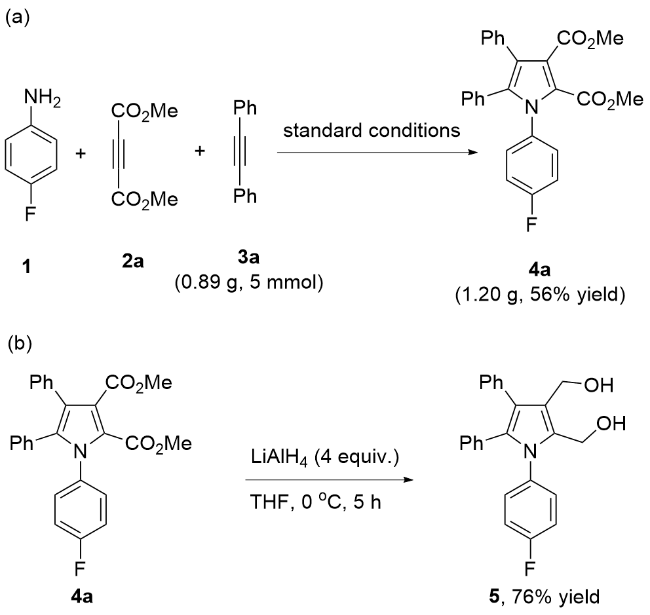
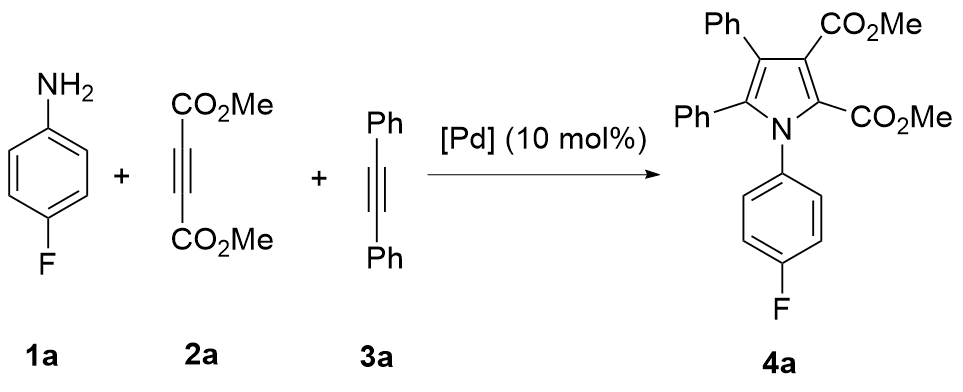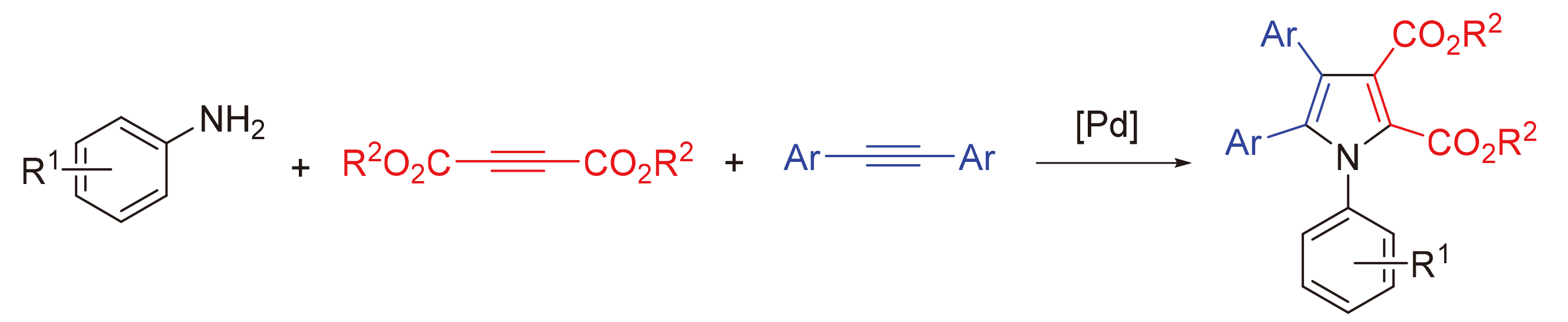A mixture of Pd(TFA)₂ (6.6 mg, 0.02 mmol), K2S2O8 (108.1 mg, 0.4 mmol), 4-fluoroaniline (44.4 mg, 0.4 mmol), dimethyl acetylenedicarboxylate (56.8 mg, 0.4 mmol), diphenyl acetylene (35.6 mg, 0.2 mmol), HOAc (240.2 mg, 4.0 mmol) and MeCN (2 mL) was stirred at room temperature under N2. Then the mixture was heated to 80 ℃ and stirred at 80 ℃ for 24 h. After completion, the mixture was cooled to room temperature. The mixture was passed through the celite with ethyl acetate as an eluent. After evaporation of the solvent, the residue was purified by preparative thin-layer chromatography on silica gel (eluent: petroleum ether/EtOAc, V∶V=3∶1) to give the product 4a. Compounds 4b~4ad were prepared using the same procedure.
