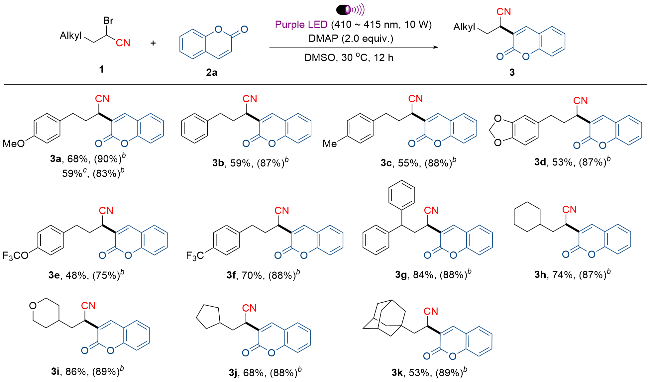
4-(4-甲氧基苯基)-2-(2-氧代-2H-苯并吡喃-3-基)丁腈(3a): 产率为68%[43.2 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率90% (263.0 mg). 白色固体, m.p. 116~118 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.88 (s, 1H), 7.56~7.50 (m, 2H), 7.32~7.29 (m, 2H), 7.11 (d, J=8.4 Hz, 2H), 6.80 (d, J,=8.8 Hz, 2H), 4.03 (dd, J=9.2 Hz, J=4.8 Hz, 1H), 3.73 (s, 3H), 2.90~2.74 (m, 2H), 2.33~2.24 (m, 1H), 2.16~2.06 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 159.5, 158.1, 153.2, 140.3, 132.1, 131.2, 129.3, 128.0, 124.8, 123.4, 119.0, 118.3, 116.4, 113.9, 55.1 (d, J=4.2 Hz), 33.4, 32.3, 32.1. HRMS (ESI) calcd for C20H18O3N [M+H]+ 320.1281, found 320.1280.
2-(2-氧代-2H-苯并吡喃-3-基)-4-苯基丁腈(3b): 产率为59% [34.0 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率87% (254.3 mg). 白色固体, m.p. 105~107 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.93 (s, 1H), 7.59~7.53 (m, 2H), 7.35 (d, J=9.2 Hz, 2H), 7.29 (t, J=7.6 Hz, 2H), 7.24~7.18 (m, 3H), 4.10~4.07 (m, 1H), 2.98~2.83 (m, 2H), 2.40~2.31 (m, 1H), 2.20~2.10 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 159.7, 153.4, 140.5 (d, J=8.6 Hz), 139.3, 132.3 (d, J=13.4 Hz), 128.8, 128.6 (d, J=8.9 Hz), 128.2 (d, J=20.0 Hz), 126.6 (d, J=35.2 Hz), 125.0 (d, J=26.9 Hz), 123.6, 119.1, 118.4, 116.7 (d, J=27.1 Hz), 33.5, 33.2, 32.6 (d, J=21.2 Hz). HRMS (ESI) calcd for C19H16O2N [M+H]+ 290.1176, found 290.1175.
2-(2-氧代-2H-苯并吡喃-3-基)-4-(对甲苯基)丁腈(3c): 产率为55% [33.4 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率88% (257.2 mg). 白色固体, m.p. 104~106 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.91 (s, 1H), 7.56 (dd, J=17.6, 8.4 Hz, 2H), 7.36~7.31 (m, 2H), 7.10 (dd, J=11.2, 8.0 Hz, 4H), 4.08 (dd, J=9.2, 4.8 Hz, 1H), 2.94~2.79 (m, 2H), 2.37~2.31 (m, 1H), 2.28 (s, 3H), 2.19~2.10 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 159.7, 153.4, 140.4 (d, J=3.7 Hz), 136.2 (d, J=9.3 Hz), 132.3 (d, J=11.4 Hz), 129.5, 129.3 (d, J=14.6 Hz), 128.4 (d, J=13.6 Hz), 128.1 (d, J=13.6 Hz), 125.0 (d, J=21.8 Hz), 123.6, 119.1, 118.4, 116.7 (d, J=21.7 Hz), 33.5, 32.7 (d, J=6.1 Hz), 32.5, 21.0 (dd, J=8.8, 4.5 Hz). HRMS (ESI) calcd for C20H18O2N [M+H]+ 304.1332, found 304.1331.
4-(苯并[
d][
1,
3]二氧杂环戊烯-5-基)-2-(2-氧代-2
H-色烯-3-基)丁腈(
3d): 产率为53% [35.2 mg, 洗脱液:
V(石油醚)∶
V(乙酸乙酯)=15∶1], 香豆素回收率87% (254.3 mg). 白色固体, m.p. 116~118 ℃;
1H NMR (400 MHz, CDCl
3)
δ: 7.90 (s, 1H), 7.58~7.52 (m, 2H), 7.35~7.30 (m, 2H), 6.72~6.65 (m, 3H), 5.88~5.87 (m, 2H), 4.06 (dd,
J=9.2, 4.8 Hz, 1H), 2.89~2.74 (m, 2H), 2.32~2.24 (m, 1H), 2.15~2.06 (m, 1H);
13C NMR (101 MHz, CDCl
3)
δ: 159.7, 153.4, 147.8, 146.2, 140.4, 133.0, 132.3, 128.1, 125.0, 123.6, 121.4, 119.1, 118.4, 116.7, 108.8, 108.4, 100.9, 33.6, 32.9, 32.4. HRMS (ESI) calcd for C
20H
16O
4N [M+H]
+ 334.1074, found 334.1072.
