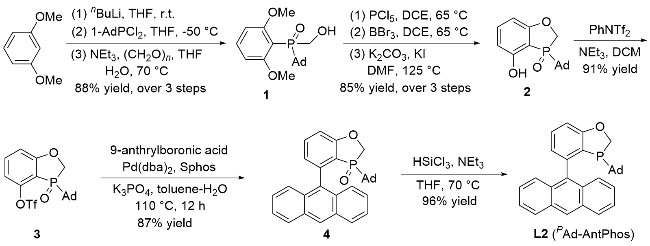
取一干净的250 mL三口烧瓶, 加入1,3-二甲氧基苯(11 g, 80 mmol), 抽换氮气三次, 加入四氢呋喃(100 mL), 在-60 ℃下加入正丁基锂(1.0 equiv.), 加完后缓慢升至室温, 继续搅拌反应8 h, 反应完成后将锂盐溶液在-40 ℃以下加入金刚烷基二氯化磷的四氢呋喃溶液, 搅拌10 min. 在冰浴下加入水(50 mL), 恢复至室温搅拌反应1 h, 加入三乙胺(24 g, 240 mmol, 3.0 equiv.)和多聚甲醛(6.0 g, 400 mmol, 5 equiv.), 加热至回流, 反应12 h. 反应完全后加入浓盐酸, 调节pH到2~3, 分液, 水层用二氯甲烷(30 mL×3)萃取, 合并有机相, 饱和食盐水(100 mL)洗涤, 无水硫酸钠干燥, 浓缩得灰白色粉末状固体. 然后用乙酸乙酯(20 mL)和石油醚(100 mL)混合溶液结晶, 得到金刚烷基(2,6-二甲氧基苯基)(羟甲基)膦氧(1): 白色固体(24.6 g, 88% yield), m.p. 191~194 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.39 (t, J=8.3 Hz, 1H), 6.54 (d, J=8.3 Hz, 2H), 4.31 (d, J=13.8 Hz, 1H), 3.94 (d, J=12.1 Hz, 1H), 3.79 (s, 6H), 3.44 (s, 1H), 1.94 (s, 3H), 1.85 (s, 6H), 1.72~1.62 (m, 6H); 13C NMR (126 MHz, CDCl3) δ: 163.29 (s), 134.41 (s), 104.66, 104.27 (d, J=280 Hz), 104.25 (d, J=6.1 Hz), 37.83 (d, J=68.9 Hz), 36.48 (s), 34.69 (s), 27.35 (d, J=10.5 Hz); 31P NMR (161 MHz, CDCl3) δ: 43.19 (s); IR ν: 3171, 2901, 2843, 1470, 1427, 1252, 1153, 1105, 1051, 777, 522 cm-1; ESI-MS m/z: 350.90 [M+H]+, 372.85 [M+Na]+; HRMS (ESI) calcd for C19H28O4P [M+H]+ 351.1720, found 351.1722; HRMS (ESI) calcd for C19H27O4PNa [M+Na]+ 373.1539, found 373.1539.