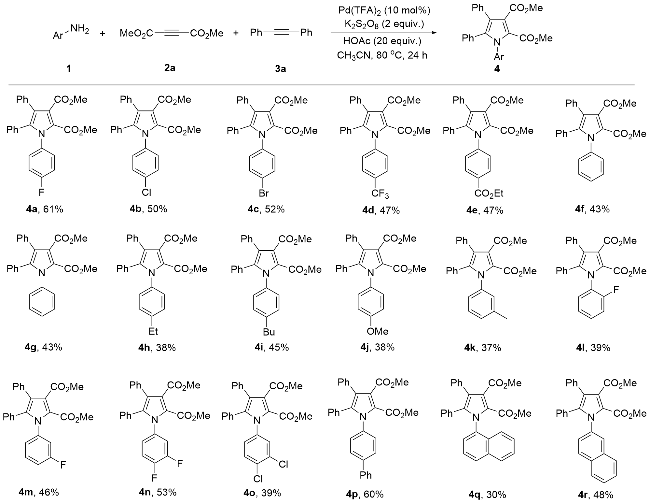
Dimethyl 1-(4-fluorophenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (4a): Yellow solid, 52.1 mg (61% yield). m.p. 167~168 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.20~7.06 (m, 10H), 6.99~6.95 (m, 2H), 6.90 (d, J=8.0 Hz, 2H), 3.80 (s, 3H), 3.71 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.8, 162.2 (d, J=250.5 Hz), 160.4, 137.5, 134.1 (d, J=3.0 Hz), 133.1, 131.2, 130.3 (d, J=8.1 Hz), 129.93 (d, J=16.2 Hz), 129.85, 128.13, 128.06, 126.9, 123.1 (d, J=13.1 Hz), 122.7, 115.5 (d, J=23.2 Hz), 52.5, 52.0; 19F NMR (376 MHz, CDCl3) δ: -112.4; HRMS (ESI) calcd for C26H21FNO4 (M+H)+ 430.1455, found 430.1454.
Dimethyl 1-(4-chlorophenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (4b): White solid, 44.7 mg (50% yield). m.p. 174~175 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.38 (d, J=8.2 Hz, 2H), 7.32~7.19 (m, 10H),7.02 (d, J=7.2 Hz, 2H), 3.92 (s, 3H), 3.84 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.8, 160.4, 137.4, 136.6, 134.4, 133.0, 131.2, 129.9, 129.8, 128.8, 128.1, 127.0, 123.3, 123.2, 122.5, 52.5, 52.1; HRMS (ESI) calcd for C26H21ClNO4 (M+H)+ 446.1159, found 446.1157.
Dimethyl 1-(4-bromophenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (4c): Yellow solid, 51.1 mg (52% yield). m.p. 173~174 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.41 (d, J=8.0 Hz, 2H), 7.20~7.03 (m, 10H), 6.90 (d, J=8.0 Hz, 2H), 3.80 (s, 3H), 3.72 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.4, 160.4, 137.3, 137.2, 133.0, 131.8, 131.2, 130.2, 129.8, 128.2, 127.0, 123.4, 123.2, 122.52, 122.47, 52.5, 52.1; HRMS (ESI) calcd for C26H21BrNO4 (M+H)+ 490.0654, found 490.0661.
Dimethyl 1-(4-trifluoromethylphenyl)-4,5-diphenylpyr- role-2,3-dicarboxylate (4d): White solid, 44.7 mg (47% yield). m.p. 161~162 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.56 (d, J=8.0 Hz, 2H), 7.29 (d, J=8.0 Hz, 2H), 7.21~7.06 (m, 8H), 6.88 (d, J=8.0 Hz, 2H), 3.81 (s, 3H), 3.71 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.7, 160.3, 141.3, 137.4, 132.9, 131.2, 130.5 (q, J=33.3 Hz), 129.8, 129.7, 129.2, 129.0, 128.3, 128.2, 127.11 (q, J=28.3 Hz), 127.10, 125.7 (q, J=4.0 Hz), 123.7, 123.6, 123.5 (q, J=273.7 Hz), 122.3, 119.8, 52.6, 52.1; 19F NMR (376 MHz, CDCl3) δ: -62.5; HRMS (ESI) calcd for C27H21F3NO4 (M+H)+ 480.1423, found 480.1425.