2-(2-氧代-2H-苯并吡喃-3-基)-4-(4-三氟甲基苯基)丁腈(3e): 产率为48% [35.9 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率75% (219.2 mg). 黄色固体, m.p. 68~70 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.93 (s, 1H), 7.57~7.52 (m, 2H), 7.35~7.30 (m, 2H), 7.23 (d, J=8.4 Hz, 2H), 7.11 (d, J=8.4 Hz, 2H), 4.09~4.05 (m, 1H), 2.97~2.82 (m, 2H), 2.38~2.29 (m, 1H), 2.18~2.09 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 159.6, 153.3, 147.8, 140.5 (d, J=7.6 Hz), 138.1, 132.3 (d, J=14.8 Hz), 129.7 (d, J=32.2 Hz), 128.1 (d, J=17.5 Hz), 125.0 (d, J=26.4 Hz), 123.3, 121.1 (d, J=34.9 Hz), 120.4 (q, J=258.1 Hz), 118.9, 118.3, 116.6 (d, J=27.4 Hz), 33.3, 32.6, 32.4; 19F NMR (376 MHz, CDCl3) δ: -57.92 (s, 3F). HRMS (ESI) calcd for C20H15O3NF3 [M+H]+ 374.0999, found 374.0998.
2-(2-氧代-2H-苯并吡喃-3-基)-4-(4-三氟甲氧基苯基)丁腈(3f): 产率为70% [49.9 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率88% (257.2 mg). 白色固体, m.p. 93~95 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.94 (s, 1H), 7.60~7.52 (m, 4H), 7.35~7.32 (m, 4H), 4.11~4.07 (m, 1H), 3.04~2.89 (m, 2H), 2.42~2.33 (m, 1H), 2.22~2.13 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 159.7, 153.4, 143.5, 140.6 (d, J=11.7 Hz), 132.6~132.4 (m), 128.9 (q, J=32.6 Hz), 128.8 (d, J=29.2 Hz), 128.2 (d, J=18.4 Hz), 125.7~125.4 (m), 125.1 (d, J=29.9 Hz), 124.1 (q, J=270.7 Hz), 123.2 (d, J=1.4 Hz), 118.9, 116.7 (d, J=30.2 Hz), 33.0 (d, J=8.5 Hz), 32.8, 32.6; 19F NMR (376 MHz, CDCl3) δ: -62.41 (s, 3F). HRMS (ESI) calcd for C20H15O2NF3 [M+H]+ 358.1049, found 358.1047.
2-(2-氧代-2H-苯并吡喃-3-基)-4,4-二苯基丁腈(3g): 产率为84% [61.1 mg, 洗脱液: V(石油醚)∶V(乙酸乙 酯)=15∶1], 香豆素回收率88% (257.2 mg). 黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 7.85 (s, 1H), 7.56~7.53 (m, 1H), 7.48 (d, J=8.0 Hz, 1H), 7.43 (d, J=7.6 Hz, 2H), 7.39~7.28 (m, 5H), 7.24 (d, J=4.4 Hz, 4H), 7.16~7.10 (m, 1H), 4.35 (dd, J=11.2, 5.2 Hz, 1H), 3.95 (dd, J=10.4, 4.4 Hz, 1H), 2.93~2.86 (m, 1H), 2.46~2.39 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 159.6, 153.3, 143.4, 141.2, 140.4 (d, J=5.6 Hz), 132.2 (d, J=15.4 Hz), 129.2, 128.9, 128.5, 128.1 (d, J=27.7 Hz), 127.7, 127.3, 127.0 (d, J=8.5 Hz), 126.5, 124.9 (d, J=30.9 Hz), 123.6, 118.4, 116.6 (d, J=30.3 Hz), 49.3 (d, J=17.4 Hz), 37.7~37.2 (m), 32.2 (d, J=22.7 Hz). HRMS (ESI) calcd for C25H20- O2N [M+H]+ 366.1489, found 366.1488.
3-环己基-2-(2-氧代-2H-苯并吡喃-3-基)丙腈(3h): 产率为74% [41.8 mg, 洗脱液: V(石油醚)∶V(乙酸乙 酯)=20∶1], 香豆素回收率87% (254.3 mg). 白色固体, m.p. 135~137 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.92 (s, 1H), 7.57~7.52 (m, 2H), 7.35~7.30 (m, 2H), 4.13 (dd, J=10.4, 4.8 Hz, 1H), 1.90 (d, J=12.4 Hz, 1H), 1.80~1.56 (m, 7H), 1.32~1.13 (m, 3H), 1.04~0.95 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 159.8, 153.3, 140.0 (d, J=6.2 Hz), 132.2 (d, J=13.3 Hz), 128.1 (d, J=16.6 Hz), 124.9 (d, J=25.0 Hz), 124.7, 119.7, 118.6, 116.6 (d,J=25.8 Hz), 40.3, 35.8, 33.5 (d, J=14.4 Hz), 31.7 (d, J=11.5 Hz), 30.3 (d, J=20.0 Hz), 26.3~25.8 (m). HRMS (ESI) calcd for C18H20O2N [M+H]+ 282.1489, found 282.1488.
