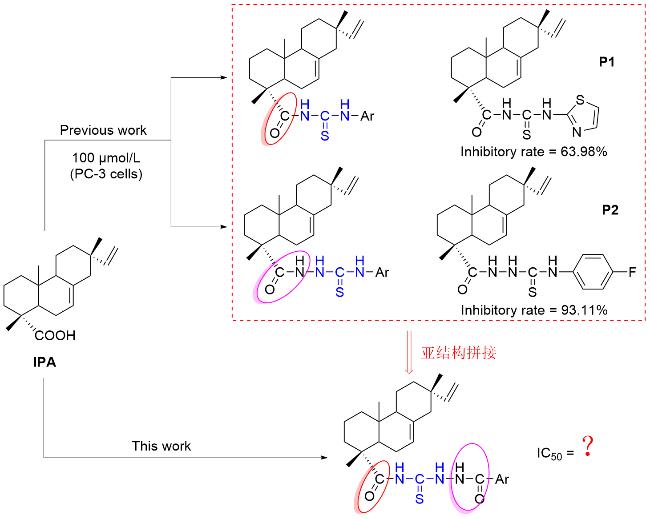
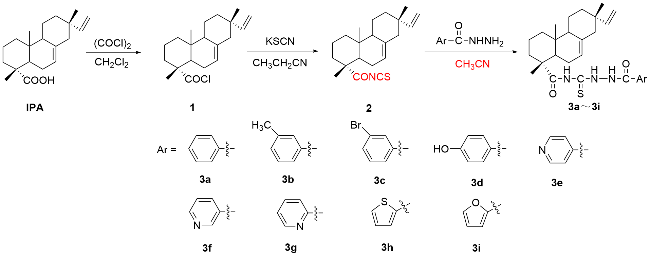
将3.0 mmol芳基酰肼混合于10 mL丙酮中, 滴加上述合成的异海松酰基异硫氰酸酯(2). 滴毕, 50 ℃回流反应, 溶液中有沉淀产生, 薄层色谱(TLC)监测反应进程, 至原料点消失, 结束反应. 反应液冷却至室温, 过滤沉淀, 丙酮洗涤得到化合物3.
异海松酰基-N'-苯甲酰氨基硫脲(3a): 白色固体, 产率85.4%. m.p. 132.2~133.6 ℃; 1H NMR (500 MHz, CDCl3) δ: 13.56 (s, 1H, CONH), 9.95 (s, 1H, CONH), 8.56 (s, 1H, CSNH), 7.91 (d, J=25 Hz, 2H, ArH), 7.63 (t, J=7.5 Hz, 1H, ArH), 7.53 (t, J=8 Hz, 2H, ArH), 5.87 (dd, J=10.5, 17.5 Hz, 1H, C=CH), 5.34 (brd, 1H, C=CH), 4.99 (dd, J=17.5, 32.5 Hz, 2H, C=CH2), 2.21 (s, 1H), 2.08~1.89 (m, 6H), 1.85~1.80 (m, 2H), 1.71~1.60 (m, 7H), 1.42 (s, 3H, Me), 0.98 (s, 3H, Me), 0.91 (s, 3H, Me); 13C NMR (125 MHz, CDCl3) δ: 179.6, 170.5, 162.6, 150.1, 136.0, 132.8, 130.7, 128.9 (2C), 127.2 (2C), 120.41, 109.4, 52.0, 47.7, 45.9, 45.7, 38.5, 37.1, 36.7, 35.9, 35.3, 25.0, 21.5, 20.1, 17.9, 17.1, 15.4; HRMS calcd for C28H36N3- O2S [M-H]+ 478.2514, found 478.2512.
