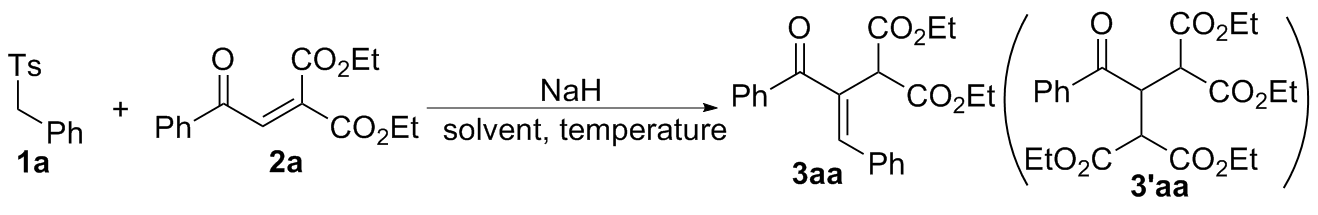
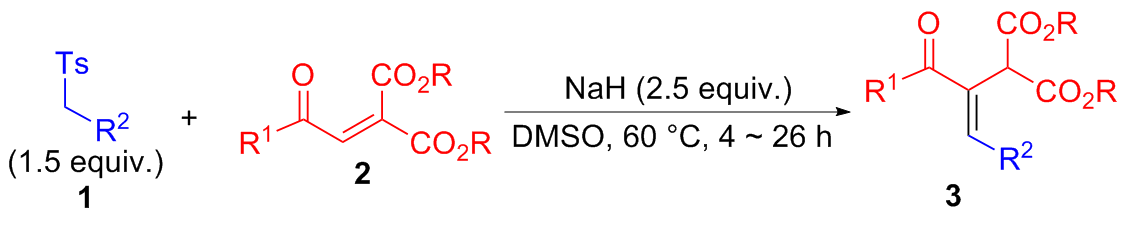
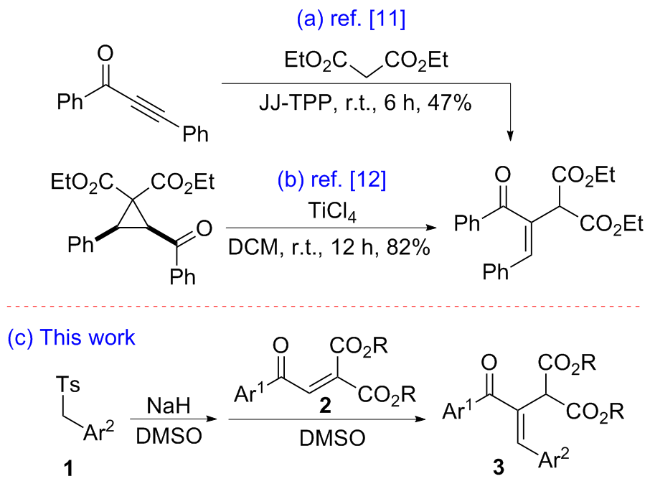
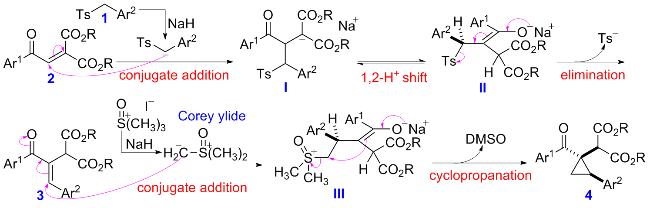
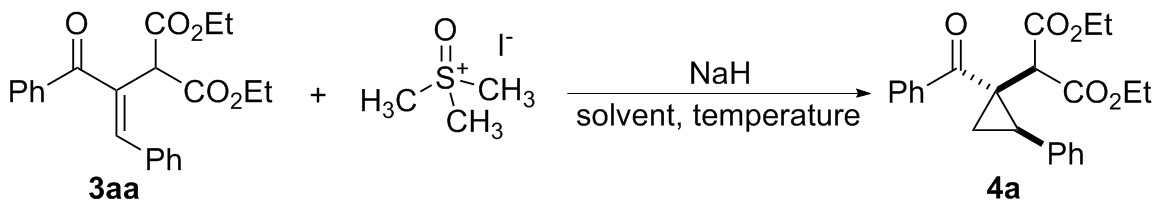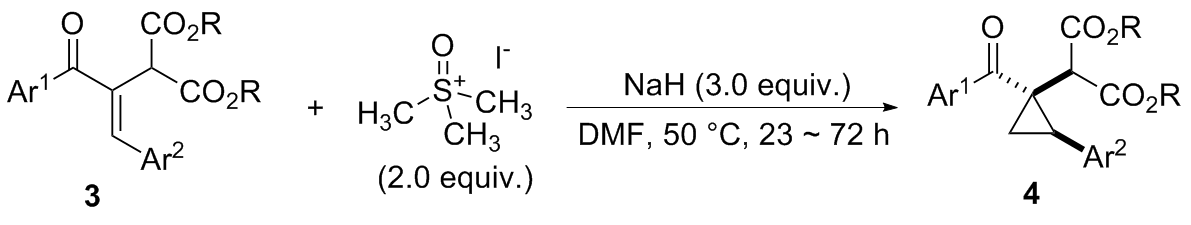在经无水无氧处理过的反应管中加4.5 mmol芳甲基对甲基苯基砜1, 20 mL无水DMSO和7.5 mmol氢化钠(质量分数为60%), 于室温下搅拌0.5 h, 再加入3 mmol 2-芳甲酰基甲亚基丙二酸二酯2, 然后将该反应管置于60 ℃的金属浴中搅拌反应, TLC跟踪至化合物2消失, 冷却至室温并加入30 mL饱和氯化铵水溶液淬灭反应, 用乙酸乙酯萃取(100 mL×3), 合并有机相, 用饱和氯化钠水溶液洗涤有机相(20 mL×3), 无水硫酸钠干燥, 过滤, 回收乙酸乙酯, 柱层析分离得到纯的产物3.
(
E)-2-(3-氧代-1,3-二苯基丙-1-烯-2-基)丙二酸二乙酯(
3aa)
[11-12,20]: 柱层析[
V(石油醚)∶
V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率90%.
1H NMR (CDCl
3, 500 MHz)
δ: 7.90~7.82 (m, 2H), 7.61~7.54 (m, 1H), 7.51~7.45 (m, 2H), 7.44~7.34 (m, 6H), 4.88 (s, 1H), 4.30~4.16 (m, 4H), 1.25 (t,
J=7.1 Hz, 6H);
13C NMR (125 MHz, CDCl
3)
δ: 196.7, 167.7, 144.1, 137.5, 134.4, 134.3, 132.2, 129.9, 129.2, 128.8, 128.3, 61.9, 51.5, 14.0; HRMS (ESI) calcd for C
22H
23O
5 [M+H]
+ 367.1540, found 367.1548.
2-苯甲酰基丙烷-1,1,3,3-四羧酸四乙酯(
3'aa)
[21]: 柱层析[
V(石油醚)∶
V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率10%.
1H NMR (CDCl
3, 500 MHz)
δ: 8.07~7.98 (m, 2H), 7.60~7.52 (m, 1H), 7.50~7.42 (m, 2H), 4.85 (t,
J=8.0 Hz, 1H), 4.23~4.12 (m, 4H), 4.06 (d,
J=8.0 Hz, 2H), 4.03~3.90 (m, 4H), 1.23 (t,
J=7.2 Hz, 6H), 1.10 (t,
J=7.2 Hz, 6H);
13C NMR (125 MHz, CDCl
3)
δ: 199.0, 167.9, 167.7, 136.9, 133.4, 129.1, 128.6, 62.0, 52.9, 43.8, 14.0, 13.8.
