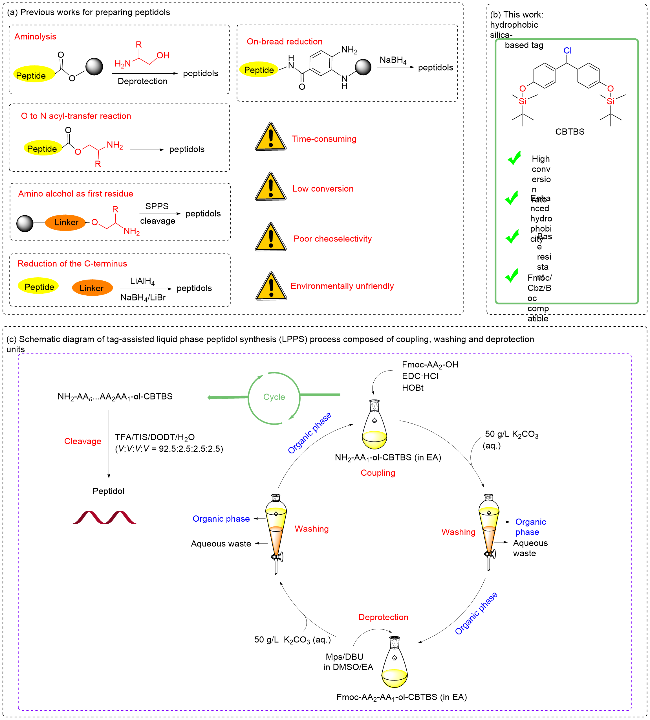
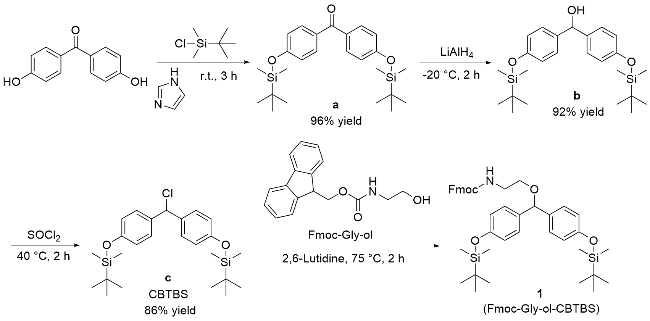
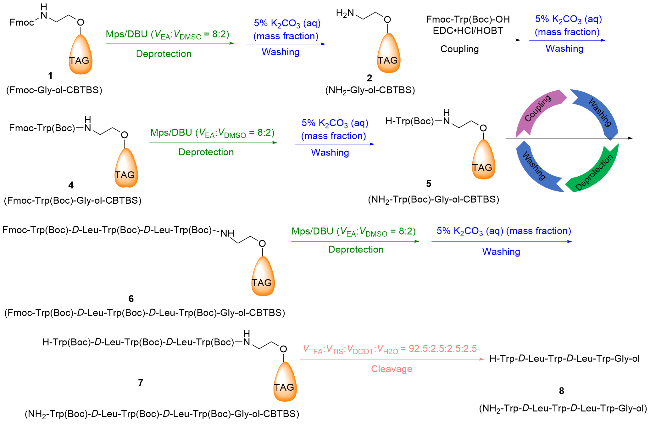
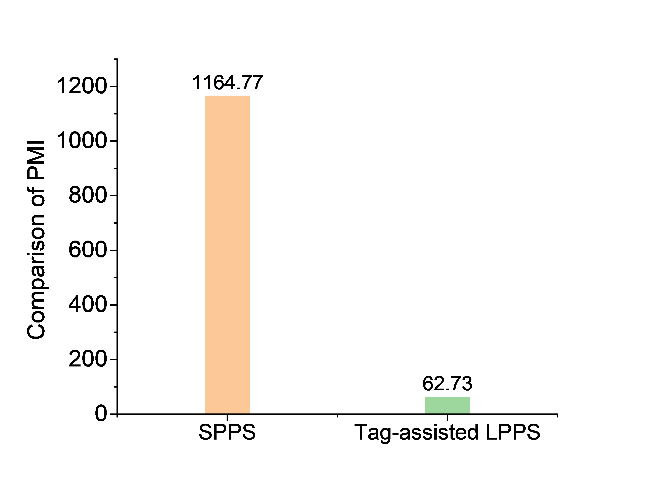将Fmoc-Gly-ol (0.62 g, 2.2 mmol)、2,6-二甲基吡啶(0.24 g, 2.2 mmol)和化合物c (0.92 g, 2.0 mmol)溶解于20 mL EA中, 75 ℃下反应2 h. 经HPLC检测反应完成后, 加入等体积的50 g/L的K2CO3水溶液洗涤(20 mL×3), 之后经减压浓缩和柱层析纯化[洗脱剂为V(EA)∶ V(PE)=1∶15]得到白色固体(9H-芴-9-基)甲基-2-(双(4-(叔丁基二甲基硅氧基)苯基)甲氧基)乙基氨基甲酸酯(Fmoc-Gly-ol-CBTBS, 1) 1.35 g, 1.9 mmol). 1H NMR (400 MHz, Chloroform-d) δ: 7.77 (d, J=7.5 Hz, 2H), 7.60 (d, J=7.5 Hz, 2H), 7.40 (t, J=7.4 Hz, 2H), 7.34~7.27 (m, 2H), 7.20~7.09 (m, 4H), 6.87~6.64 (m, 4H), 5.26 (s, 1H), 5.18 (s, 1H), 4.38 (d, J=7.1 Hz, 2H), 4.23 (t, J=7.1 Hz, 1H), 3.52 (t, J=5.0 Hz, 2H), 3.44 (t, J=5.2 Hz, 2H), 0.97 (s, 18H), 0.18 (s, 12H); 13C NMR (101 MHz, Chloroform-d) δ: 156.23, 154.85, 143.79, 141.10, 134.48, 127.99, 127.50, 127.47, 126.84, 124.91, 119.77, 119.66, 83.01, 67.55, 66.55, 47.04, 40.92, 25.46, 17.96, -4.61; HRMS- ESI calcd for C42H55NNaO5Si2 [M+Na]+ 732.3546, found 732.3571.
