found 341.1655.
1-(4-Fluorophenyl)-3-(2-phenylimidazo[1,2-a]pyridin-3-yl)propan-1-one (4c): 39.9 mg, 58% yield, yellow solid, m.p. 171.7~172.6 °C. 1H NMR (500 MHz, CDCl3) δ: 8.09 (d, J=6.9 Hz, 1H), 7.97~7.88 (m, 2H), 7.81 (d, J=7.1 Hz, 2H), 7.68 (d, J=9.0 Hz, 1H), 7.48 (t, J=7.7 Hz, 2H), 7.38 (t, J=7.4 Hz, 1H), 7.26~7.17 (m, 1H), 7.11 (t, J=8.6 Hz, 2H), 6.88 (t, J=6.8 Hz, 1H), 3.59 (t, J=7.6 Hz, 2H), 3.33 (t, J=7.7 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 197.0, 165.9 (d, JC—F=253.8 Hz), 144.6, 142.5, 134.5, 132.8 (d, JC—F=2.5 Hz), 130.7 (d, JC—F=8.8 Hz), 128.7, 128.2, 128.1, 127.7, 123.6 (d, JC—F=120.0 Hz), 119.1, 117.7, 115.8 (d, JC—F=22.5 Hz), 112.4, 36.4, 18.0; 19F NMR (471 MHz, CDCl3) δ: -104.4. HRMS (ESI-TOF) calcd for C22H18FN2O [M+H]+ 345.1398, found 345.1406.
1-(4-Chlorophenyl)-3-(2-phenylimidazo[1,2-a]pyridin-3-yl)propan-1-one (4d): 37.4 mg, 52% yield, yellow solid, m.p. 173.2~174.3 °C. 1H NMR (500 MHz, CDCl3) δ: 8.08 (d, J=6.9 Hz, 1H), 7.82 (dd, J=13.8, 7.9 Hz, 4H), 7.68 (d, J=9.1 Hz, 1H), 7.48 (t, J=7.6 Hz, 2H), 7.40 (dd, J=11.7, 7.8 Hz, 3H), 7.22 (t, J=7.8 Hz, 1H), 6.88 (t, J=6.9 Hz, 1H), 3.59 (t, J=7.7 Hz, 2H), 3.32 (t, J=7.6 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 197.4, 144.6, 142.5, 140.0, 134.6, 129.4, 129.0, 128.7, 128.1, 127.8, 124.1, 124.1, 123.1, 119.0, 117.7, 112.5, 112.5, 36.5, 18.0. HRMS (ESI- TOF) calcd for C22H18ClN2O [M+H]+ 361.1102, found 361.1105.
1-(4-Bromophenyl)-3-(2-phenylimidazo[1,2-a]pyridin-3-yl)propan-1-one (4e): 38.0 mg, 47% yield, yellow solid, m.p. 177.9~178.6 °C. 1H NMR (500 MHz, CDCl3) δ: 8.09 (d, J=6.9 Hz, 1H), 7.80 (d, J=7.1 Hz, 2H), 7.75 (d, J=8.4 Hz, 2H), 7.69 (d, J=9.0 Hz, 1H), 7.58 (d, J=8.6 Hz, 2H), 7.48 (t, J=7.6 Hz, 2H), 7.38 (t, J=7.5 Hz, 1H), 7.25~7.18 (m, 1H), 6.88 (t, J=6.8 Hz, 1H), 3.59 (t, J=7.7 Hz, 2H), 3.31 (t, J=7.6 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 197.6, 144.6, 142.5, 135.0, 134.5, 132.0, 129.5, 128.7, 128.2, 128.1, 127.8, 124.1, 123.1, 119.0, 117.7, 112.5, 36.5, 18.0. HRMS (ESI-TOF) calcd for C22H18- BrN2O [M+H]+ 405.0597, found 405.0604.