2-(2-氧代-2H-苯并吡喃-3-基)-3-(四氢-2H-吡喃-4-基)丙腈(3i): 产率为86% [49.6 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=20∶1], 香豆素回收率89% (260.1 mg). 白色固体, m.p. 163~165 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.92 (s, 1H), 7.58~7.52 (m, 2H), 7.34~7.30 (m, 2H), 4.14~4.11 (m, 1H), 3.99~3.92 (m, 2H), 3.39 (t, J=11.6 Hz, 2H), 1.90~1.74 (m, 4H), 1.65 (d, J=13.2 Hz, 1H), 1.42~1.31 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 159.7, 153.3, 140.2 (d, J=3.0 Hz), 132.2 (d, J=12.2 Hz), 128.1 (d, J=14.5 Hz), 125.0 (d, J=20.9 Hz), 124.1, 119.3, 118.4, 116.6 (d, J=21.4 Hz), 67.6~67.4 (m), 39.6, 33.3~32.9 (m), 31.8~31.5 (m), 29.9 (d, J=18.4 Hz). HRMS (ESI) calcd for C17H18O3N [M+H]+ 284.1281, found 284.1280.
3-环戊基-2-(2-氧代-2H-苯并吡喃-3-基)丙腈(3j): 产率为68% [36.2 mg, 洗脱液: V(石油醚)∶V(乙酸乙 酯)=20∶1), 香豆素回收率88% (257.2 mg). 白色固体, m.p. 88~90 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.92 (s, 1H), 7.58~7.53 (m, 2H), 7.36~7.31 (m, 2H), 4.07 (t, J=7.6 Hz, 1H), 2.14~2.03 (m, 1H), 1.95~1.80 (m, 4H), 1.70~1.51 (m, 4H), 1.30~1.12 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 159.8, 153.4, 140.0 (d, J=8.6 Hz), 132.2 (d, J=14.3 Hz), 128.1 (d, J=17.1 Hz), 125.0 (d, J=27.7 Hz), 124.5, 119.7, 118.6, 116.7 (d, J=27.5 Hz), 39.1, 38.2, 33.0, 32.4, 32.2, 31.7, 25.2. HRMS (ESI) calcd for C17H18- O2N [M+H]+ 268.1332, found 268.1331.
3-((3R,5R,7R)-金刚烷-1-基)-2-(-氧代-2H-苯并吡喃- 3-基)丙腈(3k): 产率为53% [35.2 mg, 洗脱液: V(石油醚)∶V(乙酸乙 酯)=20∶1], 香豆素回收率89% (260.1 mg). 白色固体, m.p. 171~173 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.92 (s, 1H), 7.56~7.52 (m, 2H), 7.34~7.29 (m, 2H), 4.10~4.06 (m, 1H), 2.00 (s, 3H), 1.73~1.62 (m, 13H), 1.55 (dd, J=14.0, 2.4 Hz, 1H); 13C NMR (101 MHz, CDCl3) δ: 159.6, 153.3, 139.9 (d, J=7.0 Hz), 132.0 (d, J=15.0 Hz), 128.0 (d, J=17.1 Hz), 125.6, 124.9 (d, J=27.2 Hz), 121.0, 118.6, 116.6 (d, J=26.7 Hz), 47.8~47.4 (m), 42.1~41.8 (m), 36.9~36.5 (m), 33.0, 28.4 (d, J=9.7 Hz), 26.7, 26.5. HRMS (ESI) calcd for C22H24O2N [M+H]+ 334.1802, found 334.1799.
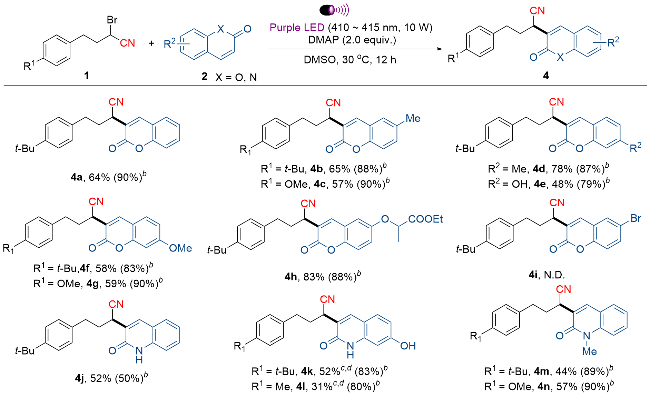
4-(4-(叔丁基)苯基)-2-(2-氧代-2H-苯并吡喃-3-基)丁腈(4a): 产率为64% [44.2 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率90% (262.7 mg). 白色固体, m.p. 84~86 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.92 (s, 1H), 7.59~7.53 (m, 2H), 7.36~7.30 (m, 4H), 7.16 (d, J=8.0 Hz, 2H), 4.12~4.09 (m, 1H), 2.96~2.81 (m, 2H), 2.40~2.32 (m, 1H), 2.22~2.12 (m, 1H), 1.28 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 159.7, 153.4, 149.4, 140.5, 136.3, 132.3, 128.1, 125.5, 124.9, 124.0, 123.6, 120.0, 118.4, 116.7, 34.4, 33.4, 32.7, 32.6, 31.4 (d, J=3.3 Hz). HRMS (DART) calced for C23H24O2N [M+H]+ 346.1802, found 346.1800.