取一干净的100 mL史莱克管加入化合物4 (0.8 g)后, 抽换氮气三次, 依次加入四氢呋喃(10 mL)、三乙胺(1 mL)和三氯硅烷(0.4 mL). 体系在70 ℃下加热反应6 h, 利用31P NMR检测反应. 反应完成后在0 ℃下加入氢氧化钠溶液(30%, 15 mL), 反应体系在65 ℃下搅拌2 h. 加入甲叔醚, 用甲叔醚(15 mL×3)萃取, 合并有机相, 饱和食盐水(50 mL)洗涤, 无水硫酸钠干燥, 柱层析[洗涤剂: V(乙酸乙酯):V(乙醚)=1:10, 250 mL]得到PAd- Ant-Phos (L2): 743 mg, 96% yield. 1H NMR (500 MHz, CDCl3) δ: 8.49 (s, 1H), 8.07~7.98 (m, 2H), 7.92 (d, J=8.5 Hz, 1H), 7.80 (d, J=8.5 Hz, 1H), 7.48 (t, J=7.4 Hz, 2H), 7.45~7.36 (m, 3H), 7.14~7.01 (m, 2H), 4.94 (d, J=12.4 Hz, 1H), 4.53 (dd, J=26.2, 12.5 Hz, 1H), 1.45~1.37 (m, 4H), 1.28 (d, J=12.1 Hz, 5H), 1.06 (d, J=11.6 Hz, 3H), 0.94 (d, J=11.6 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 164.20 (s), 142.34 (d, J=17.1 Hz), 136.53 (s), 131.35 (d, J=14.6 Hz), 130.92 (s), 130.83 (s), 128.91 (s), 128.43 (d, J=28.1 Hz), 128.14 (s), 127.22 (s), 127.12 (s), 127.07 (s), 125.53 (s), 125.32 (s), 125.04 (d, J=3.9 Hz), 124.88 (s), 68.84 (d, J=26.5 Hz), 37.93 (d, J=10.9 Hz), 36.29 (s), 34.01 (d, J=20.3 Hz), 30.32 (s), 28.08 (d, J=8.3 Hz); 31P NMR (162 MHz, CDCl3) δ: -18.72 (s); IR ν: 2901, 2845, 1583, 1562, 1556, 1462, 1443, 1410, 1350, 1303, 1283, 1219, 1011, 970, 961, 933, 845, 883, 845, 802, 785, 735, 644, 630, 601, 596, 556, 511, 496 cm-1; ESI-MS m/z: 449.00 [M+H]+; HRMS (ESI) calcd for C31H30OP [M+H]+ 449.1762, found 449.1765.
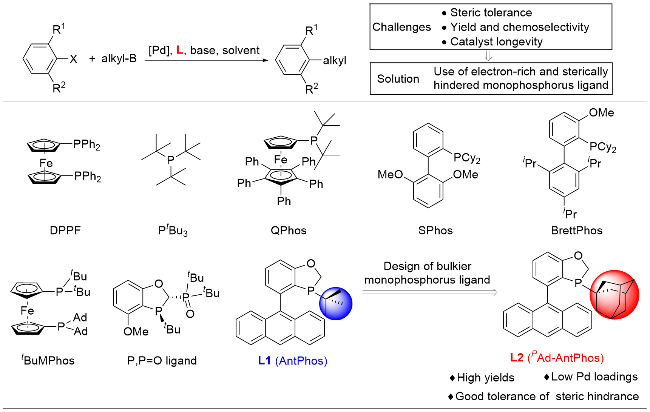
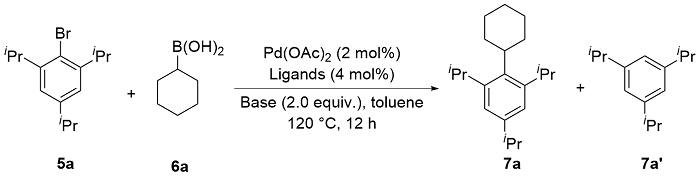
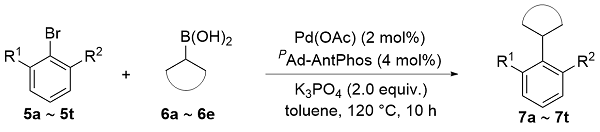
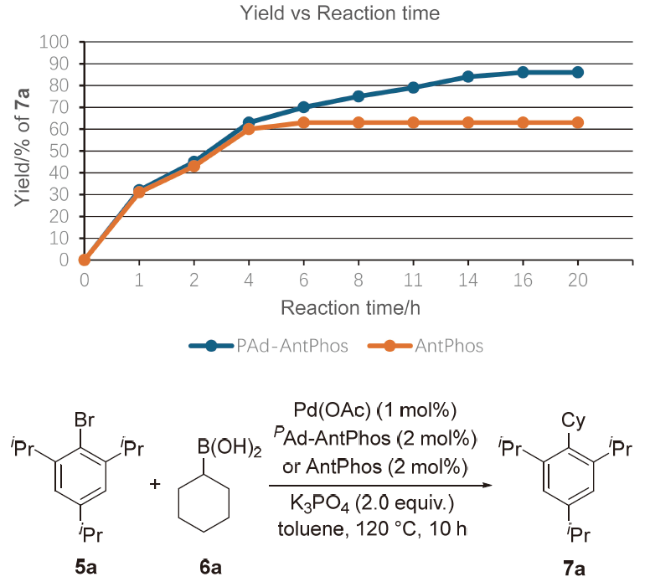
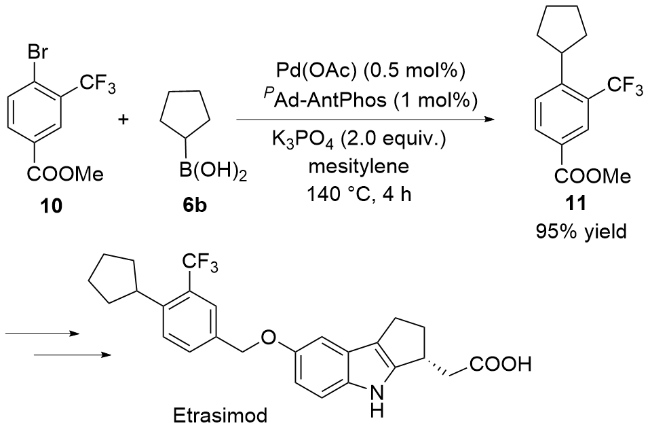
偶联反应操作方法: 取一干净的25 mL史莱克管, 依次加入芳基溴化物5a~5t (0.2 mmol)、环烷基硼酸6a~6e (0.4 mmol, 50.4 mg, 2.0 equiv.)、磷酸钾(85 mg, 0.4 mmol, 2.0 equiv.)、醋酸钯(0.9 mg, 0.004 mmol, 2 mol%)和配体(0.008 mmol, 4 mol%), 抽换氮气三次, 加入甲苯(2 mL), 反应体系在120 ℃下加热反应10 h, 反应结束后柱层析分离得到7a~7t.
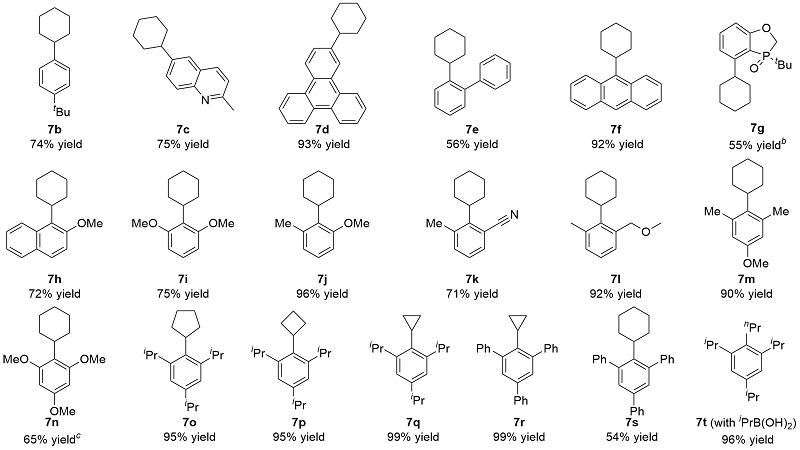
2-环己基-1,3,5-三异丙基苯(
7a)
[8]: 无色液体(49.7 mg, 87% yield).