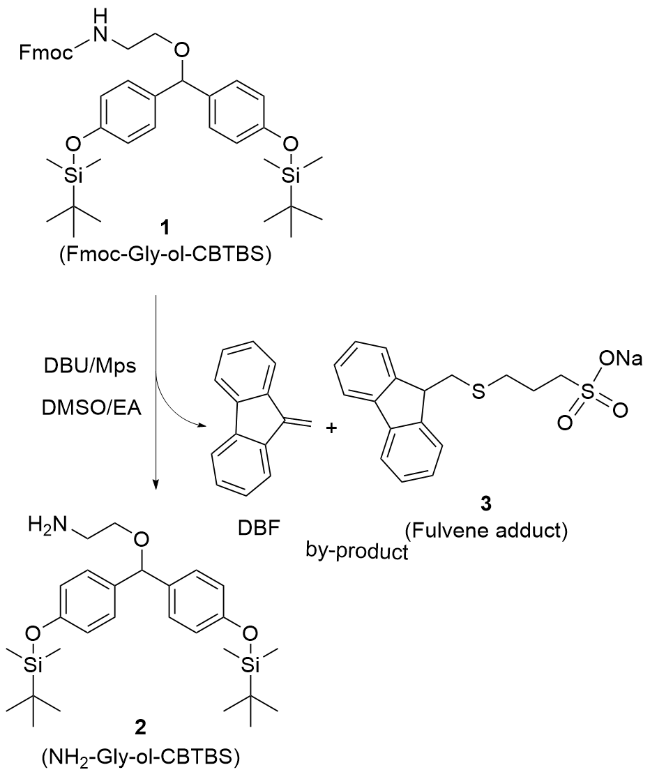
将Fmoc-Gly-ol-CBTBS (1, 1.35 g, 1.9 mmol)溶解在20 mL的DMSO/EA (V∶V=2∶8)混合溶剂中, 再加入3.8 mmol Mps和1.9 mmol DBU, 40 ℃下进行脱保护20 min, 经HPLC检测反应完全, 用等体积的5 g/L K2CO3水溶液洗涤(20 mL×3), 以去除体系中的加成产物、过量DBU和Mps, 以及偶联步骤中未彻底水洗去除的无保护基氨基酸. 减压浓缩有机相, 得到2-(双(4-(叔丁基二甲基硅氧基)苯基)甲氧基)乙胺(NH2-Gly-ol-CBTBS, 2) 0.91 g, 1.86 mmol. 1H NMR (400 MHz, Chloroform-d) δ: 7.77 (d, J=7.5 Hz, 2H), 7.60 (d, J=7.5 Hz, 2H), 7.40 (t, J=7.4 Hz, 2H), 7.34~7.27 (m, 2H), 7.20~7.09 (m, 4H), 6.87~6.64 (m, 4H), 5.26 (s, 1H), 5.18 (s, 1H), 4.38 (d, J=7.1 Hz, 2H), 4.23 (t, J=7.1 Hz, 1H), 3.52 (t, J=5.0 Hz, 2H), 3.44 (t, J=5.2 Hz, 2H), 0.97 (s, 18H), 0.18 (s, 12H); 13C NMR (101 MHz, Chloroform-d) δ: 156.23, 154.85, 143.79, 141.10, 134.48, 127.99, 127.50, 127.47, 126.84, 124.91, 119.77, 119.66, 83.01, 67.55, 66.55, 47.04, 40.92, 25.46, 17.96, -4.61; HRMS-ESI calcd for C27H45- NNaO3Si2 [M+Na]+ 510.2878, found 510.2848.