4-(4-叔丁基苯基)-2-(6-甲基-2-氧代-2H-苯并吡喃-3-基)丁腈(4b): 产率为65% [46.8 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率88% (280.4 mg). 白色固体, m.p. 103~105 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.86 (s, 1H), 7.36 (d, J=8.8 Hz, 1H), 7.32~7.30 (m, 3H), 7.23 (d, J=8.4 Hz, 1H), 7.16 (d, J=8.0 Hz, 2H), 4.10 (dd, J=9.2, 4.8 Hz, 1H), 2.95~2.80 (m, 2H), 2.42 (s, 3H), 2.40~2.31 (m, 1H), 2.21~2.11 (m, 1H), 1.28 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 159.5, 152.1, 149.4, 140.4, 136.3, 134.7, 133.3, 128.1, 127.9 (d, J=9.4 Hz), 125.5, 123.4, 119.2, 118.2, 116.4, 34.4, 33.4, 32.7, 32.6, 31.4, 20.8 (d, J=4.4 Hz). HRMS (ESI) calcd for C24H25O2NNa [M+Na]+ 382.1778, found 382.1775.
4-(4-甲氧基苯基)-2-(6-甲基-2-氧代-2H-苯并吡喃-3-基)丁腈(4c): 产率为57% [38.2 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率90% (288.3 mg). 白色固体, m.p. 121~123 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.85 (s, 1H), 7.36 (d, J=8.4 Hz, 1H), 7.30 (s, 1H), 7.23 (d, J=8.4 Hz, 1H), 7.13 (d, J=8.4 Hz, 2H), 6.82 (d, J=8.4 Hz, 2H), 4.05 (dd, J=9.2, 4.8 Hz, 1H), 3.76 (s, 3H), 2.91~2.76 (m, 2H), 2.42 (s, 3H), 2.34~2.25 (m, 1H), 2.14~2.05 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 161.1, 158.2, 151.5, 140.4, 134.7, 133.3, 131.3, 129.4, 127.9, 123.4, 119.2, 118.2, 116.4, 114.0, 55.2, 33.7, 32.5, 32.3, 20.8. HRMS (DART) calcd for C21H20O3N [M+H]+ 334.1438, found 334.1436.
4-(4-叔丁基苯基)-2-(7-甲基-2-氧代-2H-苯并吡喃-3-基)丁腈(4d): 产率为78% [56.0 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率87% (278.7 mg). 白色固体, m.p. 87~89 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.88 (s, 1H), 7.41 (d, J=8.4 Hz, 1H), 7.31 (d, J=8.4 Hz, 2H), 7.17~7.13 (m, 4H), 4.11~4.07 (m, 1H), 2.95~2.80 (m, 2H), 2.46 (s, 3H), 2.39~2.30 (m, 1H), 2.21~2.11 (m, 1H), 1.29 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 160.0, 153.5, 149.3, 143.7, 140.4 (d, J=3.2 Hz), 136.3, 128.2, 127.8 (d, J=24.4 Hz), 126.2 (d, J=27.0 Hz), 125.5 (d, J=26.7 Hz), 122.3, 119.3, 116.8 (d, J=29.1 Hz), 116.0, 34.4, 33.4, 32.6 (d, J=6.7 Hz), 32.4, 31.3 (dd, J=26.3, 13.2 Hz), 21.9 (d, J=12.7 Hz). HRMS (DART) calcd for C24H26O2N [M+H]+ 360.1958, found 360.1957.
4-(4-(叔丁基)苯基)-2-(7-羟基-2-氧代-2H-苯并吡喃-3-基)丁腈(4e): 产率为48% [34.6 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=3∶1], 香豆素回收率79% (257.2 mg). 黄色固体, m.p. 183~185 ℃; 1H NMR (400 MHz, CD3OD) δ: 7.88 (s, 1H), 7.45 (d, J=8.4 Hz, 1H), 7.26 (d, J=8.0 Hz, 2H), 7.10 (d, J=8.4 Hz, 2H), 6.80~6.77 (m, 1H), 6.68 (d, J=2.0 Hz, 1H), 4.01~3.98 (m, 1H), 2.87~2.70 (m, 2H), 2.26~2.16 (m, 2H), 1.24 (s, 9H); 13C NMR (101 MHz, CD3OD) δ: 163.1, 161.8, 156.4, 150.0, 142.6, 137.9, 130.7, 128.9, 126.2, 120.4, 119.3, 114.6, 112.5, 103.0, 34.9, 33.9, 33.4, 33.1, 31.6. HRMS (ESI) calcd for C23H23O3NNa [M+Na]+ 384.1570, found 384.1568.