1H NMR (500 MHz, CDCl
3)
δ: 7.01~6.90 (m, 2H), 3.57 (dt,
J=13.5, 6.8 Hz, 1H), 3.23 (dt,
J=13.5, 6.7 Hz, 1H), 3.09 (t,
J=12.2 Hz, 1H), 2.85 (ddt,
J=20.8, 13.8, 6.8 Hz, 2H), 1.96~1.79 (m, 4H), 1.74 (t,
J=16.6 Hz, 2H), 1.36 (dd,
J=21.6, 9.4 Hz, 2H), 1.31~1.20 (m, 19H);
13C NMR (126 MHz, CDCl
3)
δ: 148.62 (s), 147.97 (s), 145.47 (s), 138.12 (s), 122.82 (s), 122.03 (s), 39.13 (s), 34.23 (s), 33.82 (s), 31.94 (s), 29.50 (s), 27.87 (s), 26.22 (s), 24.99 (s), 24.16 (d,
J=13.4 Hz), 23.94 (s).
4-环己基-1-叔丁基苯(
7b)
[20]: 无色液体(32 mg, 74% yield).
1H NMR (500 MHz, CDCl
3)
δ: 7.31 (d,
J=7.4 Hz, 2H), 7.14 (d,
J=7.4 Hz, 2H), 2.47 (t,
J=9.3 Hz, 1H), 1.85 (d,
J=14.2 Hz, 4H), 1.74 (d,
J=12.4 Hz, 1H), 1.47~1.36 (m, 4H), 1.35~1.18 (m, 10H);
13C NMR (126 MHz, CDCl
3)
δ: 148.38 (s), 144.99 (s), 126.37 (s), 125.08 (s), 43.95 (s), 34.38 (d,
J=19.2 Hz), 31.41 (s), 26.95 (s), 26.19 (s).
2-甲基-6-环己基喹啉(7c): 无色液体(34 mg, 75% yield). 1H NMR (500 MHz, CDCl3) δ: 8.00~7.95 (m, 1H), 7.93 (d, J=8.4 Hz, 1H), 7.81 (dd, J=71.3, 9.0 Hz, 1H), 7.59~7.52 (m, 1H), 7.27~7.22 (m, 1H), 2.72 (s, 3H), 2.66 (t, J=11.4 Hz, 1H), 1.96 (d, J=12.5 Hz, 2H), 1.88 (d, J=12.4 Hz, 2H), 1.78 (d, J=13.1 Hz, 1H), 1.47 (tt, J=25.5, 12.6 Hz, 3H), 1.34~1.25 (m, 1H); 13C NMR (126 MHz, CDCl3) δ: 135.89 (s), 135.12 (s), 132.80 (s), 130.39 (s), 129.69 (s), 128.28 (s), 124.00 (s), 122.84 (s), 121.82 (s), 44.37 (s), 34.39 (s), 26.86 (s), 26.15 (s), 25.24 (s); ESI-MS m/z: 226.00 [M+H]+; HRMS (ESI) calcd for C18H20N [M+H]+ 226.1590, found 226.1589.
2-环己基苯并菲(
7d)
[21]: 黄色固体(58 mg, 93% yield), m.p. 127~129 ℃;
1H NMR (500 MHz, CDCl
3)
δ: 8.73~8.61 (m, 4H), 8.58 (d,
J=8.4 Hz, 1H), 8.48 (s, 1H), 7.69~7.62 (m, 5H), 7.54 (d,
J=8.4 Hz, 1H), 2.78 (t,
J=11.9 Hz, 1H), 2.05 (d,
J=12.4 Hz, 2H), 1.94 (d,
J=12.6 Hz, 2H), 1.83 (d,
J=12.1 Hz, 1H), 1.63 (dd,
J=25.0, 12.5 Hz, 2H), 1.54~1.44 (m, 2H), 1.43~1.33 (m, 1H);
13C NMR (126 MHz, CDCl
3)
δ: 146.97 (s), 129.91 (s), 129.89 (s), 129.87 (s), 129.77 (s), 129.70 (s), 129.42 (s), 127.87 (s), 127.20 (s), 127.12 (s), 127.02 (s), 127.00 (s), 126.73 (s), 126.48 (s), 123.29 (s), 123.22 (s), 123.10 (s), 121.10 (s), 44.92 (s), 34.61 (s), 26.98 (s), 26.23 (s).