9-亚甲基-9H-芴(DBF): 1H NMR (400 MHz, Chloroform-d) δ: 7.69 (dd, J=16.8, 7.5 Hz, 4H), 7.32 (dtd, J=28.6, 7.5, 1.2 Hz, 4H), 6.05 (s, 2H); 13C NMR (101 MHz, Chloroform-d) δ: 143.39, 140.20, 138.08, 128.79, 127.11, 121.07, 119.81, 107.86; HRMS-ESI calcd for C14H11 [M+H]+ 179.0159, found 179.0186.
3-(((9H-芴-9-基)甲基)硫基)丙烷-1-磺酸钠(3): 1H NMR (400 MHz, Chloroform-d) δ: 7.61 (dd, J=30.6, 7.4 Hz, 4H), 7.26 (dt, J=28.1, 7.4 Hz, 4H), 3.99 (t, J=6.2 Hz, 1H), 3.02 (dd, J=48.9, 7.0 Hz, 4H), 2.49 (t, J=6.9 Hz, 2H), 2.17~1.98 (m, 2H); 13C NMR (101 MHz, Chloroform-d) δ: 145.96, 141.12, 127.71, 127.17, 124.90, 120.01, 46.90, 36.00, 31.28, 23.76; HRMS-ESI calcd for C17H18- NaO3S2 [M+H]+ 357.0134, found 357.0104.
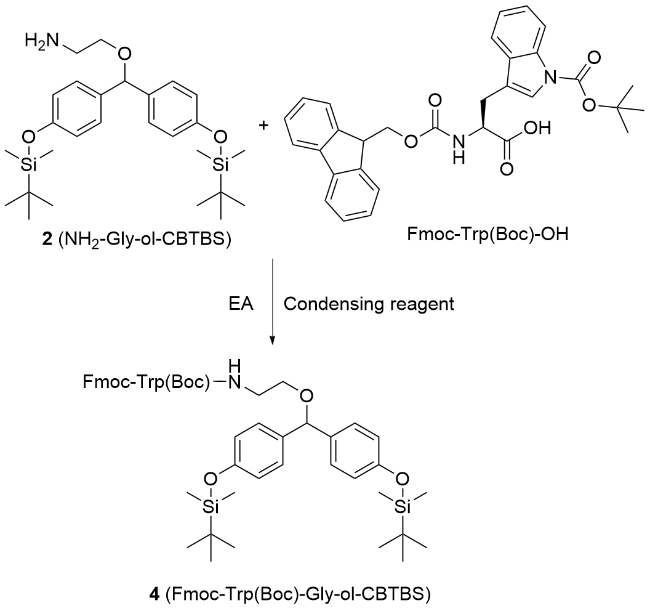
活化氨基酸: 称取相对于化合物2 (0.91 g, 1.86 mmol)为1.05 equiv.的Fmoc-Trp(Boc)-OH (1.03 g, 1.95 mmol), 1.1 equiv.的偶联试剂EDC•HCl (0.39 g, 2.05 mmol)和HOBt (0.28 g, 2.05 mmol)溶解在20 mL EA中, 在室温下预活化30 min, 可以活化氨基酸羧基以便进行偶联反应. Gramicidin A片段序列中所需要的Fmoc-D- Leu-OH、Fmoc-Trp(Boc)-OH、Fmoc-D-Leu-OH和Fmoc- Trp(Boc)-OH使用上述方法进行活化.