3-(2-Phenylimidazo[1,2-a]pyridin-3-yl)-1-(4-(trifluoro-methyl)phenyl)propan-1-one (4f): 37.8 mg, 48% yield, yellow solid, m.p. 173.8~174.9 °C. 1H NMR (500 MHz, CDCl3) δ: 8.09 (d, J=7.0 Hz, 1H), 7.99 (d, J=8.1 Hz, 2H), 7.86~7.76 (m, 1H), 7.70 (t, J=8.6 Hz, 3H), 7.48 (t, J=7.7 Hz, 2H), 7.42~7.35 (m, 1H), 7.23 (ddd, J=9.1, 6.7, 1.3 Hz, 1H), 6.89 (td, J=6.8, 1.2 Hz, 1H), 3.62 (t, J=7.7 Hz, 2H), 3.37 (t, J=7.6 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 197.7, 144.6, 142.6, 138.9, 135.1, 134.6 (q, JC—F=66.3 Hz), 128.3 (q, JC—F=115.0 Hz), 125.8 (q, JC—F=3.8 Hz), 124.6, 123.6 (d, JC—F=136 Hz), 122.4, 118.8, 117.7, 112.5, 36.8, 17.9; 19F NMR (471 MHz, CDCl3) δ: -63.2. HRMS (ESI-TOF) calcd for C23H18F3- N2O [M+H]+ 395.1366, found 395.1373.
1-(4-Nitrophenyl)-3-(2-phenylimidazo[1,2-a]pyridin-3-yl)propan-1-one (4g): 33.4 mg, 45% yield, yellow solid, m.p. 189.9~190.9 °C. 1H NMR (500 MHz, CDCl3) δ: 8.28 (d, J=8.8 Hz, 2H), 8.13 (d, J=6.9 Hz, 1H), 8.03 (d, J=8.9 Hz, 2H), 7.85~7.77 (m, 2H), 7.73 (d, J=9.0 Hz, 1H), 7.49 (t, J=7.7 Hz, 2H), 7.43~7.35 (m, 1H), 7.28~7.22 (m, 1H), 6.93 (td, J=6.8, 1.2 Hz, 1H), 3.64 (t, J=7.7 Hz, 2H), 3.41 (t, J=7.6 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 197.1, 150.5, 144.4, 142.3, 140.6, 134.1, 129.1, 128.8, 128.1, 128.0, 124.5, 123.9, 123.1, 118.6, 117.7, 112.8, 37.1, 17.8. HRMS (ESI-TOF) calcd for C22H18N3O3 [M+H]+ 372.1343, found 372.1349.
4-(3-(2-Phenylimidazo[1,2-a]pyridin-3-yl)propanoyl)-benzonitrile (4h): 17.5 mg, 25% yield, red solid, m.p. 205.8~206.5 °C. 1H NMR (500 MHz, CDCl3) δ: 8.09 (d, J=6.9 Hz, 1H), 7.97 (d, J=8.4 Hz, 2H), 7.85~7.78 (m, 2H), 7.74 (d, J=8.5 Hz, 2H), 7.69 (dt, J=9.0, 1.2 Hz, 1H), 7.48 (t, J=7.7 Hz, 2H), 7.43~7.36 (m, 1H), 7.23 (ddd, J=9.1, 6.7, 1.2 Hz, 1H), 6.90 (td, J=6.8, 1.2 Hz, 1H), 3.62 (t, J=7.6 Hz, 2H), 3.36 (t, J=7.6 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 197.4, 144.6, 142.7, 139.1, 134.5, 132.6, 128.8, 128.4, 128.1, 127.8, 124.1, 123.0, 118.6, 117.8, 117.8, 116.7, 112.5, 36.9, 17.9. HRMS (ESI- TOF) calcd for C23H18N3O [M+H]+ 352.1444, found 352.1454.
1-(4-Phenoxyphenyl)-3-(2-phenylimidazo[1,2-a]pyridin-3-yl)propan-1-one (4i): 58.5 mg, 70% yield, brown liquid. 1H NMR (500 MHz, CDCl3) δ: 8.11 (dt, J=6.9, 1.2 Hz, 1H), 7.92-7.85 (m, 2H), 7.82 (dd, J=8.2, 1.4 Hz, 2H), 7.69 (dt, J=9.0, 1.1 Hz, 1H), 7.48 (dd, J=8.4, 7.0 Hz, 2H), 7.40 (dt, J=9.2, 7.3 Hz, 3H), 7.22 (ddd, J=8.9, 6.5, 1.2 Hz, 2H), 7.12-7.03 (m, 2H), 7.01-6.94 (m, 2H), 6.88 (td, J=6.8, 1.2 Hz, 1H), 3.59 (t, J=7.6 Hz, 2H), 3.32 (t, J=7.6 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 197.2, 162.3, 155.3, 144.5, 142.3, 134.5, 131.0, 130.3, 130.1, 128.7, 128.1, 127.7, 124.8, 124.1, 123.2, 120.2, 119.3, 117.6, 117.4, 112.5, 36.3, 18.1. HRMS (ESI-TOF) calcd for C28H23N2O2 [M+H]+ 419.1754, found 419.1756.