2-环己基-1,1'-联苯(7
e)
[22]: 无色液体(43.4 mg, 92% yield).
1H NMR (500 MHz, CDCl
3)
δ: 7.46~7.38 (m, 3H), 7.38~7.31 (m, 3H), 7.28 (d,
J=7.0 Hz, 1H), 7.20 (q,
J=7.5 Hz, 2H), 2.65 (t,
J=12.0 Hz, 1H), 1.74 (d,
J=11.3 Hz, 4H), 1.65 (d,
J=10.7 Hz, 1H), 1.44 (dd,
J=24.7, 12.4 Hz, 2H), 1.25~1.09 (m, 3H);
13C NMR (126 MHz, CDCl
3)
δ: 133.07 (s), 130.01 (s), 129.30 (d,
J=13.2 Hz), 127.91 (s), 127.43 (s), 126.62 (s), 126.31 (s), 125.23 (s), 39.96 (s), 34.63 (s), 26.75 (s), 26.13 (s).
9-环己基蒽(
7f)
[8]: 黄色固体(51 mg, 99% yield), m.p. 121~124 ℃;
1H NMR (500 MHz, CDCl
3)
δ: 8.70 (s, 1H), 8.44 (s, 1H), 8.34 (s, 1H), 8.01 (d,
J=8.4 Hz, 2H), 7.60~7.35 (m, 4H), 4.15 (t,
J=12.6 Hz, 1H), 2.63~2.49 (m, 2H), 2.04 (d,
J=12.6 Hz, 2H), 1.96 (d,
J=12.5 Hz, 3H), 1.70~1.54 (m, 3H);
13C NMR (126 MHz, CDCl
3)
δ: 139.36 (s), 129.58 (d,
J=21.0 Hz), 128.16 (s), 126.41 (s), 126.19 (s), 125.33 (s), 124.48 (s), 123.84 (s), 40.42 (s), 31.97 (s), 27.94 (s), 26.41 (s).
3-(叔丁基)-4-环己基-2
H-苯并[
d][
1,
3]氧代膦-3-氧化物(
7g): 白色固体(32 mg, 55% yield), m.p. 135~137 ℃;
1H NMR (500 MHz, CDCl
3)
δ: 7.49 (dd,
J=7.5, 2.0 Hz, 1H), 7.10 (t,
J=7.9 Hz, 1H), 6.91 (dd,
J=8.2, 2.6 Hz, 1H), 4.62 (d,
J=13.8 Hz, 1H), 4.39 (dd,
J=13.6, 10.7 Hz, 1H), 3.99 (t,
J=9.2 Hz, 1H), 2.05 (d,
J=11.1 Hz, 2H), 1.84 (s, 1H), 1.78 (d,
J=5.5 Hz, 2H), 1.64 (dd,
J=20.8, 11.1 Hz, 2H), 1.36 (dd,
J=24.5, 11.5 Hz, 3H), 1.29 (d,
J=16.3 Hz, 9H);
13C NMR (126 MHz, CDCl
3)
δ: 165.77 (d,
J=18.3 Hz), 135.57 (s), 133.60 (d,
J=7.6 Hz), 113.77 (d,
J=5.1 Hz), 109.99 (s), 94.38 (d,
J=5.1 Hz), 66.46 (s), 65.97 (s), 60.32 (s), 36.65 (s), 35.21 (s), 25.09 (s), 24.85 (s);
31P NMR (376 MHz, CDCl
3)
δ: 49.61; ESI-MS
m/
z: 293.20 [M+H]
+; HRMS (ESI) calcd for C
17H
26PO
2 [M+H]
+ 293.1665, found 293.1673.