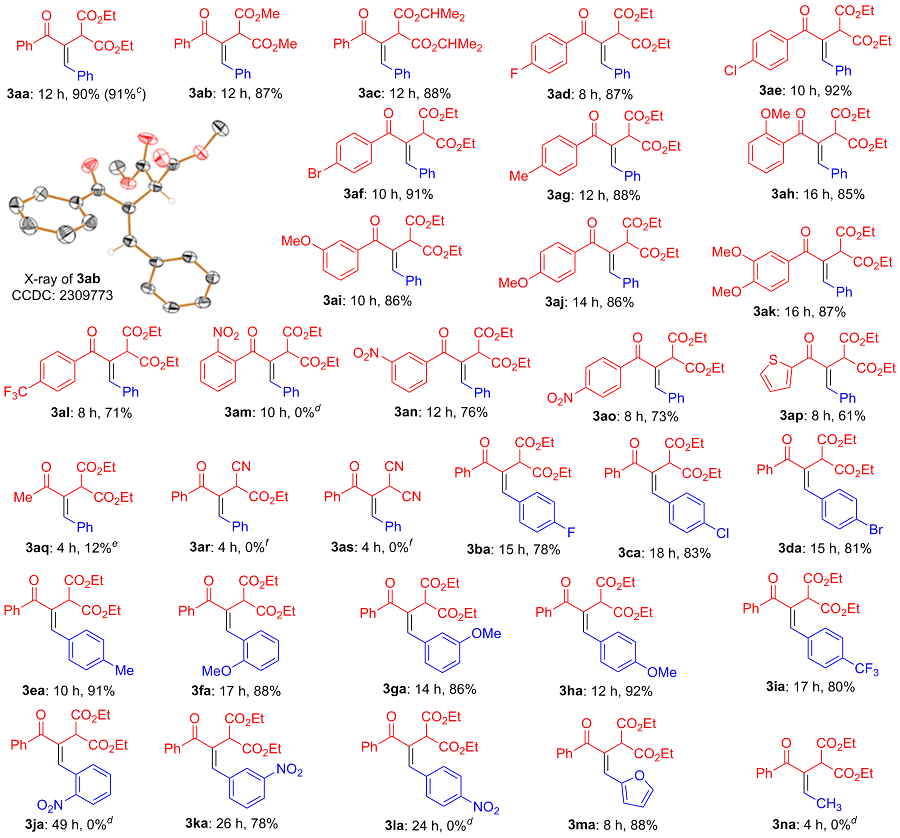
(E)-2-(3-氧代-1,3-二苯基丙-1-烯-2-基)丙二酸二甲酯(3ab): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色固体, 产率87%. m.p. 114~115 ℃; 1H NMR (CDCl3, 500 MHz) δ: 7.89~7.81 (m, 2H), 7.61~7.54 (m, 1H), 7.51~7.46 (m, 2H), 7.45~7.35 (m, 6H), 4.92 (s, 1H), 3.77 (s, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.7, 168.2, 144.4, 137.4, 134.2, 134.1, 132.3, 129.9, 129.4, 128.9, 128.8, 128.4, 53.0, 51.1; HRMS (ESI) calcd for C20H19O5 [M+H]+ 339.1227, found 339.1226.
(E)-2-(3-氧代-1,3-二苯基丙-1-烯-2-基)丙二酸二异丙酯(3ac): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率88%. 1H NMR (500 MHz, CDCl3) δ: 7.91~7.82 (m, 2H), 7.60~7.52 (m, 1H), 7.50~7.33 (m, 8H), 5.18~5.05 (m, 2H), 4.82 (s, 1H), 1.28 (d, J=6.3 Hz, 6H), 1.23 (d, J=6.3 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.4, 167.1, 143.7, 137.6, 134.7, 134.5, 134.2, 132.0, 129.7, 129.1, 128.8, 128.7, 128.1, 69.4, 51.8, 21.5; HRMS (ESI) calcd for C24H27O5 [M+H]+ 395.1853, found 395.1858.
(E)-2-(3-(4-氟苯基)-3-氧代-1-苯基丙-1-烯-2-基)丙二酸二乙酯(3ad): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率87%. 1H NMR (500 MHz, CDCl3) δ: 7.86~7.84 (m, 2H), 7.48~7.36 (m, 5H), 7.33 (s, 1H), 7.20~7.12 (m, 2H), 4.88 (s, 1H), 4.33~4.15 (m, 4H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 195.3, 167.7, 165.3 (d, 1JFC=253.7 Hz), 143.5, 134.3, 134.1, 133.7 (d, 4JFC=2.8 Hz), 132.5 (d, 3JFC=9.1 Hz), 129.3, 128.9, 128.8, 115.5 (d, 2JFC=21.9 Hz), 62.0, 51.5, 14.0; 19F NMR (470 MHz, CDCl3) δ: -106.3; HRMS (ESI) calcd for C22H22FO5 [M+H]+ 385.1446, found 385.1451.
(E)-2-(3-(4-氯苯基)-3-氧代-1-苯基丙-1-烯-2-基)丙二酸二乙酯(3ae): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色固体, 产率92%. m.p. 71~72 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.87~7.76 (m, 2H), 7.50~7.36 (m, 7H), 7.33 (s, 1H), 4.88 (s, 1H), 4.32~4.14 (m, 4H), 1.26 (t, J=7.2 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 195.5, 167.7, 143.9, 138.7, 135.8, 134.2, 134.0, 131.3, 129.4, 128.9, 128.8, 128.7, 62.0, 51.4, 14.0; HRMS (ESI) calcd for C22H22ClO5 [M+H]+ 401.1150, found 401.1147.
(E)-2-(3-(4-溴苯基)-3-氧代-1-苯基丙-1-烯-2-基)丙二酸二乙酯(3af): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率91%. 1H NMR (500 MHz, CDCl3) δ: 7.80~7.68 (m, 2H), 7.67~7.58 (m, 2H), 7.48~7.36 (m, 5H), 7.33 (s, 1H), 4.87 (s, 1H), 4.32~4.14 (m, 4H), 1.26 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 195.7, 167.6, 144.0, 136.3, 134.2, 134.0, 131.6, 131.4, 129.4, 128.9, 128.8, 127.3, 62.0, 51.4, 14.0; HRMS (ESI) calcd for C22H22BrO5 [M+H]+ 445.0645, found 445.0642.
(E)-2-(3-氧代-1-苯基-3-(p-甲基苯基)丙-1-烯-2-基)丙二酸二乙酯(3ag): 柱层析[V(石油醚)∶V(乙酸乙 酯)=15∶1]分离得黄色油状液体, 产率88%. 1H NMR (500 MHz, CDCl3) δ: 7.83~7.72 (m, 2H), 7.48~7.33 (m, 6H), 7.31~7.26 (m, 2H), 4.87 (s, 1H), 4.32~4.13 (m, 4H), 2.43 (s, 3H), 1.26 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.4, 167.8, 143.5, 143.0, 134.8, 134.4, 130.1, 129.1, 129.0, 128.8, 61.9, 51.6, 21.6, 14.0; HRMS (ESI) calcd for C23H25O5 [M+H]+ 381.1697, found 381.1695.
(E)-2-(3-(2-甲氧基苯基)-3-氧代-1-苯基丙-1-烯-2-基)丙二酸二乙酯(3ah): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色油状液体, 产率85%. 1H NMR (500 MHz, CDCl3) δ: 7.50~7.30 (m, 8H), 7.05~6.94 (m, 2H), 4.81 (s, 1H), 4.29~4.11 (m, 4H), 3.83 (s, 3H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.0, 167.7, 157.4, 146.7, 135.8, 134.5, 131.7, 129.9, 129.3, 128.9, 128.6, 128.1, 120.3, 111.6, 61.7, 55.8, 50.7, 14.0; HRMS (ESI) calcd for C23H25O6 [M+H]+ 397.1646, found 397.1649.
(E)-2-(3-(3-甲氧基苯基)-3-氧代-1-苯基丙-1-烯-2-基)丙二酸二乙酯(3ai): 柱层析[V(石油醚)∶V(乙酸乙酯)=12∶1]分离得黄色油状液体, 产率86%. 1H NMR (500 MHz, CDCl3) δ: 7.47~7.33 (m, 9H), 7.15~7.08 (m, 1H), 4.87 (s, 1H), 4.31~4.15 (m, 4H), 3.85 (s, 3H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.4, 167.7, 159.6, 144.2, 138.8, 134.33, 134.26, 129.24, 129.21, 128.8, 122.6, 118.7, 114.2, 61.9, 55.4, 51.5, 14.0; HRMS (ESI) calcd for C23H25O6 [M+H]+ 397.1646, found 397.1645.