Dimethyl 1-(4-ethoxycarbonylphenyl)-4,5-diphenylpyr- role-2,3-dicarboxylate (4e): Yellow solid, 45.0 mg (47% yield). m.p. 160~161 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.98 (d, J=8.0 Hz, 2H), 7.24~7.04 (m, 10H), 6.89 (d, J=8.0 Hz, 2H), 4.35 (q, J=6.6 Hz, 2H), 3.80 (s, 3H), 3.69 (s, 3H), 1.37 (t, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.8, 165.8, 160.4, 142.1, 137.2, 133.0, 131.2, 130.4, 129.93, 129.87, 128.7, 128.19, 128.16, 127.0, 123.5, 123.4, 122.6, 61.4, 2.6, 52.1, 14.4; HRMS (ESI) calcd for C29H26- NO6 (M+H)+ 484.1760, found 484.1762.
Dimethyl 1,4,5-triphenylpyrrole-2,3-dicarboxylate (
4f): Yellow solid, 35.4 mg (43% yield). m.p. 167~168 ℃(lit.
[15] 162.1~162.9 ℃);
1H NMR (400 MHz, CDCl
3)
δ: 7.30~7.19 (m, 10H), 7.10~7.03 (m, 3H), 6.91 (d,
J=8.0 Hz, 2H), 3.80 (s, 3H), 3.69 (s, 3H).
13C NMR (101 MHz, CDCl
3)
δ: 166.8, 160.6, 138.1, 137.2, 133.3, 131.2, 130.2, 129.9, 128.6, 128.5, 128.4, 128.1, 127.91, 127.87, 126.8, 123.1, 122.9, 122.6, 52.4, 52.0; HRMS (ESI) calcd for C
26H
22NO
4 (M+H)
+ 412.1549, found 412.1543.
Dimethyl 1-(4-methylphenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (4g): Yellow solid, 36.3 mg (43% yield). m.p. 157~158 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.23~7.16 (m, 5H), 7.11~7.04 (m, 7H), 6.92 (d, J=8.0 Hz, 2H), 3.79 (s, 3H), 3.71 (s, 3H), 2.32 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.9, 160.7, 138.3, 137.2, 135.5, 133.4, 131.2, 130.3, 130.0, 129.2, 128.2, 128.1, 127.9, 127.8, 126.8, 123.04, 122.99, 122.4, 52.4, 52.0, 21.3; HRMS (ESI) calcd for C27H24NO4 (M+H)+ 426.1705, found 426.1703.
Dimethyl 1-(4-ethylphenyl)-4,5-diphenylpyrrole-2,3-di- carboxylate (4h): White solid, 33.5 mg (38% yield). m.p. 117~118 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.23~7.15 (m, 5H), 7.11~7.03 (m, 7H), 6.90 (d, J=8.0 Hz, 2H), 3.79 (s, 3H), 3.70 (s, 3H), 3.63 (q, J=7.6 Hz, 2H), 6.90 (t, J=7.6 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.9, 160.7, 144.5, 137.2, 135.6, 133.5, 131.3, 130.4, 130.0, 128.3, 128.1, 127.9, 127.8, 126.8, 123.1, 122.4, 52.4, 52.0, 28.5, 15.2; HRMS (ESI) calcd for C28H26NO4 (M+H)+ 440.1862, found 440.1865.
Dimethyl 1-(4-butylphenyl)-4,5-diphenylpyrrole-2,3-di- carboxylate (4i): Yellow liquid, 42.0 mg (45% yield). 1H NMR (400 MHz, CDCl3) δ: 7.23~7.17 (m, 5H), 7.10~7.03 (m, 7H), 6.90 (d, J=7.4 Hz, 2H), 3.80 (s, 3H), 3.70 (s, 3H), 2.60 (t, J=7.4 Hz, 2H), 1.62~1.54 (m, 2H), 1.36~1.27 (m, 2H), 0.92 (t, J=7.4 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.8, 160.6, 143.2, 137.1, 135.6, 133.5, 131.2, 130.3, 130.0, 128.4, 128.2, 128.0, 127.8, 127.7, 126.8, 123.1, 123.0, 122.3, 52.3, 52.0, 35.3, 33.2, 22.3, 14.0; HRMS (ESI) calcd for C30H30NO4 (M+H)+ 468.2175, found 468.2178.
Dimethyl 1-(4-methoxyphenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (
4j): Yellow solid, 33.8 mg (38% yield). m.p. 182~183 ℃ (lit.
[13a] 182~183 ℃);
1H NMR (400 MHz, CDCl
3)
δ: 7.23~7.03 (m, 10H), 6.92 (d,
J=8.0 Hz, 2H), 6.79 (d,
J=8.0 Hz, 2H), 3.79 (s, 3H), 3.76 (s, 3H), 3.71 (s, 3H);
13C NMR (101 MHz, CDCl
3)
δ: 166.9, 160.7, 159.2, 137.4, 133.4, 131.2, 130.8, 130.3, 130.0, 129.6, 128.1, 128.0, 127.8, 126.8, 123.1, 123.0, 122.3, 113.6, 55.4, 52.4, 52.0; HRMS (ESI) calcd for C
27H
24NO
5 (M+H)
+ 442.1654, found 442.1649.