将NH2-Gly-ol-CBTBS (2)溶解于Fmoc-Trp(Boc)- OH的活化液中, 40 ℃下搅拌40 min, 经HPLC检测反应完全后, 用等体积的50 g/L的K2CO3水溶液洗涤(20 mL×3), 去除体系中过量氨基酸及偶联试剂残留后, 获得目标产物Fmoc-Trp(Boc)-Gly-ol-CBTBS (4, 1.81 g, 1.82 mmol), 之后再溶解于20 mL的DMSO/EA (V∶V=2∶8)混合溶剂中, 往其加入Mps (0.65 g, 3.62 mmol)和DBU (0.28 g, 1.81 mmol), 脱保护得到NH2-Trp(Boc)- Gly-ol-CBTBS (5, 1.38 g, 1.79 mmol).
(S)-3-(2-(((9H-芴-9-基)甲氧基)羰基)氨基)-3-((2-(双(4-(叔丁基二甲基硅氧基)苯基)甲氧基)乙基)氨基)-3-氧代丙基)-1H-吲哚1羧酸叔丁酯(4): 1H NMR (400 MHz, Chloroform-d) δ: 8.17 (s, 1H), 7.78 (d, J=7.6 Hz, 2H), 7.66~7.50 (m, 3H), 7.47~7.38 (m, 2H), 7.38~7.30 (m, 3H), 7.26 (d, J=7.4 Hz, 1H), 7.06~6.93 (m, 4H), 6.74 (dd, J=8.3, 1.5 Hz, 4H), 6.02 (d, J=6.0 Hz, 1H), 5.58 (d, J=7.9 Hz, 1H), 5.02 (s, 1H), 4.50~4.41 (m, 2H), 4.39~4.31 (m, 1H), 4.15 (q, J=7.1 Hz, 1H), 3.37 (d, J=5.2 Hz, 3H), 3.28 (d, J=19.3 Hz, 1H), 3.17 (d, J=7.7 Hz, 2H), 2.07 (s, 1H), 1.64 (s, 7H), 1.34~1.25 (m, 2H), 0.98 (d, J=2.9 Hz, 18H), 0.18 (d, J=2.6 Hz, 12H); 13C NMR (101 MHz, Chloroform-d) δ: 170.76, 155.99, 155.11, 155.06, 149.60, 143.83, 143.80, 141.37, 135.55, 134.59, 134.42, 130.34, 128.25, 128.17, 127.84, 127.21, 125.19, 124.85, 124.43, 122.94, 120.10, 119.93, 119.88, 119.17, 115.51, 83.84, 83.16, 67.28, 67.20, 55.44, 47.19, 28.79, 28.28, 25.75, 18.24, -4.32; HRMS-ESI calcd for C58H73N3Na- O8Si2 [M+Na]+ 1018.4889, found 1018.4860.