(E)-2-(3-(4-甲氧基苯基)-3-氧代-1-苯基丙-1-烯-2-基)丙二酸二乙酯(3aj): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色固体, 产率86%. m.p. 95~96 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.94~7.84 (m, 2H), 7.46~7.35 (m, 6H), 7.34 (s, 1H), 7.00~6.93 (m, 2H), 4.87 (s, 1H), 4.31~4.13 (m, 4H), 3.87 (s, 3H), 1.24 (t, J=7.2 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 195.4, 167.8, 163.1, 142.5, 134.4, 134.2, 132.3, 129.9, 129.0, 128.8, 128.7, 113.6, 61.8, 55.5, 51.7, 13.9; HRMS (ESI) calcd for C23H25O6 [M+H]+ 397.1646, found 397.1643.
(E)-2-(3-(3,4-二甲氧基苯基)-3-氧代-1-苯基丙-1-烯- 2-基)丙二酸二乙酯(3ak): 柱层析[V(石油醚)∶V(乙酸乙酯)=2∶1]分离得黄色油状液体, 产率87%. 1H NMR (500 MHz, CDCl3) δ: 7.53 (dd, J=8.3, 1.9 Hz, 1H), 7.48 (d, J=1.9 Hz, 1H), 7.46~7.34 (m, 6H), 6.91 (d, J=8.4 Hz, 1H), 4.87 (s, 1H), 4.32~4.13 (m, 4H), 3.95 (s, 3H), 3.93 (s, 3H), 1.24 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 195.4, 167.8, 152.9, 149.0, 142.5, 134.4, 134.1, 130.0, 129.1, 128.79, 128.75, 125.0, 112.1, 109.8, 61.9, 56.1, 56.0, 51.7, 14.0; HRMS (ESI) calcd for C24H27O7 [M+H]+ 427.1751, found 427.1749.
(E)-2-(3-氧代-1-苯基-3-(4-(三氟甲基)苯基)丙-1-烯- 2-基)丙二酸二乙酯(3al): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率71%. 1H NMR (500 MHz, CDCl3) δ: 7.94 (d, J=8.0 Hz, 2H), 7.74 (d, J=8.1 Hz, 2H), 7.48~7.37 (m, 5H), 7.34 (s, 1H), 4.89 (s, 1H), 4.33~4.16 (m, 4H), 1.27 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 195.7, 167.6, 144.9, 140.8, 134.3, 133.9, 133.6 (q, 2JFC=32.6 Hz), 130.1, 129.6, 128.94, 128.88, 125.4 (q, 3JFC=3.6 Hz), 123.7 (q, 1JFC=272.8 Hz), 62.1, 51.4, 14.0; 19F NMR (470 MHz, CDCl3) δ: -63.0; HRMS (ESI) calcd for C23H22F3O5 [M+H]+ 435.1414, found 435.1422.
(E)-2-(3-(3-硝基苯基)-3-氧代-1-苯基丙-1-烯-2-基)丙二酸二乙酯(3an): 柱层析[V(石油醚)∶V(乙酸乙 酯)=10∶1]分离得黄色油状液体, 产率76%. 1H NMR (500 MHz, CDCl3) δ: 8.72~8.64 (m, 1H), 8.46~8.40 (m, 1H), 8.22~8.15 (m, 1H), 7.74~7.66 (m, 1H), 7.50~7.39 (m, 5H), 7.33 (s, 1H), 4.90 (s, 1H), 4.34~4.17 (m, 4H), 1.28 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 194.4, 167.5, 148.1, 144.5, 139.0, 135.4, 134.0, 133.6, 129.7, 129.6, 129.0, 128.9, 126.6, 124.7, 62.1, 51.4, 14.0; HRMS (ESI) calcd for C22H22NO7 [M+H]+ 412.1391, found 412.1388.
(E)-2-(3-(4-硝基苯基)-3-氧代-1-苯基丙-1-烯-2-基)丙二酸二乙酯(3ao): 柱层析[V(石油醚)∶V(乙酸乙 酯)=12∶1]分离得黄色油状液体, 产率73%. 1H NMR (500 MHz, CDCl3) δ: 8.39~8.30 (m, 2H), 8.03~7.94 (m, 2H), 7.48~7.37 (m, 5H), 7.31 (s, 1H), 4.90 (s, 1H), 4.33~4.19 (m, 4H), 1.28 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 195.0, 167.5, 149.8, 145.3, 143.0, 134.3, 133.6, 130.6, 129.8, 129.0, 128.9, 123.6, 62.1, 51.2, 14.0; HRMS (ESI) calcd for C22H22NO7 [M+H]+ 412.1391, found 412.1398.
(E)-2-(3-氧代-1-苯基-3-(噻吩-2-基)丙-1-烯-2-基)丙二酸二乙酯(3ap): 柱层析[V(石油醚)∶V(乙酸乙酯)=5∶1]分离得黄色油状液体, 产率61%. 1H NMR (CDCl3, 500 MHz) δ: 7.76 (dd, J=3.8, 1.0 Hz, 1H), 7.69 (dd, J=4.9, 1.0 Hz, 1H), 7.64 (s, 1H), 7.48~7.35 (m, 5H), 7.15 (dd, J=4.9, 3.8 Hz, 1H), 4.86 (s, 1H), 4.31~4.11 (m, 4H), 1.22 (t, J=7.2 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 187.9, 167.5, 142.8, 141.9, 134.23, 134.17, 134.1, 133.9, 129.1, 128.81, 128.76, 127.8, 61.9, 51.7, 13.8; HRMS (ESI) calcd for C20H21O5S [M+H]+ 373.1104, found 373.1101.
(E)-2-(3-氧代-1-苯基丁-1-烯-2-基)丙二酸二乙酯(3aq): 柱层析[V(石油醚)∶V(乙酸乙酯)=4∶1]分离得无色油状液体, 产率12%. 1H NMR (CDCl3, 500 MHz) δ: 7.80 (s, 1H), 7.48~7.37 (m, 5H), 4.66 (s, 1H), 4.25~4.11 (m, 4H), 2.53 (s, 3H), 1.26 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 197.9, 167.8, 143.3, 136.0, 134.3, 129.4, 128.81, 128.77, 61.7, 50.4, 25.7, 14.0; HRMS (ESI) calcd for C17H21O5 [M+H]+ 305.1384, found 305.1386.
(E)-2-(1-(4-氟苯基)-3-氧代-3-苯基丙-1-烯-2-基)丙二酸二乙酯(3ba): 柱层析[V(石油醚)∶V(乙酸乙酯)=12∶1]分离得黄色油状液体, 产率78%. 1H NMR (500 MHz, CDCl3) δ: 7.83 (d, J=7.3 Hz, 2H), 7.57 (t, J=7.4 Hz, 1H), 7.53~7.39 (m, 4H), 7.33 (s, 1H), 7.10 (t, J=8.6 Hz, 2H), 4.84 (s, 1H), 4.33~4.13 (m, 4H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.6, 167.6, 163.1 (d, 1JFC=250.4 Hz), 142.9, 137.3, 134.4, 132.3, 130.8 (d, 3JFC=8.3 Hz), 130.2 (d, 4JFC=3.2 Hz), 129.9, 128.3, 115.9 (d, 2JFC=21.7 Hz), 62.0, 51.3, 14.0; 19F NMR (470 MHz, CDCl3) δ: -111.0; HRMS (ESI) calcd for C22H22FO5 [M+H]+ 385.1446, found 385.1451.
(E)-2-(1-(4-氯苯基)-3-氧代-3-苯基丙-1-烯-2-基)丙二酸二乙酯(3ca): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色固体, 产率83%. m.p. 64~65 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.90~7.77 (m, 2H), 7.57 (t, J=7.4 Hz, 1H), 7.53~7.44 (m, 2H), 7.43~7.34 (m, 4H), 7.31 (s, 1H), 4.82 (s, 1H), 4.34~4.12 (m, 4H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.4, 167.5, 142.5, 137.2, 135.3, 134.9, 132.6, 132.4, 130.1, 129.9, 129.0, 128.3, 62.0, 51.4, 14.0; HRMS (ESI) calcd for C22H22ClO5 [M+H]+ 401.1150, found 401.1140.