1-环己基-2-甲氧基萘(
7h)
[8]: 白色固体(41 mg, 85% yield), m.p. 67~69 ℃;
1H NMR (500 MHz, CDCl
3)
δ: 8.24~8.09 (m, 1H), 7.78 (d,
J=8.1 Hz, 1H), 7.71 (d,
J=8.9 Hz, 1H), 7.46 (t,
J=6.3 Hz, 1H), 7.33 (t,
J=7.3 Hz, 1H), 7.28~7.24 (m, 1H), 3.93 (s, 3H), 3.54~3.31 (m, 1H), 2.26 (d,
J=11.4 Hz, 2H), 1.89 (d,
J=11.2 Hz, 2H), 1.81 (d,
J=10.3 Hz, 1H), 1.71 (d,
J=8.7 Hz, 2H), 1.53~1.33 (m, 3H).
2-环己基-1,3-二甲氧基苯(
7i)
[8]: 白色蜡状固体(33 mg, 75% yield), m.p. 97~98 ℃;
1H NMR (500 MHz, CDCl
3)
δ: 7.09 (t,
J=8.3 Hz, 1H), 6.54 (d,
J=8.3 Hz, 2H), 3.79 (s, 6H), 3.21 (t,
J=12.2 Hz, 1H), 2.05 (q,
J=12.6 Hz, 2H), 1.77 (d,
J=12.6 Hz, 2H), 1.70 (d,
J=11.9 Hz, 1H), 1.52 (s, 1H), 1.44~1.20 (m, 4H);
13C NMR (126 MHz, CDCl
3)
δ: 126.47 (s), 123.81 (s), 104.63 (s), 55.77 (s), 35.00 (s), 30.01 (s), 27.46 (s), 26.35 (s).
2-环己基-3-甲基苯甲醚(7j): 无色液体(39 mg, 96% yield). 1H NMR (500 MHz, CDCl3) δ: 7.03 (t, J=7.9 Hz, 1H), 6.74 (t, J=8.2 Hz, 2H), 3.78 (s, 3H), 2.91~2.74 (m, 1H), 2.33 (s, 3H), 2.08 (dd, J=23.6, 12.2 Hz, 2H), 1.80 (d, J=11.6 Hz, 2H), 1.71 (d, J=8.6 Hz, 1H), 1.55 (s, 2H), 1.39~1.27 (m, 3H); 13C NMR (126 MHz, CDCl3) δ: 162.65 (s), 133.65 (s), 126.06 (s), 109.62 (s), 55.18 (s), 29.77 (s), 27.56 (s), 26.30 (s), 21.01 (s); HRMS (EI) calcd for C14H20O 204.1507, found 204.1509.
2-环己基-3-甲基苯甲腈(7k): 无色液体(28 mg, 71% yield). 1H NMR (500 MHz, CDCl3) δ: 7.47 (d, J=7.6 Hz, 1H), 7.33 (d, J=7.6 Hz, 1H), 7.13 (t, J=7.7 Hz, 1H), 3.01 (s, 1H), 2.39 (s, 3H), 2.17 (s, 2H), 1.89 (d, J=6.5 Hz, 2H), 1.75 (s, 1H), 1.68 (d, J=12.6 Hz, 2H), 1.38 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 148.62 (s), 136.98 (s), 135.16 (s), 132.98 (s), 125.93 (s), 120.03 (s), 77.28 (s), 77.03 (s), 76.77 (s), 30.37 (s), 27.16 (s), 25.64 (s), 20.79 (s), 14.19 (s); HRMS (EI) calcd for C14H17N 199.1357, found 199.1356.
2-环己基-1-甲氧甲基-3-甲基苯(7l): 无色液体(40 mg, 92% yield). 1H NMR (500 MHz, CDCl3) δ: 7.12 (t, J=7.9 Hz, 1H), 6.91 (d, J=7.3 Hz, 1H), 6.84 (d, J=8.2 Hz, 1H), 4.46 (s, 2H), 3.80 (s, 3H), 3.37 (s, 3H), 2.80 (dd, J=19.5, 9.7 Hz, 1H), 2.17~2.04 (m, 2H), 1.80 (d, J=11.7 Hz, 2H), 1.71 (d, J=8.7 Hz, 1H), 1.42~1.19 (m, 5H); 13C NMR (126 MHz, CDCl3) δ: 126.20 (s), 122.08 (s), 111.74 (s), 77.25 (s), 77.00 (s), 76.74 (s), 73.65 (s), 57.88 (s), 55.23 (s), 29.99 (s), 27.48 (s), 26.27 (s); HRMS (EI) calcd for C15H22O 218.1671, found 218.1673.