(S)-3-(2-氨基-3-((2-(双(4-(叔丁基二甲基硅氧基) 苯基)甲氧基)乙基)氨基)-3-氧代丙基)-1H-吲哚-1-羧酸叔丁酯(5): 1H NMR (400 MHz, Chloroform-d) δ: 8.23~8.08 (m, 1H), 7.68 (dd, J=18.5, 6.3 Hz, 2H), 7.57~7.46 (m, 1H), 7.40~7.31 (m, 1H), 7.26 (d, J=6.9 Hz, 1H), 7.15 (dd, J=8.4, 6.7 Hz, 4H), 6.79 (d, J=8.3 Hz, 4H), 5.21 (s, 1H), 4.15 (q, J=7.1 Hz, 1H), 3.73 (dd, J=9.5, 3.9 Hz, 1H), 3.61~3.34 (m, 5H), 2.83 (dd, J=14.5, 9.5 Hz, 1H), 2.07 (s, 1H), 1.69 (s, 9H), 0.99 (s, 18H), 0.20 (s, 12H). 13C NMR (101 MHz, Chloroform-d) δ: 174.42, 155.11, 134.88, 134.81, 131.04, 130.45, 128.96, 128.34, 124.76, 124.21, 122.77, 119.89, 119.41, 116.97, 115.44, 83.13, 67.72, 65.68, 60.51, 55.05, 39.11, 30.84, 28.33, 25.76, 18.27, -4.30; HRMS-ESI calcd for C38H54N3O4Si2 [M-Boc+H]+ 701.4992, found 701.4963.
肽链延伸阶段采用循环合成策略, 依次执行上述偶联与脱保护操作, 最终得到带有CBTBS标签的全保护Gramicidin A片段Fmoc-Trp(Boc)-D-Leu-Trp (Boc)-D- Leu-Trp(Boc)-Gly-ol-CBTBS (6, 3.00 g, 1.66 mmol). 之后将化合物6通过脱保护得到NH2-Trp(Boc)-D-Leu-Trp- (Boc)-D-Leu-Trp(Boc)-Gly-ol-CBTBS (7, 2.59 g, 1.65 mmol). 在30 ℃下, 采用体积比为92.5∶2.5∶2.5∶2.5的TFA-TIS-DODT-H₂O四元混合体系, 对CBTBS保护基及侧链保护基进行2 h的裂解反应, 随后通过甲基叔丁基醚沉降处理, 经离心分离和氮气吹干, 最终获得白色固体Gramicidin A片段粗肽产品NH2-Trp-D-Leu-Trp- D-Leu-Trp-Gly-ol (8) 1.35 g, 产率80%, 纯度95%.
Fmoc-Trp(Boc)-D-Leu-Trp(Boc)-D-Leu-Trp(Boc)-Gly-ol-CBTBS (6): 1H NMR (400 MHz, Chloroform-d) δ: 7.95~7.80 (m, 4H), 7.48 (q, J=7.8 Hz, 6H), 7.30 (d, J=30.3 Hz, 14H), 6.97 (q, J=6.9 Hz, 16H), 6.57~6.53 (m, 4H), 5.09 (s, 1H), 5.06 (s, 1H), 4.34 (s, 2H), 3.97 (s, 4H), 3.68 (s, 2H), 3.54 (d, J=17.0 Hz, 2H), 3.32 (d, J=11.3 Hz, 6H), 1.41 (d, J=21.7 Hz, 32H), 0.76 (d, J=2.6 Hz, 18H), 0.68~0.56 (m, 12H), -0.04 (d, J=3.8 Hz, 12H); 13C NMR (101 MHz, Chloroform-d) δ: 173.99, 173.44, 172.27, 171.50, 154.56, 141.01, 136.66, 135.03, 130.60, 128.34, 128.05, 127.55, 126.85, 126.77, 124.98, 124.32, 122.70, 122.32, 119.68, 119.44, 117.27, 115.33, 115.21, 114.84, 83.06, 53.24, 46.64, 42.95, 27.98, 26.62, 25.46, 24.78, 20.92, 17.95, -4.61, -4.63; HRMS-ESI calcd for C102H131N9KO16Si2 [M+K+H]2+ 917.4478, found 917.4453.