(E)-2-(1-(4-溴苯基)-3-氧代-3-苯基丙-1-烯-2-基)丙二酸二乙酯(3da): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率81%. 1H NMR (500 MHz, CDCl3) δ: 7.83 (d, J=7.2 Hz, 2H), 7.62~7.52 (m, 3H), 7.51~7.44 (m, 2H), 7.35~7.27 (m, 3H), 4.81 (s, 1H), 4.32~4.13 (m, 4H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.4, 167.5, 142.4, 137.2, 135.0, 133.1, 132.4, 132.0,130.3, 129.9, 128.3, 123.6, 62.0, 51.4, 13.9; HRMS (ESI) calcd for C22H22BrO5 [M+H]+ 445.0645, found 445.0650.
(E)-2-(3-氧代-3-苯基-1-(p-甲基苯基)丙-1-烯-2-基)丙二酸二乙酯(3ea): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率91%. 1H NMR (500 MHz, CDCl3) δ: 7.89~7.78 (m, 2H), 7.61~7.51 (m, 1H), 7.51~7.43 (m, 2H), 7.36 (s, 1H), 7.33~7.28 (m, 2H), 7.25~7.18 (m, 2H), 4.90 (s, 1H), 4.33~4.14 (m, 4H), 2.38 (s, 3H), 1.26 (t, J=7.2 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.8, 167.8, 144.5, 139.6, 137.7, 133.8, 132.1, 131.4, 129.8, 129.5, 128.9, 128.2, 61.8, 51.5, 21.4, 14.0; HRMS (ESI) calcd for C23H25O5 [M+H]+ 381.1697, found 381.1688.
(E)-2-(1-(2-甲氧基苯基)-3-氧代-3-苯基丙-1-烯-2-基)丙二酸二乙酯(3fa): 柱层析[V(石油醚)∶V(乙酸乙酯)=8∶1]分离得黄色油状液体, 产率88%. 1H NMR (500 MHz, CDCl3) δ: 7.91 (d, J=7.7 Hz, 2H), 7.62~7.52 (m, 1H), 7.52~7.33 (m, 5H), 6.99 (t, J=7.5 Hz, 1H), 6.91 (d, J=8.3 Hz, 1H), 4.75 (s, 1H), 4.33~4.11 (m, 4H), 3.81 (s, 3H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.8, 168.0, 157.2, 140.9, 137.6, 134.0, 132.2, 130.8, 130.2, 129.6, 128.2, 123.4, 120.6, 110.6, 61.8, 55.5, 51.9, 14.0; HRMS (ESI) calcd for C23H25O6 [M+H]+ 397.1646, found 397.1650.
(E)-2-(1-(3-甲氧基苯基)-3-氧代-3-苯基丙-1-烯-2-基)丙二酸二乙酯(3ga): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色油状液体, 产率86%. 1H NMR (500 MHz, CDCl3) δ: 7.84 (d, J=7.4 Hz, 2H), 7.56 (t, J=7.4 Hz, 1H), 7.52~7.43 (m, 2H), 7.36 (s, 1H), 7.32 (t, J=7.9 Hz, 1H), 7.01~6.88 (m, 3H), 4.91 (s, 1H), 4.32~4.14 (m, 4H), 3.81 (s, 3H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.6, 167.7, 159.7, 144.0, 137.5, 135.5, 134.5, 132.2, 129.9, 129.8, 128.3, 121.1, 115.2, 113.8, 61.9, 55.3, 51.6, 13.9; HRMS (ESI) calcd for C23H25O6 [M+H]+ 397.1646, found 397.1654.
(E)-2-(1-(4-甲氧基苯基)-3-氧代-3-苯基丙-1-烯-2-基)丙二酸二乙酯(3ha): 柱层析[V(石油醚)∶V(乙酸乙酯)=8∶1]分离得黄色固体, 产率92%. m.p. 53~54 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.86~7.76 (m, 2H), 7.55 (t, J=7.4 Hz, 1H), 7.51~7.43 (m, 2H), 7.42~7.32 (m, 3H), 6.93 (d, J=8.7 Hz, 2H), 4.92 (s, 1H), 4.37~4.11 (m, 4H), 3.84 (s, 3H), 1.26 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.9, 167.9, 160.5, 144.5, 137.9, 132.8, 132.0, 130.8, 129.8, 128.2, 126.6, 114.3, 61.9, 55.4, 51.4, 14.0; HRMS (ESI) calcd for C23H25O6 [M+H]+ 397.1646, found 397.1647.
(E)-2-(3-氧代-3-苯基-1-(4-(三氟甲基)苯基)丙-1-烯-2-基)丙二酸二乙酯(3ia): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率80%. 1H NMR (500 MHz, CDCl3) δ: 7.93~7.81 (m, 2H), 7.68 (d, J=8.2 Hz, 2H), 7.63~7.44 (m, 5H), 7.36 (s, 1H), 4.78 (s, 1H), 4.33~4.12 (m, 4H), 1.25 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.2, 167.4, 141.7, 137.9, 137.0, 136.1, 132.6, 131.0 (q, 2JFC=32.5 Hz), 130.0, 129.0, 128.4, 125.7 (q, 3JFC=3.5 Hz), 123.8 (q, 1JFC=272.7 Hz), 62.1, 51.5, 14.0; 19F NMR (470 MHz, CDCl3) δ: -62.8; HRMS (ESI) calcd for C23H22F3O5 [M+H]+ 435.1414, found 435.1422.
(E)-2-(1-(3-硝基苯基)-3-氧代-3-苯基丙-1-烯-2-基)丙二酸二乙酯(3ka): 柱层析[V(石油醚)∶V(乙酸乙 酯)=8∶1]分离得黄色油状液体, 产率78%. 1H NMR (500 MHz, CDCl3) δ: 8.33 (s, 1H), 8.25 (d, J=8.2 Hz, 1H), 7.93~7.86 (m, 2H), 7.82 (d, J=7.6 Hz, 1H), 7.68~7.58 (m, 2H), 7.56~7.48 (m, 2H), 7.35 (s, 1H), 4.75 (s, 1H), 4.34~4.17 (m, 4H), 1.27 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.0, 167.2, 148.4, 140.1, 136.74, 136.71, 135.9, 134.6, 132.8, 129.99, 129.96, 128.5, 123.8, 123.7, 62.3, 51.5, 13.9; HRMS (ESI) calcd for C22H22NO7 [M+H]+ 412.1391, found 412.1393.
(E)-2-(1-(呋喃-2-基)-3-氧代-3-苯基丙-1-烯-2-基)丙二酸二乙酯(3ma): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色油状液体, 产率88%. 1H NMR (500 MHz, CDCl3) δ: 7.80~7.67 (m, 2H), 7.62~7.50 (m, 2H), 7.50~7.40 (m, 2H), 7.01 (s, 1H), 6.63 (d, J=3.4 Hz, 1H), 6.54~6.45 (m, 1H), 5.40 (s, 1H), 4.31~4.15 (m, 4H), 1.24 (t, J=7.1 Hz, 6H); 13C NMR (125 MHz, CDCl3) δ: 196.5, 167.8, 149.9, 145.6, 137.9, 131.8, 130.7, 129.8, 129.5, 128.3, 117.8, 112.5, 61.7, 51.8, 14.0; HRMS (ESI) calcd for C20H21O6 [M+H]+ 357.1333, found 357.1335.
在经无水无氧处理过的反应管中加0.6 mmol三甲基碘化亚砜, 4 mL无水DMF, 0.9 mmol 60%氢化钠和0.3 mmol化合物3, 搅拌10 min后将反应管移入50 ℃的金属浴中搅拌反应, TLC跟踪至化合物3消失, 冷却至室温, 并加入5 mL饱和氯化铵水溶液淬灭反应, 用乙酸乙酯萃取(5 mL×3), 合并有机相, 用饱和氯化钠水溶液洗涤(5 mL×3), 无水硫酸钠干燥, 过滤, 回收乙酸乙酯, 柱层析分离得纯的产物4.