Dimethyl 1-(3-methylphenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (4k): Yellow solid, 31.3 mg (37% yield). m.p. 216~217 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.19~7.03 (m, 10H), 6.98 (d, J=11.5 Hz, 2H), 6.91 (d, J=8.0 Hz, 2H), 3.79 (s, 3H), 3.70 (s, 3H), 2.27 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.8, 160.7, 138.4, 137.9, 137.1, 133.4, 131.2, 130.3, 130.0, 129.2, 129.1, 128.2, 128.1, 127.9, 126.8, 125.6, 123.14, 123.07, 122.3, 52.4, 52.0, 21.3; HRMS (ESI) calcd for C27H24NO4 (M+H)+ 426.1705, found 426.1706.
Dimethyl 1-(2-fluorophenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (4l): White solid, 33.2 mg (39% yield). m.p. 148~149 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.42~7.39 (m, 1H), 7.36~7.28 (m, 5H), 7.26~7.10 (m, 8H), 3.94 (s, 3H), 3.84 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 167.0, 160.0, 158.5 (d, J=251.5 Hz), 137.8, 133.0, 130.8, 130.6 (d, J=8.1Hz), 130.5, 129.9, 129.7, 128.3, 128.1 (d, J=11.1 Hz), 127.0, 126.5 (d, J=13.1 Hz), 124.04, 123.96 (d, J=4.0 Hz), 123.2, 122.0, 115.9 (d, J=19.2 Hz), 52.6, 52.0; 19F NMR (376 MHz, CDCl3) δ: -120.3; HRMS (ESI) calcd for C26H21FNO4 (M+H)+ 430.1455, found 430.1451.
Dimethyl 1-(3-fluorophenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (4m): Yellow solid, 39.8 mg (46% yield). m.p. 132~133 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.24~7.04 (m, 11H), 6.92~6.88 (m, 3H), 3.78 (s, 3H), 3.69 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.7, 162.2 (d, J=249.5 Hz), 160.3, 139.5 (d, J=10.1 Hz), 137.3, 133.1, 131.1 129.0, 129.86, 129.6 (d, J=9.1 Hz), 128.2, 128.1, 127.0, 124.7 (d, J=3.0 Hz), 123.3, 123.1, 122.6, 116.4 (d, J=23.2 Hz), 115.7 (d, J=21.2 Hz), 52.5, 52.1; 19F NMR (376 MHz, CDCl3) δ: -111.9 (dd, J=14.9, 7.5HZ); HRMS (ESI) calcd for C26H21FNO4 (M+H)+ 430.1455, found 430.1447.
Dimethyl 1-(3,4-difluorophenyl)-4,5-diphenylpyrrole- 2,3-dicarboxylate (4n). White solid, 47.5 mg (53% yield). m.p. 151~152 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.20~7.03 (m, 10H), 6.96~6.88 (m, 3H), 3.80 (s, 3H), 3.73 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.7, 160.2, 150.3 (dd, J=252.5, 13.1 Hz), 149.6 (dd, J=251.5, 14.1 Hz), 137.6, 134.3 (dd, J=8.1, 4.0 Hz), 132.9, 131.1, 129.8, 129.7, 128.4, 128.3, 128.2, 127.1, 125.2 (dd, J=6.1, 3.0 Hz), 123.5, 123.4, 118.4 (d, J=19.2 Hz), 117.0 (d, J=18.2 Hz), 52.6, 52.1; 19F NMR (376 MHz, CDCl3) δ: -135.4~-135.5 (m), -136.3~-136.4 (m); HRMS (ESI) calcd for C26H20F2NO4 (M+H)+ 448.1360, found 448.1355.
Dimethyl 1-(3,4-dichlorophenyl)-4,5-diphenylpyrrole- 2,3-dicarboxylate (4o): White solid, 37.6 mg (39% yield). m.p. 145~146 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.35 (d, J=8.2 Hz, 1H),7.29 (s, 1H), 7.20~7.12 (m, 8H), 7.02 (d, J=8.2 Hz, 1H), 6.91 (d, J=8.0 Hz, 2H), 3.80 (s, 3H), 3.73 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.7, 160.2, 137.5, 132.9, 132.8, 132.5, 131.2, 130.6, 130.1, 129.8, 129.6, 128.4, 128.3, 128.2, 128.1, 127.1, 123.7, 123.5, 122.3, 52.6, 52.2; HRMS (ESI) calcd for C26H20Cl2NO4 (M+H)+ 480.0769, found 480.0763.