N-异海松酰基-N'-(3-甲基苯甲酰氨基)硫脲(3b): 亮灰色固体, 产率80.7%. m.p. 115.7~117.3 ℃; 1H NMR (500 MHz, CDCl3) δ: 13.57 (s, 1H, CONH), 9.91 (s, 1H, CONH), 8.55 (s, 1H, CSNH), 7.71~7.68 (m, 2H, ArH), 7.43~7.40 (m, 2H, ArH), 5.88 (dd, J=11, 17.5 Hz, 1H, C=CH), 5.35 (brd, 1H, C=CH), 4.99 (dd, J=17.5, 32.5 Hz, 2H, C=CH2), 2.47 (s, 3H, Me), 2.09~1.89 (m, 6H), 1.86~1.81 (m, 2H), 1.72~1.53 (m, 8H), 1.43 (s, 3H, Me), 0.98 (s, 3H, Me), 0.91 (s, 3H, Me); 13C NMR (125 MHz, CDCl3) δ: 179.5, 170.3, 162.4, 150.1, 138.9, 135.9, 133.5, 130.7, 128.8, 127.8, 124.2, 120.3, 109.4, 52.0, 47.7, 45.9, 45.7, 44.9, 38.5, 37.1, 36.7, 36.0, 35.2, 25.0, 21.5, 20.0, 17.9, 17.1, 15.4; HRMS calcd for C29H40N3O2S [M+H]+ 494.2836, found 494.2842.
N-异海松酰基-N'-(3-溴苯甲酰氨基)硫脲(3c): 淡黄色固体, 产率72.8%. m.p. 143.9~145.3 ℃; 1H NMR (500 MHz, CDCl3) δ: 13.54 (s, 1H, CONH), 9.93 (s, 1H, CONH), 8.56 (s, 1H, CSNH), 8.05 (s, 1H, ArH), 7.82 (d, J=7.5 Hz, 1H, ArH), 7.77 (d, J=7.5 Hz, 1H, ArH), 7.42 (t, J=8 Hz, 1H, ArH), 5.88 (dd, J=10.5, 17.5 Hz, 1H, C=CH), 5.35 (brd, 1H, C=CH), 5.00 (dd, J=17.5, 32.5 Hz, 2H, C=CH2), 2.09~1.91 (m, 6H), 1.86~1.80 (m, 2H), 1.72~1.58 (m, 7H), 1.55~1.53 (m, 1H), 1.43 (s, 3H, Me), 0.99 (s, 3H, Me), 0.91 (s, 3H, Me); 13C NMR (125 MHz, CDCl3) δ: 179.6, 170.9, 160.8, 150.1, 136.0, 135.7, 132.7, 130.5, 130.4, 125.6, 123.1, 120.4, 109.4, 52.0, 47.7, 45.9, 45.7, 38.4, 37.1, 36.7, 35.9, 35.2, 25.0, 21.5, 20.0, 17.9, 17.1, 15.3; HRMS calcd for C28H37BrN3O2S [M+ H]+ 560.1767, found 560.1769.
N-异海松酰基-N'-(4-羟基苯甲酰氨基)硫脲(3d): 白色固体, 产率57.5%. m.p. 110.6~112.2 ℃; 1H NMR (500 MHz, CDCl3) δ: 13.55 (s, 1H, NH), 10.81 (s, 1H, NH), 9.85 (s, 1H, OH), 8.56 (s, 1H, NH), 7.80 (d, J=8.5 Hz, 2H, ArH), 6.99 (d, J=13.5 Hz, 2H, ArH), 5.87 (dd, J=11, 17.5 Hz, 1H, C=CH), 5.33 (brd, 1H, C=CH), 4.99 (dd, J=17.5, 32.5 Hz, 2H, C=CH2), 2.09 (s, 1H), 2.03~1.89 (m, 7H), 1.83~1.79 (m, 2H), 1.70~1.60 (m, 6H), 1.42 (s, 3H, Me), 0.97 (s, 3H, Me), 0.90 (s, 3H, Me); 13C NMR (125 MHz, CDCl3) δ: 179.5, 170.3, 162.3, 160.5, 150.1, 135.9, 129.4 (2C), 122.4, 120.3, 115.9 (2C), 109.3, 51.9, 47.7, 45.9, 45.7, 38.4, 37.1, 36.7, 35.9, 35.2, 25.0, 21.5, 19.9, 17.9, 17.1, 15.3; HRMS calcd for C28H38N3O3S [M+H]+ 496.2628, found 496.2631.