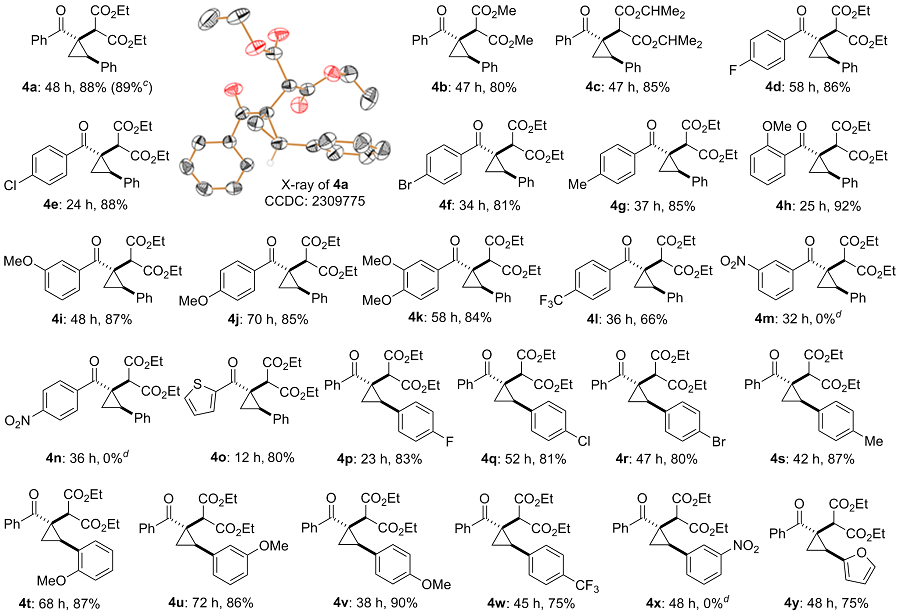
2-(R-1-苯甲酰基-trans-2-苯基环丙基)丙二酸二乙酯(4a): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得无色固体, 产率88%. m.p. 50~51 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.70~7.58 (m, 2H), 7.50~7.38 (m, 3H), 7.35~7.27 (m, 3H), 7.26~7.22 (m, 2H), 4.20~4.07 (m, 2H), 4.06~3.91 (m, 2H), 3.18~3.07 (m, 1H), 2.80 (s, 1H), 1.93 (dd, J=7.3, 6.4 Hz, 1H), 1.67 (dd, J=9.3, 6.2 Hz, 1H), 1.22 (t, J=7.1 Hz, 3H), 1.08 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.9, 169.0, 168.2, 137.8, 135.1, 130.6, 129.2, 128.4, 128.1, 127.5, 127.4, 61.6, 61.2, 53.2, 37.9, 28.5, 17.1, 13.9; HRMS (ESI) calcd for C23H24- O5Na [M+Na]+ 403.1516, found 403.1506.
2-(R-1-苯甲酰基-trans-2-苯基环丙基)丙二酸二甲酯(4b): 柱层析[V(石油醚)∶V(乙酸乙酯)=12∶1]分离得黄色油状液体, 产率80%. 1H NMR (500 MHz, CDCl3) δ: 7.68~7.60 (m, 2H), 7.51~7.39 (m, 3H), 7.34~7.26 (m, 3H), 7.26~7.23 (m, 2H), 3.69 (s, 3H), 3.52 (s, 3H), 3.19~3.08 (m, 1H), 2.83 (s, 1H), 1.93 (dd, J=7.5, 6.3 Hz, 1H), 1.69 (dd, J=9.3, 6.3 Hz, 1H); 13C NMR (125 MHz, CDCl3) δ: 202.9, 169.4, 168.7, 137.7, 134.9, 130.7, 129.2, 128.4, 128.2, 127.6, 127.5, 52.8, 52.6, 52.2, 38.0, 28.6, 16.5; HRMS (ESI) calcd for C21H21O5 [M+H]+ 353.1384, found 353.1380.
2-(R-1-苯甲酰基-trans-2-苯基环丙基)丙二酸二异丙酯(4c): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得无色固体, 产率85%. m.p. 85~86 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.72~7.59 (m, 2H), 7.49~7.35 (m, 3H), 7.33~7.26 (m, 2H), 7.26~7.19 (m, 3H), 5.07~4.97 (m, 1H), 4.95~4.83 (m, 1H), 3.09 (dd, J=9.3, 7.5 Hz, 1H), 2.71 (s, 1H), 1.93 (dd, J=7.5, 6.2 Hz, 1H), 1.61 (dd, J=9.3, 6.2 Hz, 1H), 1.213 (d, J=6.3 Hz, 3H), 1.210 (d, J=6.3 Hz, 3H), 1.12 (d, J=6.3 Hz, 3H), 0.99 (d, J=6.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.6, 168.3, 167.6, 137.7, 135.1, 130.4, 129.0, 128.3, 127.9, 127.4, 127.2, 69.0, 68.4, 53.4, 37.8, 28.1, 21.5, 21.4, 21.33, 21.27, 16.1; HRMS (ESI) calcd for C25H29O5 [M+H]+ 409.2010, found 409.2013.
2-(R-1-(4-氟苯甲酰基)-trans-2-苯基环丙基)丙二酸二乙酯(4d): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色固体, 产率86%. m.p. 81~82 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.78~7.66 (m, 2H), 7.35~7.26 (m, 3H), 7.26~7.22 (m, 2H), 7.16~7.06 (m, 2H), 4.20~4.06 (m, 2H), 4.05~3.91 (m, 2H), 3.16~3.06 (m, 1H), 2.81 (s, 1H), 1.96 (dd, J=7.4, 6.3 Hz, 1H), 1.62 (dd, J=9.3, 6.2 Hz, 1H), 1.22 (t, J=7.2 Hz, 3H), 1.08 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 201.4, 168.9, 168.3, 164.2 (d, 1JFC=251.6 Hz), 135.1, 133.9 (d, 4JFC=3.1 Hz), 130.3 (d, 3JFC=8.7 Hz), 129.2, 128.5, 127.5, 115.2 (d, 2JFC=21.8 Hz), 61.6, 61.2, 53.3, 37.6, 28.3, 16.4, 13.9; 19F NMR (470 MHz, CDCl3) δ: -108.4; HRMS (ESI) calcd for C23H24FO5 [M+H]+ 399.1602, found 399.1600.
2-(R-1-(4-氯苯甲酰基)-trans-2-苯基环丙基)丙二酸二乙酯(4e): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率88%. 1H NMR (500 MHz, CDCl3) δ: 7.66~7.57 (m, 2H), 7.44~7.35 (m, 2H), 7.34~7.20 (m, 5H), 4.18~4.06 (m, 2H), 4.04~3.91 (m, 2H), 3.08 (dd, J=9.3, 7.5 Hz, 1H), 2.80 (s, 1H), 1.95 (dd, J=7.5, 6.3 Hz, 1H), 1.62 (dd, J=9.3, 6.3 Hz, 1H), 1.21 (t, J=7.2 Hz, 3H), 1.06 (t, J=7.2 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 201.6, 168.7, 168.1, 136.8, 136.1, 134.8, 129.1, 129.0, 128.4, 128.3, 127.4, 61.5, 61.1, 53.1, 37.6, 28.3, 16.4, 13.8; HRMS (ESI) calcd for C23H24ClO5 [M+H]+ 415.1307, found 415.1304.
2-(R-1-(4-溴苯甲酰基)-trans-2-苯基环丙基)丙二酸二乙酯(4f): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色固体, 产率81%. m.p. 119~120 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.62~7.49 (m, 4H), 7.35~7.19 (m, 5H), 4.19~4.05 (m, 2H), 4.05~3.90 (m, 2H), 3.14~3.03 (m, 1H), 2.79 (s, 1H), 1.95 (dd, J=7.4, 6.3 Hz, 1H), 1.62 (dd, J=9.3, 6.2 Hz, 1H), 1.21 (t, J=7.2 Hz, 3H), 1.07 (t, J=7.2 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 201.8, 168.8, 168.2, 136.6, 134.8, 131.3, 129.3, 129.1, 128.4, 127.5, 125.2, 61.6, 61.2, 53.1, 37.7, 28.4, 16.5, 13.8; HRMS (ESI) calcd for C23H23BrO5Na [M+Na]+ 481.0621, found 481.0625.