Dimethyl 1-biphenyl-4,5-diphenylpyrrole-2,3-dicarbox- ylate (4p): White solid, 58.1 mg (60% yield). m.p. 164~165 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.70 (d, J=8.0 Hz, 2H), 7.66 (d, J=8.0 Hz, 2H), 7.57~7.45 (m, 3H), 7.38~7.34 (m, 7H), 7.24~7.18 (m, 3H), 7.08 (d, J=8.0 Hz, 2H), 3.94 (s, 3H), 3.86 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.8, 160.6, 141.0, 139.9, 137.2, 133.3, 131.2, 130.2, 129.9, 128.89,128.85, 128.1, 128.0, 127.9, 127.8, 127.2, 127.0, 126.9, 123.2, 122.9, 122.7, 52.4, 52.1. HRMS (ESI) calcd for C32H26NO4 (M+H)+ 488.1862, found 488.1862.
Dimethyl 1-(1-naphthyl)-4,5-diphenylpyrrole-2,3-dicar- boxylate (4q): White solid, 27.6 mg (30% yield). m.p. 139~140 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.82~7.77 (m, 2H), 7.46~7.35 (m, 5H), 7.23~7.18 (m, 5H), 7.00~6.97 (m, 1H), 6.91~6.85 (m, 4H), 3.84 (s, 3H), 3.52 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 167.1, 160.1, 138.3, 135.1, 133.5, 133.3, 131.7, 130.7, 130.2, 129.9, 129.4, 129.2, 128.23, 128.17, 128.0, 127.7, 127.4, 126.9, 126.8, 126.5, 124.8, 123.24, 123.16, 122.7, 118.6, 11.2, 52.5, 51.8; HRMS (ESI) calcd for C30H24NO4 (M+H)+ 462.1705, found 462.1702.
Dimethyl 1-(2-naphthyl)-4,5-diphenylpyrrole-2,3-dicar- boxylate (4r): White solid, 44.7 mg (48% yield). m.p. 171~172 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.82 (d, J=7.6 Hz, 1H), 7.77 (d, J=8.6 Hz, 1H), 7.72 (d, J=7.6 Hz, 1H), 7.62 (s, 1H), 7.51~7.44 (m, 2H), 7.31 (d, J=8.6 Hz, 1H), 7.24~7.16 (m, 5H), 7.06~6.93 (m, 5H), 3.81 (s, 3H), 3.66 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.8, 160.6, 137.4, 135.6, 133.4, 132.8, 132.7, 131.2, 130.2, 130.0, 128.3, 128.2, 128.1, 128.0, 127.9, 127.2, 126.9, 126.8, 126.7, 126.5, 123.4, 123.3, 122.7, 52.4, 52.0; HRMS (ESI) calcd for C30H24NO4 (M+H)+ 462.1705, found 462.1704.
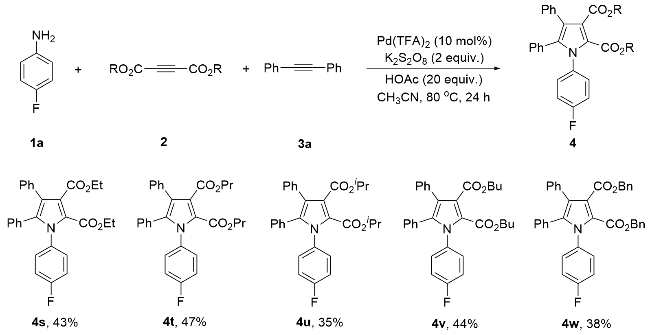
Diethyl 1-(4-fluorophenyl)-4,5-diphenylpyrrole-2,3-di- carboxylate (4s): Yellow solid, 39.4 mg (43% yield). m.p. 97~98 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.19~7.05 (m, 10H), 6.99~6.95 (m, 2H), 6.90 (d, J=8.0 Hz, 2H), 4.27 (q, J=7.2 Hz, 2H), 4.16 (q, J=7.2 Hz, 2H), 1.23 (t, J=7.2 Hz, 3H), 1.17 (t, J=7.2 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.3, 162.1 (d, J=249.5 Hz), 160.0, 137.2, 134.2 (d, J=3.0 Hz), 133.3, 131.2, 130.4 (d, J=8.1 Hz), 130.1, 129.9, 128.1 (d, J=1.0 Hz), 128.0, 126.9, 123.2 (d, J=19.2 Hz), 122.8, 115.5 (d, J=23.2 Hz), 61.4, 60.9, 14.1, 14.0; 19F NMR (376 MHz, CDCl3) δ: -112.6; HRMS (ESI) calcd for C28H25FNO4 (M+H)+ 458.1768, found 458.1767.