N-异海松酰基-N'-(4-吡啶甲酰氨基)硫脲(3e): 黄色固体, 产率80.1%. m.p. 96.3~155.8 ℃; 1H NMR (500 MHz, CDCl3) δ: 13.56 (s, 1H, CONH), 10.10 (s, 1H, CONH), 8.87 (d, J=7.5 Hz, 2H, ArH), 8.60 (s, 1H, CSNH), 7.74 (d, J=7.5 Hz, 2H, ArH), 5.88 (dd, J=11, 17.5 Hz, 1H, C=CH), 5.35 (brd, 1H, C=CH), 5.00 (dd, J=17.5, 32.5 Hz, 2H, C=CH2), 2.00~1.91 (m, 6H), 1.86~1.79 (m, 2H), 1.73~1.61 (m, 6H), 1.56~1.47 (m, 2H), 1.44 (s, 3H, Me), 0.99 (s, 3H, Me), 0.91 (s, 3H, Me); 13C NMR (125 MHz, CDCl3) δ: 179.8, 171.5, 160.3, 150.9 (2C), 150.1, 137.9, 136.1, 120.7 (2C), 120.2, 109.4, 52.0, 47.8, 46.0, 45.7, 38.4, 37.2, 36.7, 35.9, 35.3, 25.0, 21.5, 20.0, 17.9, 17.1, 15.3; HRMS calcd for C27H37N4O2S [M+H]+ 481.2722, found 481.2720.
N-异海松酰基-N'-(3-吡啶甲酰氨基)硫脲(3f): 白色固体, 产率63.7%. m.p. 102.2~103.8 ℃; 1H NMR (500 MHz, CDCl3) δ: 13.52 (s, 1H, CONH), 10.06 (s, 1H, CONH), 9.12 (s, 1H), 8.84 (d, J=7.0 Hz, 1H, ArH), 8.61 (s, 1H, CSNH), 8.21 (d, J=8.0 Hz, 1H, ArH), 7.49~7.47 (m, 1H), 5.87 (dd, J=11, 17.5 Hz, 1H, C=CH), 5.34 (brd, 1H, C=CH), 5.00 (dd, J=17.5, 32.5 Hz, 2H, C=CH2), 2.20 (s, 1H), 2.08~1.90 (m, 6H), 1.85~1.79 (m, 2H), 1.71~1.52 (m, 7H), 1.43 (s, 3H, Me), 0.98 (s, 3H, Me), 0.90 (s, 3H, Me); 13C NMR (125 MHz, CDCl3) δ: 179.7, 171.5, 160.6, 153.3, 150.1, 148.2, 136.0, 135.1, 126.9, 123.7, 120.2, 109.4, 52.0, 47.7, 45.9, 45.7, 38.4, 37.1, 36.7, 35.9, 35.2, 25.0, 21.5, 20.0, 17.9, 17.1, 15.3; HRMS calcd for C27H37N4O2S [M+H]+ 481.2602, found 481.2609.