2-(R-1-(4-甲基苯甲酰基)-trans-2-苯基环丙基)丙二酸二乙酯(4g): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色油状液体, 产率85%. 1H NMR (500 MHz, CDCl3) δ: 7.58 (d, J=8.1 Hz, 2H), 7.34~7.18 (m, 7H), 4.20~4.08 (m, 2H), 4.05~3.92 (m, 2H), 3.18~3.07 (m, 1H), 2.82 (s, 1H), 2.37 (s, 3H), 1.92 (dd, J=7.3, 6.3 Hz, 1H), 1.64 (dd, J=9.3, 6.2 Hz, 1H), 1.22 (t, J=7.2 Hz, 3H), 1.08 (t, J=7.2 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.4, 168.9, 168.1, 141.0, 135.2, 134.8, 129.1, 128.7, 128.3, 127.8, 127.3, 61.4, 61.0, 53.2, 37.7, 28.4, 21.4, 16.3, 13.8; HRMS (ESI) calcd for C24H27O5 [M+H]+ 395.1853, found 395.1853.
2-(R-1-(2-甲氧基苯甲酰基)-trans-2-苯基环丙基)丙二酸二乙酯(4h): 柱层析[V(石油醚)∶V(乙酸乙酯)=8∶1]分离得黄色固体, 产率92%. m.p. 61~62 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.42~7.14 (m, 7H), 6.99 (t, J=7.4 Hz, 1H), 6.92 (d, J=8.2 Hz, 1H), 4.25~4.08 (m, 2H), 4.05~3.92 (m, 2H), 3.86 (s, 3H), 3.04~2.94 (m, 1H), 2.81 (s, 1H), 1.85~1.73 (m, 2H), 1.25 (t, J=7.1 Hz, 3H), 1.11 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.6, 168.6, 167.7, 155.6, 134.9, 130.4, 129.2, 128.1, 128.0, 127.23, 127.20, 120.2, 111.2, 61.2, 60.8, 55.6, 52.1, 39.8, 30.4, 17.6, 13.9, 13.8; HRMS (ESI) calcd for C24H27O6 [M+H]+ 411.1802, found 411.1793.
2-(R-1-(3-甲氧基苯甲酰基)-trans-2-苯基环丙基)丙二酸二乙酯(4i): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色油状液体, 产率87%. 1H NMR (500 MHz, CDCl3) δ: 7.38~7.20 (m, 7H), 7.15 (dd, J=2.5, 1.4 Hz, 1H), 7.04~6.94 (m, 1H), 4.21~4.08 (m, 2H), 4.06~3.92 (m, 2H), 3.84 (s, 3H), 3.21~3.10 (m, 1H), 2.81 (s, 1H), 1.93 (dd, J=7.5, 6.3 Hz, 1H), 1.70 (dd, J=9.3, 6.2 Hz, 1H), 1.23 (t, J=7.1 Hz, 3H), 1.09 (t, J=7.2 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.6, 168.9, 168.1, 159.3, 139.0, 135.1, 129.2, 128.4, 127.4, 119.7, 116.5, 112.7, 61.5, 61.1, 55.4, 53.1, 38.0, 28.7, 16.7, 13.89, 13.87; HRMS (ESI) calcd for C24H27O6 [M+H]+ 411.1802, found 411.1796.
2-(R-1-(4-甲氧基苯甲酰基)-trans-2-苯基环丙基)丙二酸二乙酯(4j): 柱层析[V(石油醚)∶V(乙酸乙酯)=8∶1]分离得黄色固体, 产率85%. m.p. 71~72 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.75 (d, J=8.4 Hz, 2H), 7.38~7.18 (m, 5H), 6.92 (d, J=8.6 Hz, 2H), 4.20~4.08 (m, 2H), 4.05~3.92 (m, 2H), 3.83 (s, 3H), 3.20~3.08 (m, 1H), 2.83 (s, 1H), 2.00~1.89 (m, 1H), 1.60 (dd, J=9.2, 6.2 Hz, 1H), 1.21 (t, J=7.1 Hz, 3H), 1.06 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 201.0, 169.0, 168.3, 161.9, 135.4, 130.3, 129.9, 129.1, 128.4, 127.3, 113.3, 61.5, 61.1, 55.3, 53.6, 37.2, 28.2, 16.1, 13.84, 13.83; HRMS (ESI) calcd for C24H27O6 [M+H]+ 411.1802, found 411.1800.
2-(R-1-(3,4-二甲氧基苯甲酰基)-trans-2-苯基环丙基)丙二酸二乙酯(4k): 柱层析[V(石油醚)∶V(乙酸乙 酯)=2∶1]分离得黄色油状液体, 产率84%. 1H NMR (500 MHz, CDCl3) δ: 7.46 (dd, J=8.3, 1.9 Hz, 1H), 7.35~7.20 (m, 6H), 6.88 (d, J=8.3 Hz, 1H), 4.18~4.05 (m, 2H), 4.03~3.93 (m, 2H), 3.92 (s, 3H), 3.91 (s, 3H), 3.23~3.13 (m, 1H), 2.83 (s, 1H), 1.94 (dd, J=7.3, 6.3 Hz, 1H), 1.63 (dd, J=9.3, 6.2 Hz, 1H), 1.20 (t, J=7.2 Hz, 3H), 1.06 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 200.7, 169.0, 168.2, 151.6, 148.6, 135.4, 129.9, 129.1, 128.4, 127.3, 122.1, 111.5, 109.8, 61.5, 61.1, 56.0, 53.6, 37.1, 28.3, 16.3, 13.85, 13.83; HRMS (ESI) calcd for C25H29O7 [M+H]+ 441.1908, found 441.1902.
2-(trans-2-苯基-R-1-(4-(三氟甲基)苯甲酰基)环丙基)丙二酸二乙酯(4l): 柱层析[V(石油醚)∶V(乙酸乙 酯)=10∶1]分离得黄色固体, 产率66%. m.p. 99~100 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.74 (d, J=8.1 Hz, 2H), 7.68 (d, J=8.2 Hz, 2H), 7.35~7.21 (m, 5H), 4.18~4.04 (m, 2H), 4.04~3.91 (m, 2H), 3.13~3.04 (m, 1H), 2.80 (s, 1H), 1.98 (dd, J=7.5, 6.4 Hz, 1H), 1.67 (dd, J=9.4, 6.3 Hz, 1H), 1.20 (t, J=7.2 Hz, 3H), 1.07 (t, J=7.2 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.0, 168.8, 168.2, 141.5, 134.7, 132.2 (q, 2JFC=32.7 Hz), 129.2, 128.5, 127.8, 127.6, 125.2 (q, 3JFC=3.6 Hz), 123.7 (q, 1JFC=272.5 Hz), 61.7, 61.3, 53.0, 38.0, 28.6, 16.8, 13.8; 19F NMR (470 MHz, CDCl3) δ: -63.0; HRMS (ESI) calcd for C24H24F3O5 [M+H]+ 449.1570, found 449.1561.
2-(trans-2-苯基-R-1-(噻吩-2-甲酰基)环丙基)丙二酸二乙酯(4o): 柱层析[V(石油醚)∶V(乙酸乙酯)=5∶1]分离得黄色油状液体, 产率80%. 1H NMR (500 MHz, CDCl3) δ: 7.67 (d, J=3.6 Hz, 1H), 7.60 (d, J=4.8 Hz, 1H), 7.36~7.27 (m, 5H), 7.16~7.08 (m, 1H), 4.24~4.11 (m, 2H), 4.05~3.88 (m, 2H), 3.61~3.48 (m, 1H), 2.78 (s, 1H), 2.21~2.08 (m, 1H), 2.03~1.90 (m, 1H), 1.24 (t, J=7.1 Hz, 3H), 1.07 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 191.4, 168.7, 167.6, 141.2, 134.9, 132.6, 131.3, 129.3, 128.4, 127.7, 127.5, 61.5, 61.0, 53.7, 39.0, 31.4, 19.0, 13.9, 13.8; HRMS (ESI) calcd for C21H23O5S [M+H]+ 387.1261, found 387.1253.
