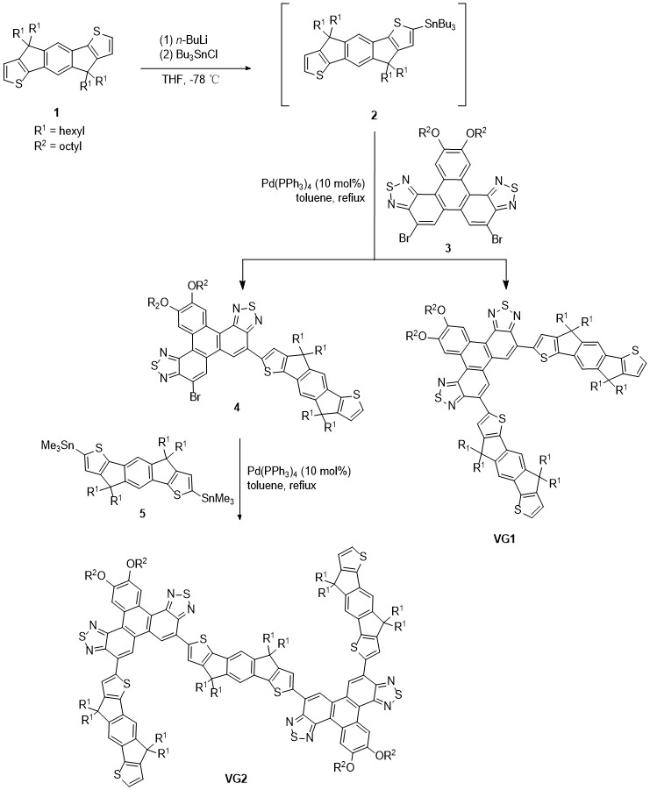
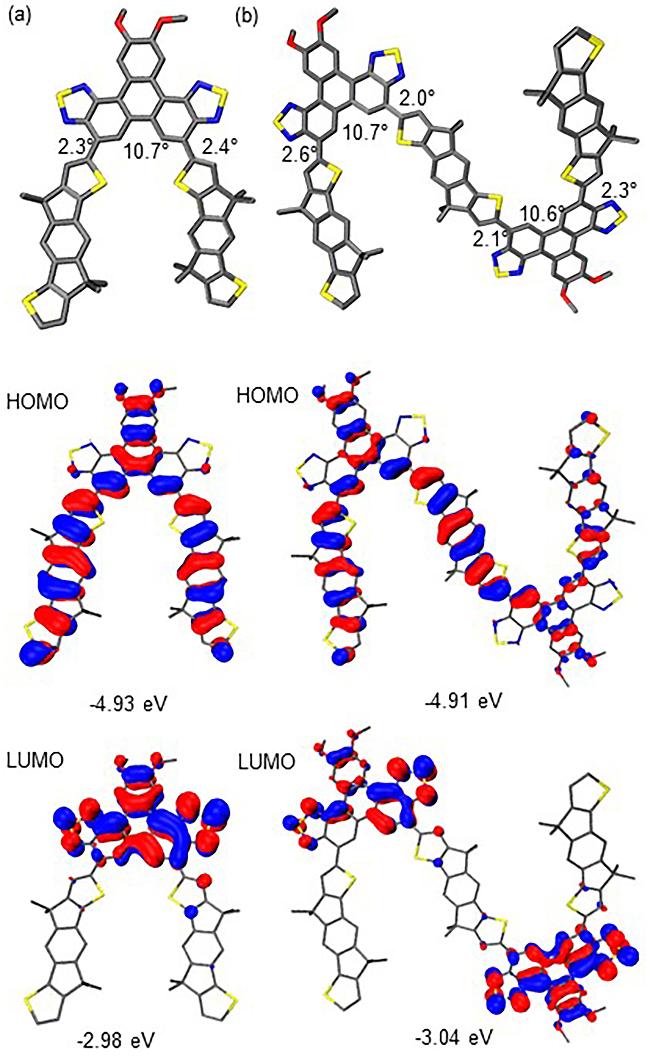
1 Introduction
2 Results and discussion
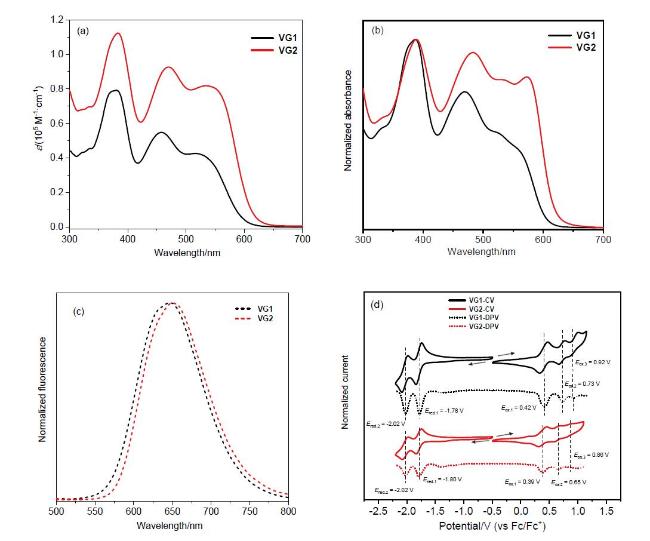
Table 1 Optical and electrochemical data of VG1 and VG2 |
| Compd. | λsolutiona/ nm | λfilma/ nm | λemb/ nm | Egopt c/ eV | EHOMOd/ eV | ELUMOd/ eV | ΦFL-solidf/ % | ΦFL-solutiong/ % | τse/ns | kr h/ (108 s-1) | knr h/ (108 s-1) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| VG1 | 380, 457, 535 | 387, 469 | 647 | 2.05 | -5.10 | -3.09 | 0.8 | 45.3 | 4.3 | 1.05 | 1.27 |
| VG2 | 383, 469, 515 | 389, 483, 572 | 650 | 2.01 | -5.07 | -3.09 | 1.8 | 41.3 | 3.4 | 1.21 | 1.72 |
. a The maximum absorption and emission wavelengths measured in CH2Cl2. b The maximum emission wavelength measured in CH2Cl2. c Optical band gaps calculated from the onset wavelength of the absorption spectra using the equation Egopt=1240/λonset. d Frontier molecular