NH2-Trp(Boc)-D-Leu-Trp(Boc)-D-Leu-Trp(Boc)-Gly-ol-CBTBS (7): 1H NMR (400 MHz, Chloroform-d) δ: 7.90 (d, J=48.5 Hz, 3H), 7.45 (d, J=7.6 Hz, 1H), 7.38~6.90 (m, 20H), 6.65~6.52 (m, 5H), 5.11 (s, 1H), 4.56 (s, 1H), 4.20 (s, 1H), 3.97 (s, 1H), 3.72 (s, 1H), 3.43~3.27 (m, 5H), 3.22 (s, 1H), 3.16 (s, 1H), 2.95 (d, J=11.8 Hz, 2H), 2.72 (d, J=14.4 Hz, 2H), 2.41 (s, 1H), 2.12 (s, 1H), 1.52~1.31 (m, 31H), 0.79 (s, 18H), 0.68~0.50 (m, 12H), -0.00 (s, 12H); 13C NMR (101 MHz, Chloroform-d) δ: 172.74, 155.20, 150.16, 135.72, 128.88, 128.78, 125.22, 124.64, 123.31, 122.95, 120.19, 120.10, 119.43, 115.86, 115.49, 84.81, 83.58, 83.31, 67.71, 54.04, 40.28, 30.12, 28.63, 26.10, 23.68, 23.19, 22.11, 21.74, 18.58, -3.96; HRMS-ESI calcd for C87H124N9O14Si2 [M+3H]3+ 525.2189, found 525.2217.
(S)-2-((S)-2-氨基-3-(1H-吲哚-3-基)丙酰胺基)-N- ((S)-1-(((S)-1-(((S)-1-((2-羟乙基)氨基)-3-(1H-吲哚-3-基)- 1-氧代丙-2-基)氨基)-4-甲基-1-氧代戊-2-基)氨基)- 3-(1H-吲哚-3-基)-1-氧代丙-2-基)-4-甲基戊酰胺(8): 1H NMR (400 MHz, DMSO-d6) δ: 10.92 (s, 1H), 10.79 (d, J=18.5 Hz, 2H), 8.45 (s, 1H), 8.19 (s, 1H), 8.09 (d, J=8.0 Hz, 1H), 7.99 (dd, J=26.2, 7.6 Hz, 1H), 7.89 (q, J=8.5, 7.1 Hz, 1H), 7.63 (t, J=8.8 Hz, 2H), 7.55 (d, J=8.1 Hz, 1H), 7.33 (dd, J=16.3, 8.1 Hz, 3H), 7.16 (d, J=7.1 Hz, 2H), 7.10 (s, 1H), 7.04 (t, J=7.7 Hz, 3H), 6.99~6.89 (m, 3H), 4.63 (dt, J=22.4, 6.4 Hz, 2H), 4.50 (q, J=6.9 Hz, 1H), 4.44~4.35 (m, 1H), 4.30 (h, J=8.2, 6.6 Hz, 2H), 3.78 (s, 1H), 2.96 (dd, J=14.7, 8.3 Hz, 2H), 2.86 (dt, J=14.6, 6.1 Hz, 1H), 1.11 (s, 6H); 13C NMR (101 MHz, DMSO-d6) δ: 171.90, 171.79, 171.40, 171.35, 168.49, 158.97, 158.62, 151.70, 136.59, 136.22, 135.13, 130.58, 127.63, 127.22, 125.32, 124.43, 123.85, 123.63, 121.32, 120.99, 120.98, 118.87, 118.57, 118.38, 117.65, 114.75, 111.62, 111.44, 110.28, 109.95, 107.03, 72.25, 53.33, 52.54, 51.44, 48.86, 41.45, 41.08, 40.15, 31.42, 29.63, 27.46, 26.95, 24.18, 23.33, 23.31, 21.81; HRMS-ESI calcd for C47H60N9O6 [M+H]+ 846.3867, found 846.3838.
辅助材料(Supporting Information) 化合物
a~
c、
1~
8和DBF的核磁与质谱图谱、不同试剂捕获DBF的液相图以及通过标签辅助液相与固相合成Gramicidin A片段的物料消耗表. 这些材料可以免费从本刊网站(
http:// sioc-journal.cn/)上下载.