Dipropyl 1-(4-fluorophenyl)-4,5-diphenylpyrrole-2,3-di- carboxylate (4t): Yellow liquid, 45.9 mg (47% yield). 1H NMR (400 MHz, CDCl3) δ: 7.19~7.05 (m, 10H), 6.99~6.95 (m, 2H), 6.90 (d, J=8.0 Hz, 2H), 4.15 (t, J=6.6 Hz, 2H), 4.07 (t, J=6.6 Hz, 2H), 1.62~1.53 (m, 4H), 0.86 (t, J=7.2 Hz, 3H), 0.79 (t, J=7.2 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.5, 162.2 (d, J=249.5 Hz), 160.2, 137.2, 134.3 (d, J=3.0 Hz), 133.3, 131.2, 130.4 (d, J=9.1 Hz), 130.1, 129.9, 128.1 (d, J=5.1 Hz), 128.0, 126.9, 123.3 (d, J=25.3 Hz), 122.8, 115.5 (d, J=23.2 Hz), 67.1, 66.7, 21.94, 21.89, 10.5, 10.4; 19F NMR (376 MHz, CDCl3) δ: -112.6; HRMS (ESI) calcd for C30H29FNO4 (M+H)+ 486.2081, found 486.2089.
Diisopropyl 1-(4-fluorophenyl)-4,5-diphenylpyrrole-2,3- dicarboxylate (4u): Yellow solid, 34.1 mg (35% yield). m.p. 152~153 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.19~7.04 (m, 10H), 6.99~6.95 (m, 2H), 6.89 (d, J=8.0 Hz, 2H), 5.17~5.11 (m, 1H), 5.07~5.01 (m, 1H), 1.17 (d, J=7.2 Hz, 6H), 1.15 (d, J=7.2 Hz, 6H); 13C NMR (101 MHz, CDCl3) δ: 165.8, 162.1 (d, J=249.5 Hz), 159.5, 136.9, 134.5 (d, J=3.0 Hz), 133.4, 131.2, 130.4 (d, J=9.1 Hz), 130.2, 130.0, 128.01, 127.97 (d, J=8.1 Hz), 126.8, 123.6, 123.1 (d, J=7.1 Hz), 115.4 (d, J=23.2 Hz), 69.0, 68.6, 21.7; 19F NMR (376 MHz, CDCl3) δ: -112.8; HRMS (ESI) calcd for C30H29FNO4 (M+H)+ 486.2081, found 486.2082.
Dibutyl 1-(4-fluorophenyl)-4,5-diphenylpyrrole-2,3-di- carboxylate (4v): Yellow liquid, 45.6 mg (44% yield). 1H NMR (400 MHz, CDCl3) δ: 7.19~7.05 (m, 10H), 6.99~6.95 (m, 2H), 6.90 (d, J=8.0 Hz, 2H), 4.19 (t, J=6.6 Hz, 2H), 4.11 (t, J=6.6 Hz, 2H), 1.56~1.49 (m, 4H), 1.30~1.17 (m, 4H), 0.88 (t, J=7.2 Hz, 3H), 0.83 (t, J=7.2 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.5, 162.2 (d, J=249.5 Hz), 160.2, 137.2, 134.3 (d, J=3.0 Hz), 133.4, 131.2, 130.4 (d, J=9.1 Hz), 130.1, 129.9, 128.1 (d, J=5.1 Hz), 128.0, 126.9, 123.3 (d, J=27.3 Hz), 122.8, 115.5 (d, J=23.2 Hz), 65.4, 64.9, 30.61, 30.58, 19.2, 19.1, 13.8, 13.7; 19F NMR (376 MHz, CDCl3) δ: -112.6; HRMS (ESI) calcd for C32H33FNO4 (M+H)+ 514.2394, found 514.2392.
Dibenzyl 1-(4-fluorophenyl)-4,5-diphenylpyrrole-2,3-di- carboxylate (4w): White solid, 43.8 mg (38% yield). m.p. 136~137 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.35~7.25 (m, 8H), 7.20~7.08 (m, 12H), 6.97~6.91 (m, 4H), 5.01 (s, 2H), 5.00 (s, 2H); 13C NMR (101 MHz, CDCl3) δ: 166.0, 162.2 (d, J=249.5 Hz), 159.8, 137.4, 135.5, 135.4, 134.1 (d, J=3.0 Hz), 133.1, 131.2, 130.3 (d, J=8.1 Hz), 130.0, 129.9, 128.6 (d, J=13.1 Hz), 128.5, 128.4, 128.2, 128.1, 128.0, 126.9, 123.3 (d, J=12.1 Hz), 122.6, 115.5 (d, J=23.2 Hz), 67.2, 66.9; 19F NMR (376 MHz, CDCl3) δ: -112.4; HRMS (ESI) calcd for C38H29FNO4 (M+H)+ 582.2081, found 582.2079.