2-(R-1-苯甲酰基-trans-2-(4-氟苯基)环丙基)丙二酸二乙酯(4p): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得白色固体, 产率83%. m.p. 88~89 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.66~7.58 (m, 2H), 7.52~7.36 (m, 3H), 7.26~7.18 (m, 2H), 7.04~6.97 (m, 2H), 4.19~4.08 (m, 2H), 4.08~3.93 (m, 2H), 3.13~3.01 (m, 1H), 2.78 (s, 1H), 1.88 (dd, J=7.3, 6.4 Hz, 1H), 1.69 (dd, J=9.3, 6.3 Hz, 1H), 1.22 (t, J=7.2 Hz, 3H), 1.11 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.6, 168.8, 168.1, 162.1 (d, 1JFC=246.6 Hz), 137.8, 130.9 (d, 4JCF=3.2 Hz), 130.8 (d, 3JCF=8.1 Hz), 130.6, 128.1, 127.5, 115.3 (d, 2JCF=21.6 Hz), 61.6, 61.3, 53.1, 37.8, 27.7, 16.7, 13.9; 19F NMR (470 MHz, CDCl3) δ: -114.7; HRMS (ESI) calcd for C23H24FO5 [M+H]+ 399.1602, found 399.1592.
2-(R-1-苯甲酰基-trans-2-(4-氯苯基)环丙基)丙二酸二乙酯(4q): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得黄色固体, 产率81%. m.p. 109~110 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.61 (d, J=7.9 Hz, 2H), 7.51~7.35 (m, 3H), 7.27 (d, J=8.2 Hz, 2H), 7.19 (d, J=8.3 Hz, 2H), 4.19~4.07 (m, 2H), 4.06~3.92 (m, 2H), 3.13~2.97 (m, 1H), 2.78 (s, 1H), 1.95~1.82 (m, 1H), 1.75~1.63 (m, 1H), 1.20 (t, J=7.1 Hz, 3H), 1.10 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.4, 168.6, 168.0, 137.6, 133.7, 133.3, 130.63, 130.55, 128.5, 128.1, 127.4, 61.6, 61.3, 53.0, 37.8, 27.8, 16.5, 13.8; HRMS (ESI) calcd for C23H24ClO5 [M+H]+ 415.1307, found 415.1313.
2-(R-1-苯甲酰基-trans-2-(4-溴苯基)环丙基)丙二酸二乙酯(4r): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得白色固体, 产率80%. m.p. 118~119 oC; 1H NMR (500 MHz, CDCl3) δ: 7.72~7.55 (m, 2H), 7.53~7.36 (m, 5H), 7.13 (d, J=8.4 Hz, 2H), 4.21~4.06 (m, 2H), 4.06~3.92 (m, 2H), 3.12~2.95 (m, 1H), 2.79 (s, 1H), 1.96~1.82 (m, 1H), 1.69 (dd, J=9.2, 6.3 Hz, 1H), 1.21 (t, J=7.1 Hz, 3H), 1.10 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.4, 168.6, 168.0, 137.6, 134.3, 131.5, 130.9, 130.7, 128.1, 127.4, 121.4, 61.6, 61.3, 53.0, 37.8, 27.9, 16.5, 13.9; HRMS (ESI) calcd for C23H24BrO5 [M+H]+ 459.0802, found 459.0808.
2-(R-1-苯甲酰基-trans-2-(p-甲基苯基)环丙基)丙二酸二乙酯(4s): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得白色固体, 产率87%. m.p. 84~85 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.69~7.58 (m, 2H), 7.50~7.35 (m, 3H), 7.20~7.04 (m, 4H), 4.22~4.07 (m, 2H), 4.06~3.94 (m, 2H), 3.15~3.01 (m, 1H), 2.81 (s, 1H), 2.32 (s, 3H), 1.89 (dd, J=7.4, 6.2 Hz, 1H), 1.66 (dd, J=9.3, 6.2 Hz, 1H), 1.22 (t, J=7.2 Hz, 3H), 1.09 (t, J=7.2 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.9, 169.0, 168.2, 137.9, 137.1, 132.0, 130.5, 129.1, 129.0, 128.1, 127.5, 61.5, 61.1, 53.1, 37.9, 28.3, 21.1, 16.5, 13.89, 13.86; HRMS (ESI) calcd for C24H27O5 [M+H]+ 395.1853, found 395.1844.
2-(R-1-苯甲酰基-trans-2-(2-甲氧基苯基)环丙基)丙二酸二乙酯(4t): 柱层析[V(石油醚)∶V(乙酸乙酯)=8∶1]分离得黄色固体, 产率87%. m.p. 61~62 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.86~7.77 (m, 2H), 7.50~7.37 (m, 3H), 7.30~7.23 (m, 1H), 7.07~7.01 (m, 1H), 6.95~6.84 (m, 2H), 4.17~3.92 (m, 4H), 3.91 (s, 3H), 3.12~3.05 (m, 1H), 3.03 (s, 1H), 1.96 (dd, J=7.7, 6.1 Hz, 1H), 1.64 (dd, J=9.4, 6.1 Hz, 1H), 1.21 (t, J=7.2 Hz, 3H), 1.06 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 203.2, 169.0, 167.8, 159.2, 137.7, 130.6, 129.0, 128.8, 128.0, 127.9, 123.9, 120.3, 109.9, 61.5, 61.0, 55.1, 52.7, 37.1, 25.5, 15.3, 13.9, 13.8; HRMS (ESI) calcd for C24H27O6 [M+H]+ 411.1802, found 411.1798.
2-(R-1-苯甲酰基-trans-2-(3-甲氧基苯基)环丙基)丙二酸二乙酯(4u): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色油状液体, 产率86%. 1H NMR (500 MHz, CDCl3) δ: 7.71~7.58 (m, 2H), 7.51~7.36 (m, 3H), 7.25~7.16 (m, 1H), 6.90~6.74 (m, 3H), 4.21~4.07 (m, 2H), 4.07~3.96 (m, 2H), 3.80 (s, 3H), 3.16~3.05 (m, 1H), 2.83 (s, 1H), 1.91 (dd, J=7.4, 6.3 Hz, 1H), 1.64 (dd, J=9.3, 6.2 Hz, 1H), 1.22 (t, J=7.1 Hz, 3H), 1.10 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.8, 169.0, 168.3, 159.6, 137.9, 136.8, 130.6, 129.5, 128.1, 127.6, 121.5, 115.3, 112.6, 61.6, 61.2, 55.2, 53.2, 37.8, 28.4, 16.7, 13.9; HRMS (ESI) calcd for C24H27O6 [M+ H]+ 411.1802, found 411.1796.
2-(R-1-苯甲酰基-trans-2-(4-甲氧基苯基)环丙基)丙二酸二乙酯(4v): 柱层析[V(石油醚)∶V(乙酸乙酯)=8∶1]分离得黄色固体, 产率90%. m.p. 71~72 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.68~7.58 (m, 2H), 7.51~7.32 (m, 3H), 7.16 (d, J=8.7 Hz, 2H), 6.82 (d, J=8.6 Hz, 2H), 4.21~4.08 (m, 2H), 4.07~3.95 (m, 2H), 3.76 (s, 3H), 3.10~2.98 (m, 1H), 2.80 (s, 1H), 1.92~1.78 (m, 1H), 1.65 (dd, J=9.3, 6.2 Hz, 1H), 1.21 (t, J=7.1 Hz, 3H), 1.10 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.9, 168.9, 168.2, 158.9, 137.8, 130.4, 130.2, 128.0, 127.3, 126.9, 113.7, 61.4, 61.1, 55.2, 53.0, 37.9, 28.0, 16.7, 13.8; HRMS (ESI) calcd for C24H27O6 [M+ H]+ 411.1802, found 411.1799.
2-(R-1-苯甲酰基-trans-2-(4-(三氟甲基)苯基)环丙基)丙二酸二乙酯(4w): 柱层析[V(石油醚)∶V(乙酸乙酯)=15∶1]分离得白色固体, 产率75%. m.p. 100~101 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.64 (d, J=7.2 Hz, 2H), 7.57 (d, J=8.1 Hz, 2H), 7.51~7.35 (m, 5H), 4.18~4.04 (m, 2H), 4.02~3.89 (m, 2H), 3.22~3.08 (m, 1H), 2.79 (s, 1H), 2.05~1.93 (m, 1H), 1.75 (dd, J=9.1, 6.4 Hz, 1H), 1.20 (t, J=7.1 Hz, 3H), 1.05 (t, J=7.2 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 202.2, 168.5, 167.9, 139.5, 137.6, 130.8, 129.7 (q, 2JFC=32.8 Hz), 129.6, 128.2, 127.5, 125.3 (q, 3JFC=3.9 Hz), 124.0 (q, 1JFC=272.1 Hz), 61.7, 61.3, 53.1, 37.9, 28.0, 16.5, 13.83, 13.76; 19F NMR (470 MHz, CDCl3) δ: -62.6; HRMS (ESI) calcd for C24H24F3O5 [M+H]+ 449.1570, found 449.1567.