3,5-二甲基-4-环己基苯甲醚(7m): 无色液体(39 mg, 92% yield). 1H NMR (500 MHz, CDCl3) δ: 6.54 (s, 2H), 3.75 (s, 3H), 2.91 (t, J=11.0 Hz, 1H), 2.39 (d, J=54.8 Hz, 6H), 1.87 (dd, J=17.9, 12.8 Hz, 4H), 1.75 (d, J=12.2 Hz, 1H), 1.65 (d, J=12.2 Hz, 2H), 1.44~1.32 (m, 2H), 1.31~1.19 (m, 1H); 13C NMR (126 MHz, CDCl3) δ: 156.65 (s), 137.54 (s), 135.61 (s), 113.79 (s), 54.96 (s), 40.95 (s), 30.71 (s), 27.79 (s), 26.39 (s), 22.37 (s); HRMS (EI) calcd for C15H22O 218.1671, found 218.1672.
2-环己基-1,3,5-三甲氧基苯(7n): 白色固体(33 mg, 65% yield), m.p. 125~127 ℃; 1H NMR (500 MHz, CDCl3) δ: 6.13 (s, 2H), 3.79 (s, 3H), 3.77 (s, 6H), 3.10 (t, J=12.2 Hz, 1H), 1.99 (q, J=12.6 Hz, 2H), 1.76 (d, J=12.7 Hz, 2H), 1.50 (d, J=12.1 Hz, 2H), 1.39~1.30 (m, 2H), 1.26 (t, J=12.8 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 158.75 (s), 116.47 (s), 91.32 (s), 55.75 (s), 55.20 (s), 34.64 (s), 30.38 (s), 27.54 (s), 26.35 (s); ESI-MS m/z: 251.00 [M+H]+; HRMS (ESI) calcd for C15H23O3 [M+H]+ 251.1642, found 251.1647.
2-环戊基-1,3,5-三异丙基苯(
7o)
[8]: 白色固体(52 mg, 95% yield), m.p. 55~57 ℃;
1H NMR (500 MHz, CDCl
3)
δ: 6.99 (s, 2H), 3.63~3.54 (m, 1H), 3.24 (dt,
J=13.4, 6.6 Hz, 2H), 2.86 (dt,
J=13.7, 6.9 Hz, 1H), 1.98 (d,
J=8.5 Hz, 4H), 1.84~1.68 (m, 4H), 1.25 (d,
J=6.7 Hz, 18H);
13C NMR (126 MHz, CDCl
3)
δ: 147.07 (s), 145.65 (s), 136.66 (s), 121.81 (s), 38.62 (s), 33.93 (s), 33.44 (s), 29.64 (s), 27.13 (s), 24.54 (s), 23.99 (s).
2-环丁基-1,3,5-三异丙基苯(
7p)
[8]: 无色液体(49 mg, 95% yield).
1H NMR (500 MHz, CDCl
3)
δ: 6.98 (s, 2H), 4.23~4.14 (m, 1H), 3.52 (dt,
J=13.4, 6.7 Hz, 2H), 2.86 (tt,
J=13.9, 6.9 Hz, 2H), 2.55~2.42 (m, 2H), 2.37 (dd,
J=18.8, 9.5 Hz, 2H), 2.15~2.06 (m, 1H), 2.01 (dd,
J=20.6, 9.9 Hz, 1H), 1.35~1.19 (m, 18H);
13C NMR (126 MHz, CDCl
3)
δ: 147.60 (s), 145.96 (s), 135.86 (s), 121.68 (s), 34.90 (s), 34.26 (s), 33.98 (s), 30.66 (s), 29.44 (s), 24.71 (s), 24.00 (s), 23.11 (s), 17.69 (s).
