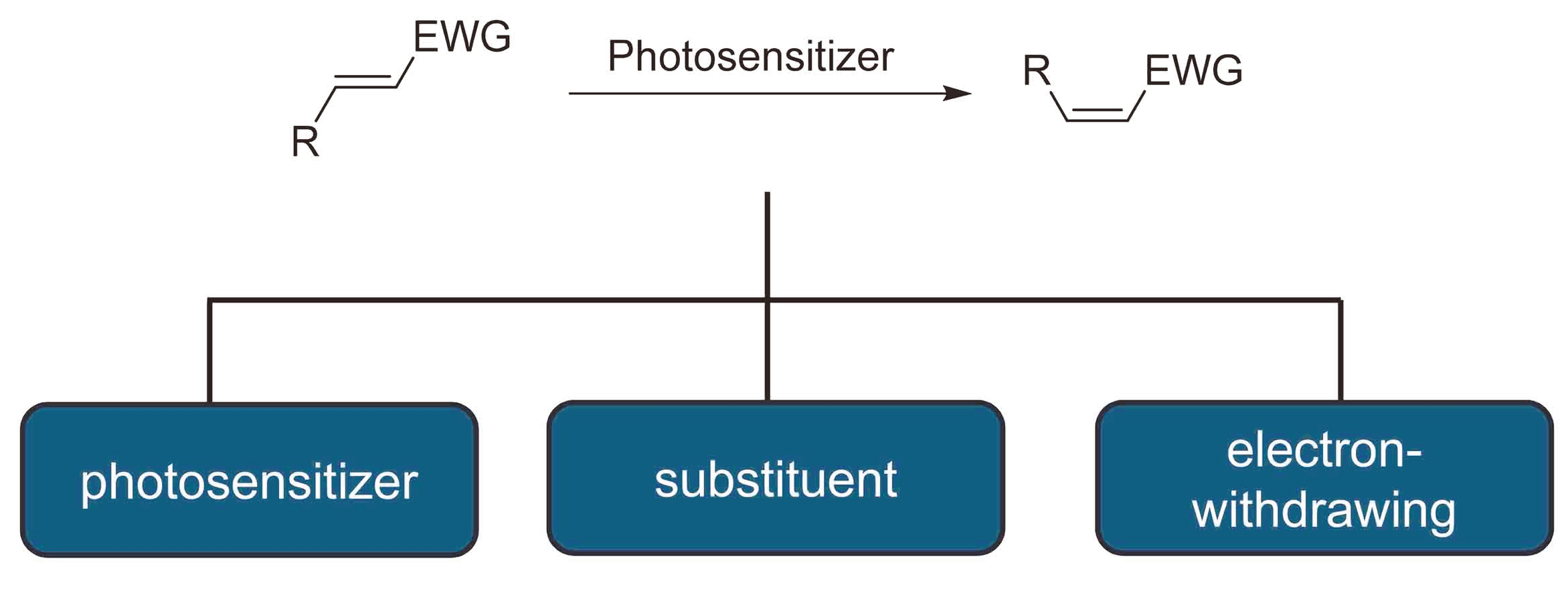
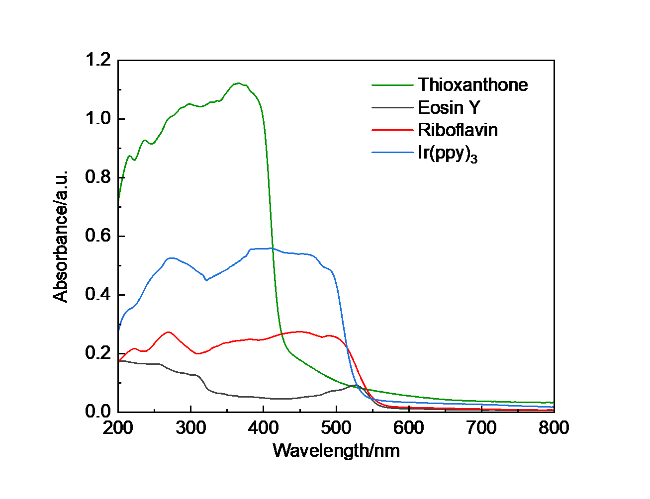
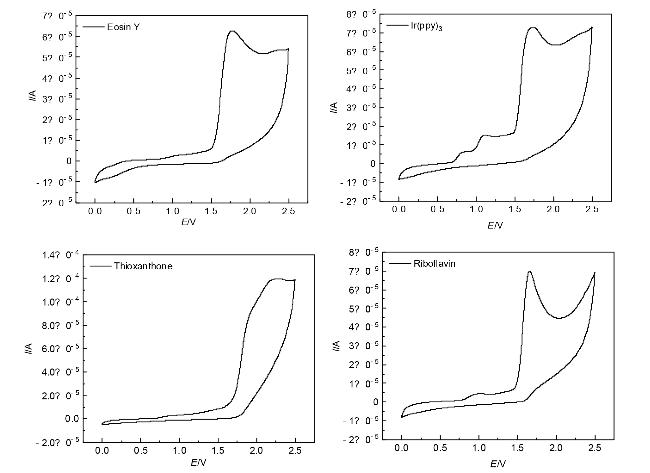
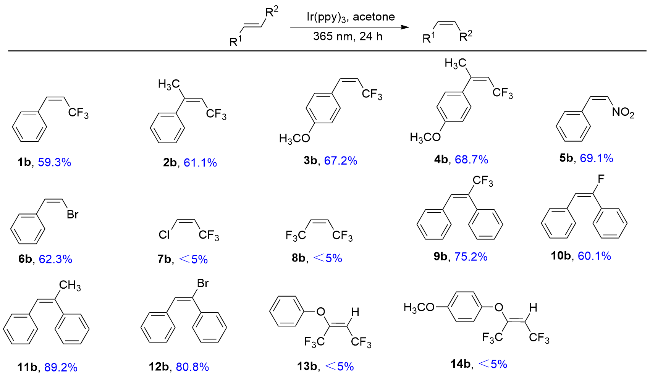
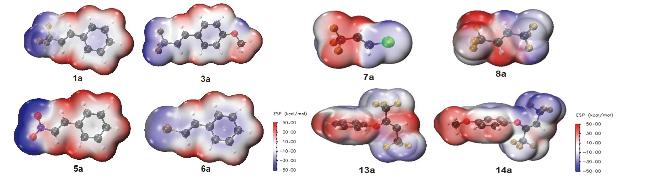
1 结果与讨论
表1 光催化异构化合成顺式-β-三氟甲基苯乙烯及其衍生物a,bTable 1 Photocatalytic isomerization synthesis of cis-β-trifluo- romethylstyrene and its derivatives  |
| Substrate | Photosensitizer | λ/nm | Conversion rate/%/% |
|---|---|---|---|
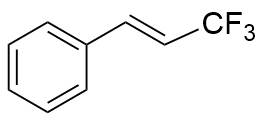 | Riboflavin | 365 | 34.9 |
| 455 | 33.2 | ||
| Ir(ppy)3 | 365 | 59.3 | |
| 455 | 51.8 | ||
| Eosin Y | 365 | 25.1 | |
| 455 | 20.3 | ||
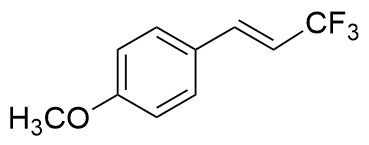 | Riboflavin | 365 | 38.7 |
| 455 | 35.1 | ||
| Ir(ppy)3 | 365 | 67.2 | |
| 455 | 60.3 | ||
| Eosin Y | 365 | 24.9 | |
| 455 | 20.1 |
a Reaction conditions (unless otherwise specified): substrate (1.0 mmol, 1.0 equiv.), Ir(ppy)3 (2 mol%), solvent (15.0 mL), 30 ℃, 25 W LEDs, b NMR yield determined by 19F NMR. |