1-(Naphthalen-2-yl)-3-(2-phenylimidazo[1,2-a]pyridin-3-yl)propan-1-one (4j): 45.1 mg, 60% yield, yellow solid, m.p. 153.8~154.6 °C. 1H NMR (500 MHz, CDCl3) δ: 8.40 (d, J=1.7 Hz, 1H), 8.15 (dt, J=6.9, 1.2 Hz, 1H), 8.02 (dd, J=8.7, 1.8 Hz, 1H), 7.95~7.82 (m, 6H), 7.72 (dd, J=9.1, 1.2 Hz, 1H), 7.62 (ddd, J=8.2, 6.8, 1.3 Hz, 1H), 7.56 (ddd, J=8.1, 6.9, 1.3 Hz, 1H), 7.50 (t, J=7.7 Hz, 2H), 7.43~ 7.36 (m, 1H), 7.24 (ddd, J=9.0, 6.7, 1.2 Hz, 1H), 6.91 (td, J=6.8, 1.1 Hz, 1H), 3.67 (t, J=7.6 Hz, 2H), 3.52 (t, J=7.6 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 198.6, 144.5, 142.4, 135.7, 134.5, 133.7, 132.4, 129.9, 129.6, 128.8, 128.7, 128.6, 128.2, 127.8, 127.8, 126.9, 124.2, 123.6, 123.2, 119.3, 117.7, 112.5, 36.6, 18.2. HRMS (ESI-TOF) calcd for C26H21N2O [M+H]+ 377.1648, found 377.1656.
3-(2-Phenylimidazo[1,2-a]pyridin-3-yl)-1-(thiophen-2-yl)propan-1-one (4k): 42.5 mg, 64% yield, orange solid, m.p. 125.0~125.7 °C. 1H NMR (500 MHz, Chloroform-d) δ: 8.10 (dd, J=6.3, 1.9 Hz, 1H), 7.87~7.75 (m, 2H), 7.69~7.64 (m, 2H), 7.62~7.58 (m, 1H), 7.48 (td, J=7.6, 1.7 Hz, 2H), 7.42~7.34 (m, 1H), 7.20 (dddd, J=9.6, 6.7, 2.9, 1.4 Hz, 1H), 7.09 (dtd, J=5.1, 3.5, 1.9 Hz, 1H), 6.87 (ddt, J=8.2, 6.9, 1.5 Hz, 1H), 3.58 (t, J=7.6 Hz, 2H), 3.28 (t, J=7.6 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 191.6, 144.6, 143.5, 142.6, 134.5, 134.2, 132.2, 128.7, 128.3, 128.1, 127.7, 124.1, 123.2, 119.0, 117.6, 112.4, 37.2, 18.3. HRMS (ESI-TOF) calcd for C20H17SN2O [M+H]+ 333.1056, found 333.1066.
(R)-1,3-Diphenyl-3-(2-phenylimidazo[2-a]pyridin-3-yl)propan-1-one (4l): 28.1 mg, 35% yield, white solid, m.p. 156.9~157.9 °C. 1H NMR (500 MHz, CDCl3) δ: 7.85 (d, J=6.9 Hz, 1H), 7.70 (d, J=9.1 Hz, 1H), 7.66 (d, J=7.8 Hz, 2H), 7.63~7.59 (m, 2H), 7.51 (t, J=7.4 Hz, 1H), 7.38~7.31 (m, 7H), 7.30~7.24 (m, 3H), 7.25~7.19 (m, 1H), 6.76 (t, J=6.8 Hz, 1H), 5.58 (t, J=7.0 Hz, 1H), 3.86~3.69 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 197.2, 144.5, 143.8, 140.5, 136.2, 134.8, 133.2, 129.5, 129.1, 128.5, 128.3, 128.0, 127.9, 127.2, 127.1, 124.5, 123.9, 121.0, 117.7, 112.4, 42.3, 35.6. HRMS (ESI-TOF) calcd for C28H23N2O [M+H]+ 403.1805, found 403.1807.
Supporting Information 1H NMR and
13C NMR spectra of compounds
3~
4. The Supporting Information is available free of charge via the Internet at
http://sioc-journal.cn/.