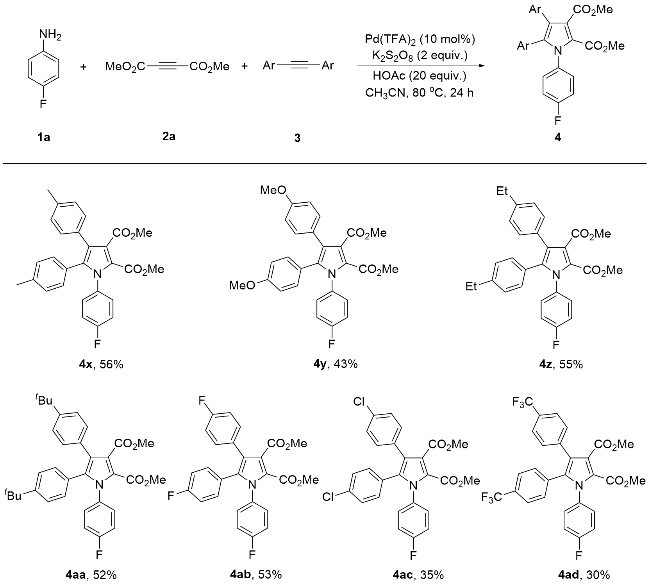
Dimethyl 1-(4-fluorophenyl)-4,5-di(4-methylphenyl)py- rrole-2,3-dicarboxylate (4x): Yellow solid, 50.9 mg (56% yield). m.p. 171~172 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.15~6.95 (m, 8H), 6.88 (d, J=8.0 Hz, 2H), 6.78 (d, J=8.0 Hz, 2H), 3.81 (s, 3H), 3.70 (s, 3H), 2.29 (s, 3H), 2.21 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 167.1, 162.1 (d, J=249.5 Hz), 160.4, 137.8, 137.7, 136.5, 134.3 (d, J=3.0 Hz), 131.0, 130.4 (d, J=9.1 Hz), 130.2, 129.6, 128.9 (d, J=10.1 Hz), 127.1, 123.1 (d, J=31.1 Hz), 122.2, 115.5 (d, J=23.2 Hz), 52.5, 52.0, 21.33, 21.31; 19F NMR (376 MHz, CDCl3) δ: -112.6; HRMS (ESI) calcd for C28H25- FNO4 (M+H)+ 458.1768, found 458.1759.
Dimethyl 1-(4-fluorophenyl)-4,5-di(4-methoxyphenyl)- pyrrole-2,3-dicarboxylate (4y): Yellow solid, 42.2 mg (43% yield). m.p. 203~204 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.13~7.09 (m, 4H), 7.00~6.96 (m, 2H), 6.81~6.75 (m, 4H), 6.61 (d, J=8.0 Hz, 2H), 3.81 (s, 3H), 3.76 (s, 3H), 3.69 (s, 6H); 13C NMR (101 MHz, CDCl3) δ: 167.1, 162.1 (d, J=248.5 Hz), 160.5, 159.2, 158.5, 137.4, 134.3 (d, J=4.0 Hz), 132.4, 131.0, 130.4 (d, J=9.1 Hz), 125.6, 123.2, 122.5 (d, J=26.3 Hz), 122.1, 115.5 (d, J=23.2 Hz), 113.6 (d, J=8.1 Hz), 55.22, 55.17, 52.5, 52.0; 19F NMR (376 MHz, CDCl3) δ: -112.7; HRMS (ESI) calcd for C28H25FNO6 (M+H)+ 490.1666, found 490.1664.
Dimethyl 1-(4-fluorophenyl)-4,5-di(4-ethylphenyl)pyr- role-2,3-dicarboxylate (4z): Yellow solid, 53.3 mg (55% yield). m.p. 187~188 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.17~7.13 (m, 2H), 7.10 (d, J=8.0 Hz, 2H), 7.04 (d, J=8.0 Hz, 2H), 6.99~6.95 (m, 2H), 6.91 (d, J=8.0 Hz, 2H), 6.82 (d, J=8.0 Hz, 2H), 3.81 (s, 3H), 3.70 (s, 3H), 2.60 (q, J=7.6 Hz, 2H), 2.52 (q, J=7.6 Hz, 2H), 1.21 (t, J=7.6 Hz, 2H), 1.14 (t, J=7.6 Hz, 2H); 13C NMR (101 MHz, CDCl3) δ: 167.1, 162.1 (d, J=249.5 Hz), 160.4, 143.9, 142.6, 137.7, 134.3 (d, J=3.0 Hz), 131.0, 130.3 (d, J=8.1 Hz), 129.6, 127.5 (d, J=13.1 Hz), 127.2, 123.1 (d, J=39.4 Hz), 122.1, 115.4 (d, J=22.2 Hz), 52.4, 51.9, 28.5, 28.4, 15.2, 15.0; 19F NMR (376 MHz, CDCl3) δ: -112.7; HRMS (ESI) calcd for C30H29FNO4 (M+H)+ 486.2081, found 486.2079.