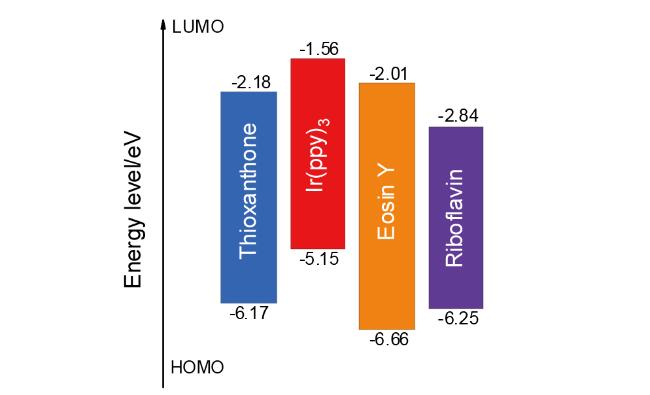
表2 光敏剂的激发能垒Table 2 Excited energy barrier of photosensitizers |
| Compd. | S0/a.u. | T1/a.u. | Excited energy barrier/(kJ•mol-1) |
|---|---|---|---|
| Ir(ppy)3 | -2518.7198827 | -2518.6319118 | 230.96 |
| Riboflavin | -1330.7519889 | -1330.72582644 | 208.76 |
| Thioxanthone | -973.8742644 | -973.7974316 | 201.72 |
| Eosin Y | -11440.2303443 | -11440.1591998 | 186.79 |
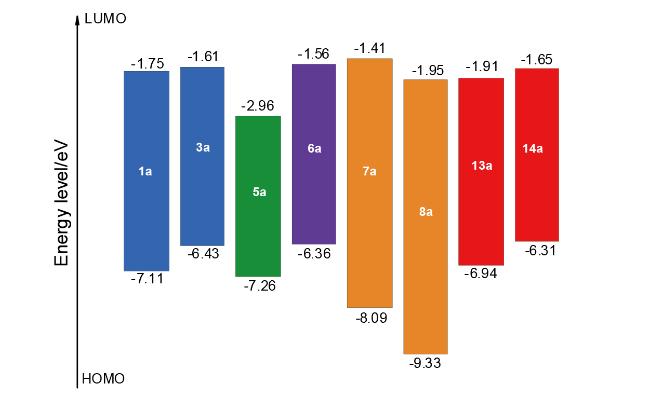
表3 底物的激发能垒Table 3 Excited energy barrier of substrates |
| Compd. | S0/a.u. | T1/a.u. | Excited energy barrier/(kJ•mol-1) |
|---|---|---|---|
| 1a | -646.9630444 | -646.867191653 | 251.66 |
| 3a | -761.5343183 | -761.444961102 | 234.61 |
| 5a | -514.3580475 | -514.283814466 | 194.90 |
| 6a | -2883.3464238 | -2883.25594811 | 237.54 |
| 7a | -875.4448938 | -875.307474280 | 360.79 |
| 8a | -753.0124878 | -752.867234534 | 381.36 |
| 13a | -1059.3803693 | -1059.25534541 | 328.25 |
| 14a | -1173.9495601 | -1173.82929702 | 315.75 |