4-(4-(叔丁基)苯基)-2-(7-甲氧基-2-氧代-2H-苯并吡喃-3-基)丁腈(4f): 产率为58% [43.2 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率83% (292.6 mg). 白色固体, m.p. 97~99 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.84 (s, 1H), 7.42 (d, J=8.8 Hz, 1H), 7.31 (d, J=8.4 Hz, 2H), 7.15 (d, J=6.8 Hz, 2H), 6.89 (dd, J=8.4, 2.4 Hz, 1H), 6.82 (d, J=2.4 Hz, 1H), 4.08~4.05 (m, 1H), 3.88 (s, 3H), 2.94~2.79 (m, 2H), 2.37~2.28 (m, 1H), 2.19~2.10 (m, 1H), 1.29 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 163.2, 160.1, 155.3, 149.3, 140.9, 137.5, 129.0 (d, J=7.2 Hz), 128.1, 125.6 (d, J=4.8 Hz), 119.8, 119.4, 113.2 (d, J=4.7 Hz), 112.1, 100.6 (d, J=4.0 Hz), 55.9 (d, J=11.2 Hz), 34.4, 33.5, 32.6, 32.5 (d, J=10.4 Hz), 31.4. HRMS (ESI) calcd for C24H26O3N [M+H]+ 376.1907, found 376.1906.
2-(7-甲氧基-2-氧代-2H-苯并吡喃-3-基)-4-(4-甲氧基苯基)丁腈(4g): 产率为59% [41.4 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率90% (317.1 mg). 黄色固体, m.p. 112~114 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.83 (s, 1H), 7.41 (d, J=8.8 Hz, 1H), 7.13 (d, J=8.8 Hz, 2H), 6.88 (dd, J=8.8, 2.4 Hz, 1H), 6.83~6.81 (m, 3H), 4.04~4.00 (m, 1H), 3.88 (s, 3H), 3.76 (s, 3H), 2.90~2.75 (m, 2H), 2.34~2.24 (m, 1H), 2.14~2.05 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 162.7, 160.1, 158.2, 155.3, 140.5, 131.4, 129.4 (d, J=8.0 Hz), 129.1 (d, J=8.2 Hz), 119.8, 119.4, 114.0, 113.2 (d, J=6.0 Hz), 112.1, 100.6 (d, J=5.2 Hz), 55.9 (d, J=12.2 Hz), 55.2 (d, J=10.8 Hz), 33.8, 32.4~32.3 (m), 29.7. HRMS (ESI) calcd for C21H20O4N [M+H]+ 350.1387, found 350.1386.
2-((3-(3-(4-叔丁基苯基)-1-氰基丙基)-2-氧代-2H-苯并吡喃-6-基)氧基)丙酸乙酯(4h): 产率为83% [76.5 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=10∶1], 香豆素回收率88% (461.6 mg). 黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 7.84 (d, J=1.6 Hz, 1H), 7.43 (d, J=8.4 Hz, 1H), 7.30 (d, J=8.4 Hz, 2H), 7.15 (d, J=8.0 Hz, 2H), 6.91~6.87 (m, 1H), 6.76~6.74 (m, 1H), 4.81 (dd, J=13.6, 6.4 Hz, 1H), 4.26~4.21 (m, 2H), 4.05 (dd, J=9.2, 4.8 Hz, 1H), 2.93~2.78 (m, 2H), 2.36~2.27 (m, 1H), 2.18~2.08 (m, 1H), 1.67 (d, J=6.8 Hz, 3H), 1.28 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 171.0, 161.0, 159.9, 655155.0, 149.3, 140.3, 136.3, 129.2, 128.0, 125.5, 120.3, 119.3, 113.6 (d, J=12.8 Hz), 112.6, 101.7 (d, J=11.0 Hz), 72.8, 61.7, 34.3, 33.4 (d, J=3.0 Hz), 32.6, 32.4 (d, J=3.8 Hz), 31.3, 18.3, 14.1. HRMS (ESI) calcd for C28H32O5N [M+H]+ 462.2275, found 462.2270.
4-(4-叔丁基苯基)-2-(2-氧代-1,2-二氢喹啉-3-基)丁腈(4j): 产率为52% [35.5 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=13∶1], 香豆素回收率50% (144.3 mg). 白色固体, m.p. 203~205 ℃; 1H NMR (400 MHz, CDCl3) δ: 12.85 (s, 1H), 8.05 (s, 1H), 7.64 (d, J=8.0 Hz, 1H), 7.58 (t, J=8.4 Hz, 1H), 7.45 (d, J=8.4 Hz, 1H), 7.34 (d, J=8.4 Hz, 2H), 7.29 (d, J=8.0 Hz, 1H), 7.22 (d, J=8.0 Hz, 2H), 4.35 (dd, J=9.2, 4.8 Hz, 1H), 2.99~2.89 (m, 2H), 2.49~2.40 (m, 1H), 2.28~2.19 (m, 1H), 1.31 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 162.5, 149.3, 138.3 (d, J=5.9 Hz), 138.0, 136.8, 131.1 (d, J=6.7 Hz), 128.3, 128.1, 127.5, 125.6, 125.4 (d, J=5.8 Hz), 123.3 (d, J=15.0 Hz), 120.2, 119.5, 116.0 (d, J=12.8 Hz), 34.5, 33.8, 32.7, 31.9 (d, J=7.8 Hz), 31.4 (d, J=7.8 Hz); HRMS (ESI) calcd for C23H25ON2 [M+H]+ 345.1961, found 345.1962.