2-(R-1-苯甲酰基-trans-2-(呋喃-2-基)环丙基)丙二酸二乙酯(4y): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色油状液体, 产率75%. 1H NMR (500 MHz, CDCl3) δ: 7.70~7.59 (m, 2H), 7.49~7.37 (m, 3H), 7.37~7.30 (m, 1H), 6.35~6.28 (m, 1H), 6.21~6.13 (m, 1H), 4.21~4.11 (m, 2H), 4.10~3.99 (m, 2H), 3.15 (s, 1H), 2.89 (dd, J=9.5, 7.2 Hz, 1H), 1.86~1.73 (m, 2H), 1.23 (t, J=7.2 Hz, 3H), 1.13 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 201.6, 168.6, 168.0, 150.6, 142.3, 137.4, 130.9, 128.1, 127.8, 110.6, 108.8, 61.6, 61.3, 53.3, 37.5, 21.8, 17.0, 13.9; HRMS (ESI) calcd for C21H22O6Na [M+Na]+ 393.1309, found 393.1304.
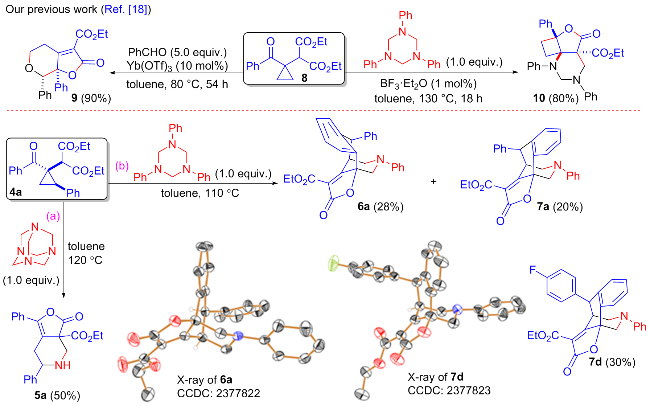
在经无水无氧处理过的反应管中加0.2 mmol环丙烷4a, 0.2 mmol乌洛托品或1,3,5-三苯基-1,3,5-三嗪烷, 2 mL无水甲苯, 搅拌10 min后将反应管移入120或110 ℃的金属浴中搅拌反应, TLC跟踪至化合物4a消失, 冷却至室温并加入2 mL饱和氯化铵水溶液淬灭反应, 用二氯甲烷萃取(5 mL×3), 合并有机相, 无水硫酸钠干燥, 过滤, 回收溶剂, 柱层析分离得纯的产物5a或6a和7a. 环丙烷4d与1,3,5-三苯基-1,3,5-三嗪烷的反应得产物7d.
3-氧代-1,6-二苯基-4,5,6,7-四氢呋喃并[3,4-c]吡啶- 3a(3H)-羧酸乙酯(5a): 柱层析[V(石油醚)∶V(乙酸乙 酯)=2∶1]分离得无色油状液体, 产率50%. 1H NMR (500 MHz, CDCl3) δ: 7.64~7.56 (m, 2H), 7.49~7.38 (m, 3H), 7.37~7.28 (m, 5H), 4.45~4.34 (m, 3H), 3.92~3.83 (m, 2H), 3.00 (d, J=12.1 Hz, 1H), 2.48 (dd, J=12.9, 11.2 Hz, 1H), 1.89 (s, 1H), 1.39 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 179.7, 167.8, 161.4, 141.3, 135.6, 129.3, 129.1, 128.8, 128.3, 127.0, 126.6, 116.8, 84.0, 63.6, 61.6, 56.9, 37.4, 14.2; HRMS (ESI) calcd for C22H22NO4 [M+H]+ 364.1543, found 364.1549.
(5-endo)-2-氧代-5,11-二苯基-4,5-二氢-2H-4,9b-(亚甲基氨基亚甲基)萘并[1,2-b]呋喃-3-羧酸乙酯(6a): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得无色固体, 产率28%. m.p. 150~151 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.66 (dd, J=7.9, 1.1 Hz, 1H), 7.35~7.29 (m, 1H), 7.25~7.12 (m, 6H), 6.94~6.79 (m, 4H), 6.70~6.62 (m, 2H), 4.63 (s, 1H), 4.28~4.18 (m, 1H), 4.16~4.00 (m, 4H), 3.32~3.25 (m, 1H), 3.00 (d, J=10.8 Hz, 1H), 0.98 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 177.7, 167.4, 160.4, 149.2, 145.2, 138.8, 135.7, 129.2, 128.9, 128.7, 128.6, 127.65, 127.61, 127.0, 124.3, 120.9, 117.3, 114.1, 81.4, 63.3, 61.0, 57.2, 55.5, 44.9, 13.8; HRMS (ESI) calcd for C29H26NO4 [M+H]+452.1856, found 452.1865.
(5-exo)-2-氧代-5,11-二苯基-4,5-二氢-2H-4,9b-(亚甲基氨基亚甲基)萘并[1,2-b]呋喃-3-羧酸乙酯(7a): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得无色油状液体, 产率20%. 1H NMR (500 MHz, CDCl3) δ: 7.66 (dd, J=7.8, 1.4 Hz, 1H), 7.52~7.29 (m, 4H), 7.24~7.07 (m, 4H), 7.05~6.90 (m, 2H), 6.84 (t, J=7.4 Hz, 1H), 6.55 (d, J=7.7 Hz, 2H), 4.82 (d, J=6.1 Hz, 1H), 4.41 (q, J=7.1 Hz, 2H), 4.28~4.17 (m, 2H), 3.55 (d, J=12.3 Hz, 1H), 2.99 (d, J=10.6 Hz, 1H), 2.86~2.76 (m, 1H), 1.42 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 180.4, 167.1, 161.1, 148.8, 140.7, 138.6, 136.4, 130.1, 129.1, 128.8, 128.64, 128.55, 128.3, 127.7, 124.6, 120.7, 116.9, 112.9, 81.3, 63.4, 61.5, 52.0, 51.6, 42.0, 14.3; HRMS (ESI) calcd for C29H26NO4 [M+H]+ 452.1856, found 452.1862.
(5-exo)-5-(4-氟苯基)-2-氧代-11-苯基-4,5-二氢-2H- 4,9b-(亚甲基氨基亚甲基)萘并[1,2-b]呋喃-3-羧酸乙酯(7d): 柱层析[V(石油醚)∶V(乙酸乙酯)=10∶1]分离得黄色固体, 产率30%. m.p. 150~151 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.66 (dd, J=7.9, 1.2 Hz, 1H), 7.37~7.29 (m, 1H), 7.23~7.10 (m, 3H), 6.99~6.88 (m, 2H), 6.88~6.78 (m, 4H), 6.72~6.59 (m, 2H), 4.62 (s, 1H), 4.26~4.18 (m, 1H), 4.17~4.02 (m, 4H), 3.32~3.21 (m, 1H), 3.00 (d, J=10.9 Hz, 1H), 1.05 (t, J=7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 177.5, 167.3, 161.8 (d, 1JFC=246.3 Hz), 160.4, 149.2, 141.1, 138.7, 135.7, 129.23, 129.18 (d, 3JFC=8.3 Hz), 129.0, 128.6, 127.8, 124.5, 121.0, 117.3, 115.5 (d, 2JFC=21.4 Hz), 114.3, 81.4, 63.4, 61.2, 57.2, 54.7, 44.9, 13.8; 19F NMR (470 MHz, CDCl3) δ: -115.6; HRMS (ESI) calcd for C29H25FNO4 [M+H]+ 470.1762, found 470.1758.
辅助材料(Supporting Information) 化合物
3aa、
3'aa、
3ab~
3al、
3an~
3aq、
3ba~
3ia、
3ka、
3ma、
4a~
4l、
4o~
4w、
4y、
5a、
6a、
7a和
7d的
1H NMR和
13C NMR图谱以及含氟化合物
3ad、
3al、
3ba、
3ia、
4d、
4l、
4p、
4w和
7d的
19F NMR图谱. 这些材料可以免费从本刊网站(
http://sioc-journal.cn/)上下载.