N-异海松酰基-N'-(2-吡啶甲酰氨基)硫脲(3g): 白色固体, 产率67.8%. m.p. 134.7~155.8 ℃; 1H NMR (500 MHz, CDCl3) δ: 11.25 (brd, 1H, CONH), 10.73 (s, 1H, CONH), 9.02 (s, 1H, CSNH), 8.68 (d, J=4.5 Hz, 1H, ArH), 8.20 (t, J=7.5 Hz, 1H, ArH), 7.93~7.89 (m, 1H, ArH), 7.54~7.48 (m, 1H, ArH), 5.87 (dd, J=10.5, 17.5 Hz, 1H, C=CH), 5.34 (brd, 1H, C=CH), 4.99 (dd, J=18.0, 32.5 Hz, 2H, C=CH2), 2.21 (s, 1H), 2.09 (s, 1H), 1.96~1.92 (m, 4H), 1.86~1.81 (m, 3H), 1.71~1.54 (m, 7H), 1.43 (s, 3H, Me), 0.98 (s, 3H, Me), 0.90 (s, 3H, Me); 13C NMR (125 MHz, CDCl3) δ: 179.5, 171.1, 159.1, 150.1, 148.8, 148.3, 137.4, 135.9, 127.0, 122.6, 120.3, 109.3, 52.0, 47.7, 45.9, 45.7, 38.4, 37.1, 36.7, 35.9, 35.2, 25.0, 21.5, 20.0, 17.9, 17.1, 15.3; HRMS calcd for C27H37N4O2S [M+H]+ 481.2742, found 481.2741.
N-异海松酰基-N'-(2-噻吩甲酰氨基)硫脲(3h): 灰色固体, 产率53.6%. m.p. 110.9~112.3 ℃; 1H NMR (500 MHz, CDCl3) δ: 13.43 (s, 1H, NH), 9.76 (s, 1H, CONH), 8.64 (s, 1H, CSNH), 7.72 (d, J=5 Hz, 1H, ArH), 7.64 (d, J=5 Hz, 1H, ArH), 7.19 (t, J=5 Hz, 1H, ArH), 5.88 (dd, J=10.0, 20.0 Hz, 1H, C=CH), 5.32~5.23 (brd, 1H, C=CH), 4.98 (dd, J=17.5, 33 Hz, 2H, C=CH2), 2.09~1.89 (m, 7H), 1.85~1.79 (m, 2H), 1.72~1.50 (m, 7H), 1.42 (s, 3H, Me), 0.98 (s, 3H, Me), 0.91 (s, 3H, Me); 13C NMR (125 MHz, CDCl3) δ: 179.5, 170.9, 157.3, 150.1, 135.9, 134.4, 131.6, 129.7, 128.0, 120.3, 109.4 (2C), 52.0, 47.7, 45.9, 45.7, 38.4, 37.1, 36.7, 35.9, 35.2, 25.0, 21.5, 20.0, 17.9, 17.1, 15.3; HRMS (ESI) calcd for C26H36N3O2S2 [M+H]+ 486.2248, found 486.2252.
N-异海松酰基-N'-(2-呋喃甲酰氨基)硫脲(3i): 淡黄色固体, 产率72.3%. m.p. 123.6~124.9 ℃; 1H NMR (500 MHz, CDCl3) δ: 13.44 (s, 1H, NH), 9.95 (s, 1H, CONH), 8.56 (s, 1H, CSNH), 7.58 (s, 1H, ArH), 7.30~7.29 (m, 1H, ArH), 6.60~6.59 (m, 1H, ArH), 5.87 (dd, J=10.5, 17.5 Hz, 1H, C=CH), 5.34 (brd, 1H, C=CH), 4.99 (dd, J=17.5, 32.5 Hz, 2H, C=CH2), 2.08~1.89 (m, 6H), 1.84~1.79 (m, 3H), 1.71~1.60 (m, 7H), 1.42 (s, 3H, Me), 0.97 (s, 3H, 17-Me), 0.90 (s, 3H, 20-Me); 13C NMR (125 MHz, CDCl3) δ: 179.5, 171.0, 153.1, 150.1, 145.2, 135.9, 120.3, 116.5, 112.4, 109.4 (2C), 52.1, 47.7, 45.9, 45.7, 38.5, 37.1, 36.7, 36.0, 35.3, 25.0, 21.5, 19.9, 17.9, 17.1, 15.3; HRMS calcd for C26H36N3O3S [M+H]+ 470.2472, found 470.2479.