4-(4-(叔丁基)苯基)-2-(7-羟基-2-氧代-1,2-二氢喹啉-3-基)丁腈(4k): 产率为52% [37.4 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=1∶1], 香豆素回收率83% (267.5 mg). 黄色固体, m.p. >220 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 11.84 (s, 1H), 10.25 (s, 1H), 7.92 (s, 1H), 7.54 (d, J=8.4 Hz, 1H), 7.29 (d, J=8.0 Hz, 2H), 7.14 (d, J=8.0 Hz, 2H), 6.72 (d, J=2.0 Hz, 1H), 6.68~6.66 (m, 1H), 4.11 (dd, J=8.4, 6.4 Hz, 1H), 2.76~2.63 (m, 2H), 2.18~2.10 (m, 2H), 1.24 (s, 9H); 13C NMR (101 MHz, DMSO-d6) δ: 160.7, 160.0, 148.4, 140.3, 137.5, 137.2, 129.7 (d, J=16.1 Hz), 128.0 (d, J=25.9 Hz), 125.2 (d, J=24.3 Hz), 122.8, 120.7, 112.6, 111.5, 99.6 (d, J=8.9 Hz), 34.1, 33.3, 32.2, 31.2 (d, J=10.2 Hz), 30.7 (d, J=18.6 Hz). HRMS (ESI) calcd for C23H25O2N2 [M+H]+ 361.1911, found 361.1909.
2-(7-羟基-2-氧代-1,2-二氢喹啉-3-基)-4-(对甲苯基)丁腈(4l): 产率为31% [19.8 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=1∶1], 香豆素回收率80% (257.8 mg). 黄色固体, m.p. 112~114 ℃; 1H NMR (400 MHz, DMSO- d6) δ: 11.84 (s, 1H), 10.26 (s, 1H), 7.92 (s, 1H), 7.54 (d, J=8.4 Hz, 1H), 7.08 (s, 4H), 6.72 (s, 1H), 6.68 (d, J=8.0 Hz, 1H), 4.10~4.06 (m, 1H), 2.76~2.61 (m, 2H), 2.24 (s, 3H), 2.17~2.06 (m, 2H); 13C NMR (101 MHz, DMSO-d6) δ: 160.7, 160.0, 140.3, 137.5 (d, J=7.1 Hz), 137.1, 135.2, 129.8 (d, J=19.3 Hz), 129.1 (d, J=35.2 Hz), 128.2 (d, J=34.9 Hz), 122.8, 120.7, 112.2 (d, J=8.7 Hz), 112.1, 99.7 (d, J=13.8 Hz), 32.8, 32.3, 30.7 (d, J=21.4 Hz), 20.7. HRMS (ESI) calcd for C20H19O2N2 [M+H]+ 319.1441, found 319.1440.
4-(4-9叔丁基)苯基)-2-(1-甲基-2-氧代-1,2-二氢喹啉-3-基)丁腈(4m): 产率为44% [31.7 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=8∶1], 香豆素回收率89% (284.6 mg); 黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 7.93 (s, 1H), 7.63~7.59 (m, 2H), 7.38 (d, J=8.0 Hz, 1H), 7.29 (t, J=8.4 Hz, 3H), 7.17 (d, J=8.0 Hz, 2H), 4.31 (dd, J=9.2 Hz, J=4.8 Hz, 1H), 3.75 (s, 3H), 2.95~2.81 (m, 2H), 2.40~2.31 (m, 1H), 2.17~2.08 (m, 1H), 1.29 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 160.4, 149.2, 139.4, 136.8, 136.4, 131.1, 129.0, 128.1, 127.5, 125.4, 122.7, 120.3, 119.7, 114.2, 34.4, 33.8, 32.8, 32.6, 31.4, 30.0. HRMS (ESI) calcd for C24H27ON2 [M+H]+ 359.2118, found 359.2117.
4-(4-甲氧基苯基)-2-(1-甲基-2-氧代-1,2-二氢喹啉-3-基)丁腈(4n): 产率为57% [37.9 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率90% (286.6 mg). 黄色固体, m.p. 103~105 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.92 (s, 1H), 7.62~7.58 (m, 2H), 7.37 (d, J=8.8 Hz, 1H), 7.28 (t, J=7.6 Hz, 1H), 7.15 (d, J=8.8 Hz, 2H), 6.82 (d, J=8.4 Hz, 2H), 4.28~4.24 (m, 1H), 3.76 (s, 3H), 3.74 (s, 3H), 2.92~2.78 (m, 2H), 2.35~2.26 (m, 1H), 2.13~2.04 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.4, 158.1, 139.4, 136.4, 131.8, 131.0, 129.4, 129.0, 127.5, 122.7, 120.3, 119.7, 114.2, 113.9, 55.2 (d, J=5.4 Hz), 34.1, 32.5, 30.0, 29.9. HRMS (ESI) calcd for C21H21- O2N2 [M+H]+ 333.1598, found 333.1596.