2-环丙基-1,3,5-三异丙基苯(
7q)
[8]: 无色液体(48 mg, 99% yield).
1H NMR (500 MHz, CDCl
3)
δ: 6.97 (s, 2H), 3.87 (dt,
J=13.6, 6.7 Hz, 2H), 2.88 (dt,
J=13.7, 6.8 Hz, 1H), 1.67 (dd,
J=13.9, 8.0 Hz, 1H), 1.26 (dd,
J=14.2, 6.9 Hz, 18H), 1.06 (d,
J=8.0 Hz, 2H), 0.59 (d,
J=4.2 Hz, 2H);
13C NMR (126 MHz, CDCl
3)
δ: 149.38 (s), 146.68 (s), 133.93 (s), 120.89 (s), 34.05 (s), 28.87 (s), 24.50 (s), 24.03 (s), 10.37 (s), 8.47 (s).
2'-环丙基-5'-苯基-1,1':3',1''-三联苯(7r): 白色固体(68 mg, 99% yield), m.p. 60~62 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.67 (d, J=7.3 Hz, 2H), 7.54 (d, J=5.5 Hz, 5H), 7.44 (q, J=7.3 Hz, 5H), 7.41~7.31 (m, 3H), 2.04 (t, J=5.9 Hz, 1H), 0.35 (d, J=7.7 Hz, 2H), -0.16 (d, J=5.3 Hz, 2H); 13C NMR (126 MHz, CDCl3) δ: 145.30 (s), 142.62 (s), 140.57 (s), 139.09 (s), 137.03 (s), 129.49 (s), 128.74 (s), 127.84 (s), 127.28 (s), 127.10 (s), 126.62 (s), 14.43 (s), 10.42 (s); IR ν: 1492, 1028, 885, 758, 748, 731, 694, 609, 578, 522, 501 cm-1. HRMS (EI) calcd for C19H22 346.1711, found 346.1617.
2'-环己基-5'-苯基-1,1':3',1''-三联苯(7s): 白色固体(21 mg, 52% yield), m.p. 71~72 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.80 (d, J=10.1 Hz, 1H), 7.69 (t, J=7.2 Hz, 4H), 7.54~7.50 (m, 2H), 7.47 (t, J=7.3 Hz, 4H), 7.43~7.33 (m, 5H), 7.30 (d, J=6.2 Hz, 1H), 2.80 (t, J=11.7 Hz, 1H), 1.83 (d, J=12.1 Hz, 2H), 1.76 (d, J=9.1 Hz, 2H), 1.67 (s, 1H), 1.27~1.18 (m, 4H); 13C NMR (126 MHz, CDCl3) δ: 141.32 (s), 141.04 (s), 130.06 (s), 128.82 (s), 127.66 (s), 127.43 (s), 127.33 (s), 127.24 (s), 127.04 (s), 126.49 (s), 125.37 (s), 124.45 (s), 40.19 (s), 34.76 (s), 26.80 (s), 26.13 (s); IR ν: 1595, 1495, 1410, 1074, 1030, 1074, 1030, 874, 750, 692, 627, 621, 611, 594, 570, 536, 504, 466, 449, 439 cm-1; HRMS (EI) calcd for C30H28 388.2191, found 388.2192.
1,3,5-三异丙基-2-正丙基苯(
7t)
[23]: 无色油状液体(35 mg, 96% yield).
1H NMR (500 MHz, CDCl
3)
δ: 7.12 (t,
J=8.3 Hz, 1H), 6.55 (d,
J=8.5 Hz, 1H), 3.81 (s, 3H), 2.66~2.58 (m, 1H), 1.51 (dd,
J=15.1, 7.5 Hz, 1H), 0.95 (t,
J=7.0 Hz, 2H).