Dimethyl 1-(4-fluorophenyl)-4,5-di(4-tert-butylphenyl)- pyrrole-2,3-dicarboxylate (4aa): Yellow solid, 56.4 mg (52% yield). m.p. 173~174 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.21 (d, J=8.0 Hz, 2H), 7.15~7.07 (m, 6H), 6.98~6.94 (m, 2H), 6.82 (d, J=8.0 Hz, 2H), 3.82 (s, 3H), 3.69 (s, 3H), 1.28 (s, 9H), 1.21 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 167.3, 162.1 (d, J=249.5 Hz), 160.4, 150.9, 149.5, 137.8, 134.3 (d, J=3.0 Hz), 130.8, 130.4 (d, J=9.1 Hz), 130.1, 129.3, 127.0, 124.9 (d, J=14.1 Hz), 123.1 (d, J=54.5 Hz), 122.0, 115.4 (d, J=23.2 Hz), 52.5, 51.9, 34.6, 34.5, 31.4, 31.2; 19F NMR (376 MHz, CDCl3) δ: -112.9; HRMS (ESI) calcd for C34H37FNO4 (M+H)+ 542.2707, found 542.2712.
Dimethyl 1,4,5-tri(4-fluorophenyl)pyrrole-2,3-dicarbo- xylate (4ab): Yellow solid, 49.2 mg (53% yield). m.p. 161~162 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.17~7.07 (m, 4H), 7.01~6.77 (m, 8H), 3.80 (s, 3H), 3.71 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.6, 162.3 (dd, J=249.5, 8.1 Hz), 162.0 (d, J=247.5 Hz), 160.5, 136.3, 133.8 (d, J=4.0 Hz), 131.0 (d, J=8.1 Hz), 131.6 (d, J=8.1 Hz), 130.3 (d, J=9.1 Hz), 128.9 (d, J=13.1 Hz), 125.9 (d, J=13.1 Hz), 123.2, 122.5 (d, J=11.1 Hz), 115.9, 115.6 (d, J=5.1 Hz), 115.4 (d, J=2.0 Hz), 115.2, 52.6, 52.2; 19F NMR (376 MHz, CDCl3) δ: -111.9, -112.3, -115.1; HRMS (ESI) calcd for C26H19F3NO4 (M+H)+ 466.1266, found 466.1266.
Dimethyl 1-(4-fluorophenyl)-4,5-di(4-chlorophenyl)pyr- role-2,3-dicarboxylate (4ac): White solid, 34.8 mg (35% yield). m.p. 194~195 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.21 (d, J=8.0 Hz, 2H), 7.15~7.07 (m, 6H), 7.02~6.98 (m, 2H), 6.80 (d, J=8.0 Hz, 2H), 3.80 (s, 3H), 3.71 (s, 3H), 2.29 (s, 3H), 2.21 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.3, 162.4 (d, J=250.5 Hz), 160.4, 135.9, 134.5, 133.7 (d, J=3.0 Hz), 133.3, 132.3, 131.4, 131.3, 130.2 (d, J=9.1 Hz), 128.6 (d, J=7.1 Hz), 128.2, 123.8, 122.4 (d, J=7.1 Hz), 115.9 (d, J=23.2 Hz), 52.6, 52.2; 19F NMR (376 MHz, CDCl3) δ: -111.7; HRMS (ESI) calcd for C26H19Cl2FNO4 (M+H)+ 498.0675, found 498.0671.
Dimethyl 1-(4-fluorophenyl)-4,5-di(4-trifluoromethyl- phenyl)pyrrole-2,3-dicarboxylate (4ad): Yellow solid, 34.0 mg (30% yield). m.p. 180~181 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.50 (d, J=7.9 Hz, 2H), 7.36 (d, J=7.9 Hz, 2H),7.27 (d, J=8.1 Hz, 2H), 7.17~7.14 (m, 2H), 7.04~6.99 (m, 4H), 3.80 (s, 3H), 3.73 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 166.0, 162.5 (d, J=250.5 Hz), 160.4, 136.6, 135.5, 133.4 (d, J=3.0 Hz), 133.3, 131.4, 130.4 (q, J=33.3 Hz), 130.3, 130.2 (d, J=9.1 Hz), 129.9 (q, J=2.0 Hz), 129.4 (q, J=32.3 Hz), 125.3 (d, J=4.0 Hz), 124.9 (q, J=36.4 Hz), 124.8, 124.2 (q, J=272.7 Hz), 123.8 (q, J=273.7 Hz), 123.5 (q, J=38.4 Hz), 122.5 (q, J=5.1 Hz), 122.4 (d, J=59.6 Hz), 116.0 (d, J=23.2 Hz), 52.6, 52.4; 19F NMR (376 MHz, CDCl3) δ: -62.5, -62.9, -111.2; HRMS (ESI) calcd for C28H19F7NO4 (M+H)+ 566.1202, found 566.1212.