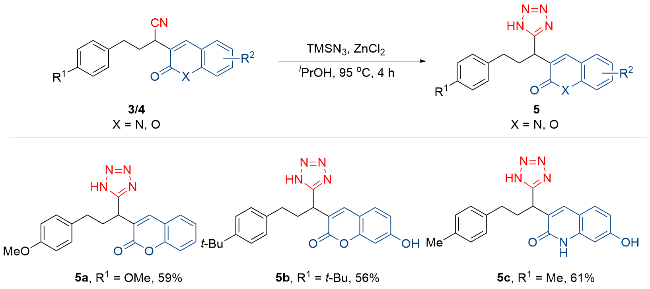
3-[3-(4-甲氧基苯基)-1-(1H-四唑-5-基)丙基]-2H-苯并吡喃-2-酮(5a): 产率为59% (42.8 mg). 白色固体, m.p. 95~97 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.88 (s, 1H), 7.56~7.50 (m, 2H), 7.32~7.29 (m, 2H), 7.11 (d, J=8.4 Hz, 2H), 6.80 (d, J=8.8 Hz, 2H), 4.03 (dd, J=9.2, 4.8 Hz, 1H), 3.73 (s, 3H), 2.90~2.74 (m, 2H), 2.33~2.24 (m, 1H), 2.16~2.06 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 159.5, 158.1, 153.2, 140.3, 132.1, 131.2, 129.3, 128.0, 124.8, 123.4, 119.0, 118.3, 116.4, 113.9, 55.1 (d, J=4.2 Hz), 33.4, 32.3, 32.1. HRMS (FI) calcd for C20H18O3N4Na [M+Na]+ 385.1271, found 385.1267.
3-[3-(4-(叔丁基)苯基)-1-(1H-四唑-5-基)丙基]-7-羟基-2H-苯并吡喃-2-酮(5b): 产率为56% (45.3 mg). 黄色固体, m.p. 176~178 ℃; 1H NMR (400 MHz, CD3OD) δ: 7.89 (s, 1H), 7.45 (d, J=8.4 Hz, 1H), 7.25 (d, J=8.4 Hz, 2H), 7.09 (d, J=8.4 Hz, 2H), 6.80 (d, J=8.8 Hz, 1H), 6.68 (s, 1H), 4.01~3.97 (m, 1H), 2.87~2.69 (m, 2H), 2.24~2.16 (m, 2H), 1.23 (s, 9H); 13C NMR (101 MHz, CD3OD) δ: 163.1, 161.8, 156.4, 150.0, 142.8, 137.9, 130.7, 128.9, 126.2, 120.4, 119.3, 114.6, 112.5, 103.0, 34.9, 33.8, 33.4, 33.1, 31.6. HRMS (FI) calcd for C23H24O3N4Na [M+Na]+ 427.1741, found 427.1739.
3-[1-(1H-四唑-5-基)-3-(对甲苯基)丙基]-7-羟基喹啉-2(1H)-酮(5c): 产率为61% (44.2 mg). 黄色固体, m.p.>220 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 11.83 (s, 1H), 10.29 (s, 1H), 7.91 (s, 1H), 7.54 (d, J=8.4 Hz, 1H), 7.08 (s, 4H), 6.72 (s, 1H), 6.68 (dd, J=8.4, 2.4 Hz, 1H), 4.09~4.05 (m, 1H), 2.76~2.61 (m, 2H), 2.23 (s, 3H), 2.17~2.06 (m, 2H); 13C NMR (101 MHz, DMSO-d6) δ: 160.8, 160.0, 140.3, 137.6, 137.2, 135.2, 129.8, 129.1, 128.3, 122.8, 120.7, 112.3, 112.1, 99.7, 32.8, 32.3, 30.7, 20.7. HRMS (FI) calcd for C20H19O2N5Na [M+Na]+ 384.1431, found 384.1426.
4-(4-(叔丁基)苯基)-2-(2-氧代-2H-苯并吡喃-3-基)丁酸甲酯(8a): 产率为52% [39.8 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率89% (260.1 mg). 白色固体, m.p. 78~80 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.67 (s, 1H), 7.53~7.46 (m, 2H), 7.34~7.28 (m, 4H), 7.13 (d, J=8.0 Hz, 2H), 3.92~3.88 (m, 1H), 3.72 (s, 3H), 2.69~2.65 (m, 2H), 2.45~2.36 (m, 1H), 2.22~2.13 (m, 1H), 1.29 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 173.2, 161.0, 153.2, 149.0, 139.9 (d, J=6.1 Hz), 137.7, 131.4 (d, J=5.2 Hz), 128.2~128.0 (m), 127.8 (d, J=9.2 Hz), 126.9, 125.4~125.3 (m), 124.5 (d, J=11.5 Hz), 119.1, 116.5 (d, J=11.0 Hz), 52.4 (d, J=12.2 Hz), 45.0 (d, J=13.7 Hz), 34.4, 33.1, 32.8, 31.4 (d, J=5.9 Hz). HRMS (ESI) calcd for C24H27O4 [M+H]+ 379.1904, found 379.1902.