orbital energy levels from cyclic voltammetry were estimated as: EHOMOcv (eV)=-(Eonsetox-EFc/Fc++4.8) (eV), ELUMOcv (eV)=-(Eonsetred-EFc/Fc++4.8) (eV). e Fluorescence lifetime (τs). f Absolute quantum yield measured in the solid state. g Absolute quantum yield measured in CH2Cl2. h Radiative (kr) and nonradiative (knr) decay rate constants calculated by the equations kr=ΦFL-solution/τs and knr=(1-ΦFL-solution)/τs. |
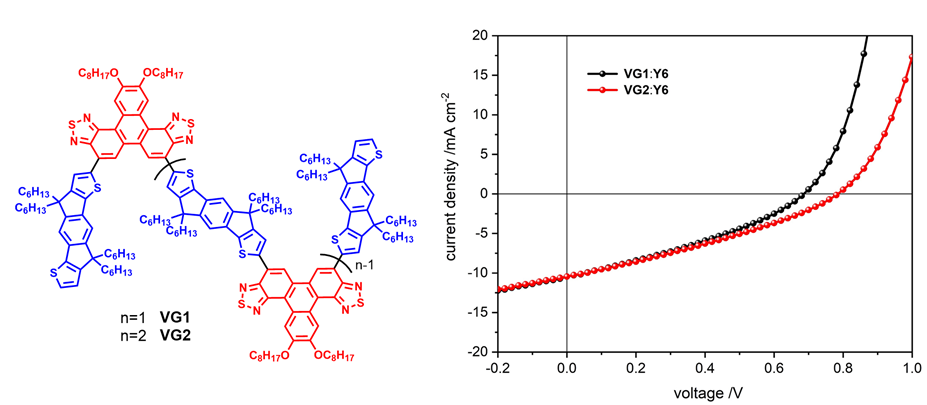
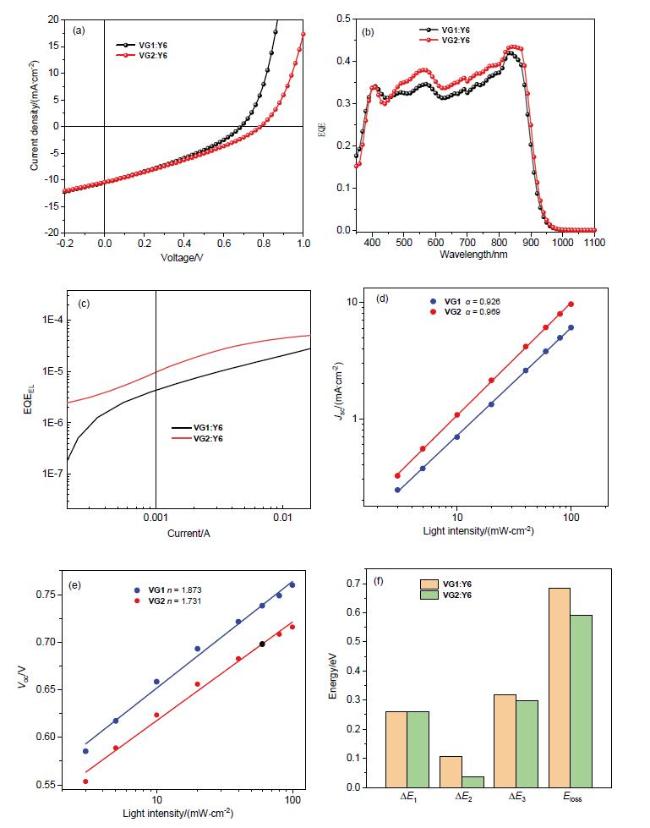
Table 2 Photovoltaic parameters of devices based on VG1 and VG2 |
| Device | Voc/V | Jsc/(mA•cm-2) | FF/% | PCE/% | Eg/eV | ΔE1/eV | ΔE2/eV | ΔE3/eV | Eloss/eV | EQEEL/% |
|---|---|---|---|---|---|---|---|---|---|---|
| VG1:Y6 | 0.68 | 10.49 | 32.95 | 2.37 | 1.382 | 0.261 | 0.105 | 0.318 | 0.684 | 4.36×10-4 |
| VG2:Y6 | 0.78 | 10.44 | 31.33 | 2.56 | 1.371 | 0.260 | 0.035 | 0.298 | 0.592 | 9.78×10-4 |