4-(4-叔丁基苯基)-2-(7-甲氧基-2-氧代-2H-苯并吡喃-3-基)丁酸甲酯(8b): 产率为28% [22.7 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率87% (306.5 mg). 黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 7.61 (s, 1H), 7.36 (d, J=8.4 Hz, 1H), 7.29 (d, J=8.0 Hz, 2H), 7.12 (d, J=8.0 Hz, 2H), 6.84 (d, J=8.4 Hz, 1H), 6.81 (s, 1H), 3.88~3.84 (m, 4H), 3.71 (s, 3H), 2.68~2.64 (m, 2H), 2.42~2.33 (m, 1H), 2.19~2.10 (m, 1H), 1.29 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 173.5, 162.5, 161.4, 154.9, 148.9, 140.1, 139.9, 137.8, 128.8 (d, J=25.6 Hz), 128.1 (d, J=30.0 Hz), 125.3 (d, J=27.1 Hz), 123.2, 112.8~112.6 (m), 100.5 (d, J=21.6 Hz), 55.8 (d, J=22.2 Hz), 52.3 (d, J=19.1 Hz), 44.7 (d, J=20.2 Hz), 34.4, 33.3, 33.0 (d, J=17.1 Hz), 31.4 (dd, J=27.8, 14.1 Hz). HRMS (ESI) calcd for C25H29O5 [M+H]+ 409.2010, found 409.2008.
4-(4-(叔丁基)苯基)-2-(2-氧代-1,2-二氢喹啉-3-基)丁酸甲酯(8c): 产率为12% [17.1 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=15∶1], 香豆素回收率89% (258.4 mg). 白色固体, m.p. 158~160 ℃; 1H NMR (400 MHz, CDCl3) δ: 12.46 (s, 1H), 7.80(s, 1H), 7.56 (d, J=8.0 Hz, 1H), 7.53~7.49 (m, 1H), 7.42 (d, J=8.0 Hz, 1H), 7.30 (d, J=8.0 Hz, 2H), 7.23 (t, J=8.0 Hz, 1H), 7.15 (d, J=8.0 Hz, 2H), 4.13~4.10 (m, 1H), 3.71 (s, 3H), 2.70 (t, J=8.0 Hz, 2H), 2.50~2.41 (m, 1H), 2.29~2.20 (m, 1H), 1.30 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 174.2, 163.6, 148.8, 138.3, 137.8, 137.7, 131.1, 130.3, 128.2, 127.7, 125.3, 122.7, 120.0, 116.0, 52.1, 44.4, 34.4, 33.2, 33.1, 31.4. HRMS (ESI) calcd for C24H28O3N [M+H]+ 378.2064, found 378.2062.
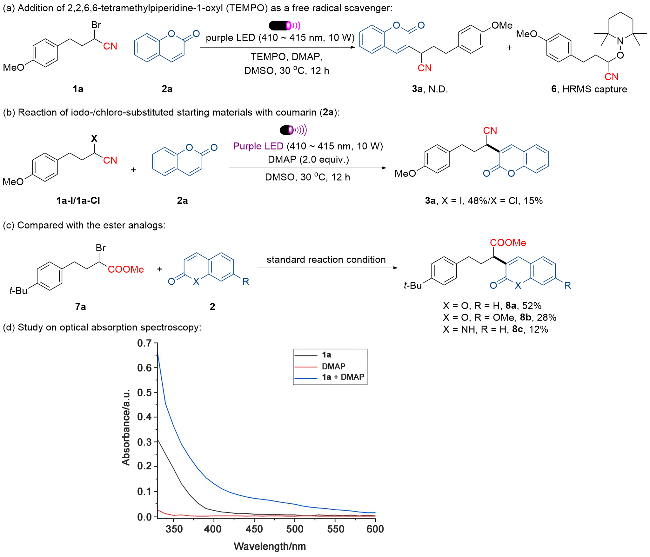
4-(4-甲氧基苯基)丁腈(1a-H): 产率为22% [3.8 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=14∶1]. 无色油状液体. 1H NMR (400 MHz, CDCl3) δ: 7.10 (d, J=8.0 Hz, 2H), 6.85 (d, J=8.0 Hz, 2H), 3.80 (s, 3H), 2.74~2.70 (m, 2H), 2.32~2.29 (m, 2H), 1.98~1.91 (m, 2H). HRMS (ESI) calcd for C11H13ONNa [M+Na]+ 198.0889, found 198.0888.
2,3-双(4-甲氧基苯乙基)丁二腈(1a-HC): 产率为15% [5.2 mg, 洗脱液: V(石油醚)∶V(乙酸乙酯)=6∶1]. 浅黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 7.06 (d, J=8.0 Hz, 4H), 6.84 (d, J=8.0 Hz, 4H), 3.79 (s, 6H), 2.88~2.83 (m, 2H), 2.72~2.60 (m, 4H), 2.25~2.16 (m, 2H), 1.94~1.85 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 158.5, 130.8, 129.4, 118.1, 114.3, 55.3, 33.3, 32.4, 31.9. HRMS (ESI) calcd for C22H24O2N2Na [M+Na]+ 371.1730, found 371.1722.
辅助材料(Supporting Information) 合成的化合物
3~
8,
1a-H和
1a-HC的
1H NMR、
13C NMR、
19F NMR和HRMS谱图. 这些材料可以免费从本刊网站(
http://sioc- journal.cn/)上下载.