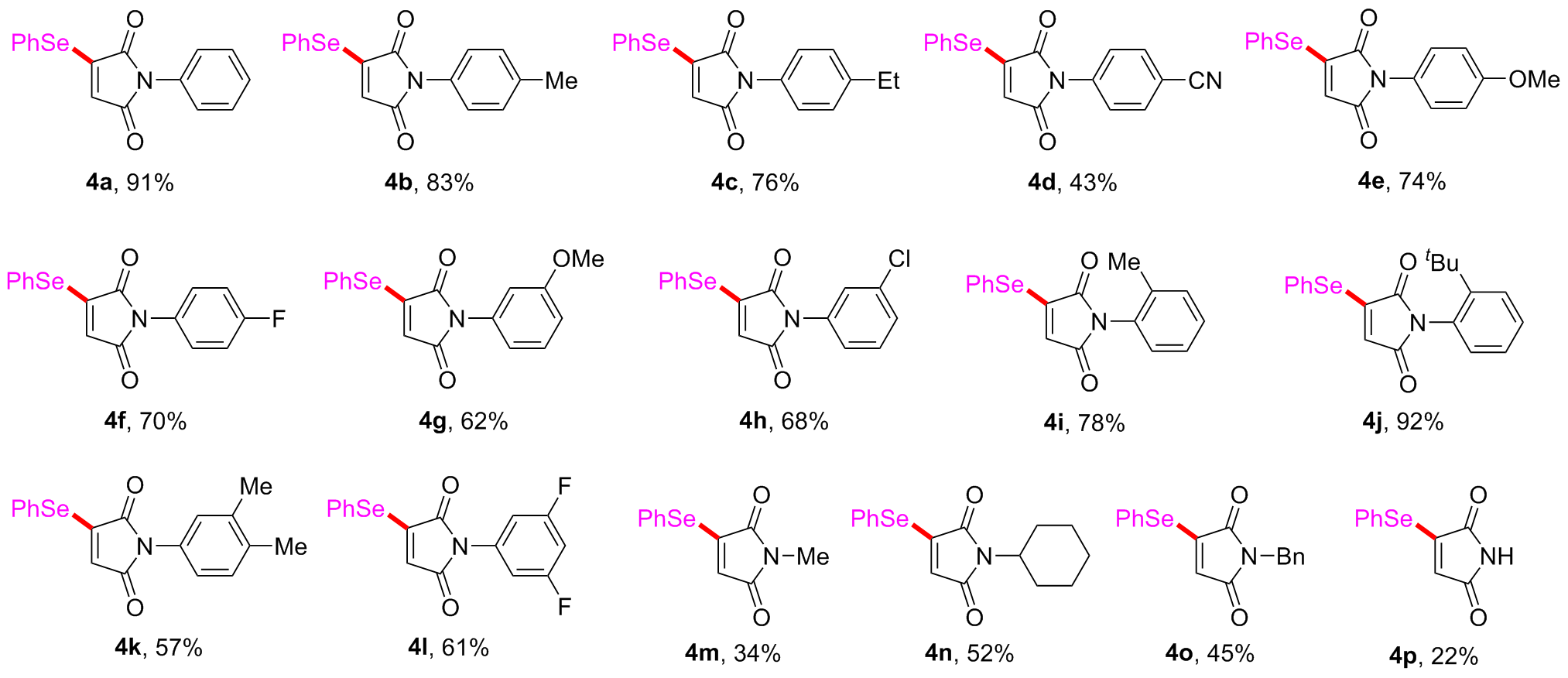
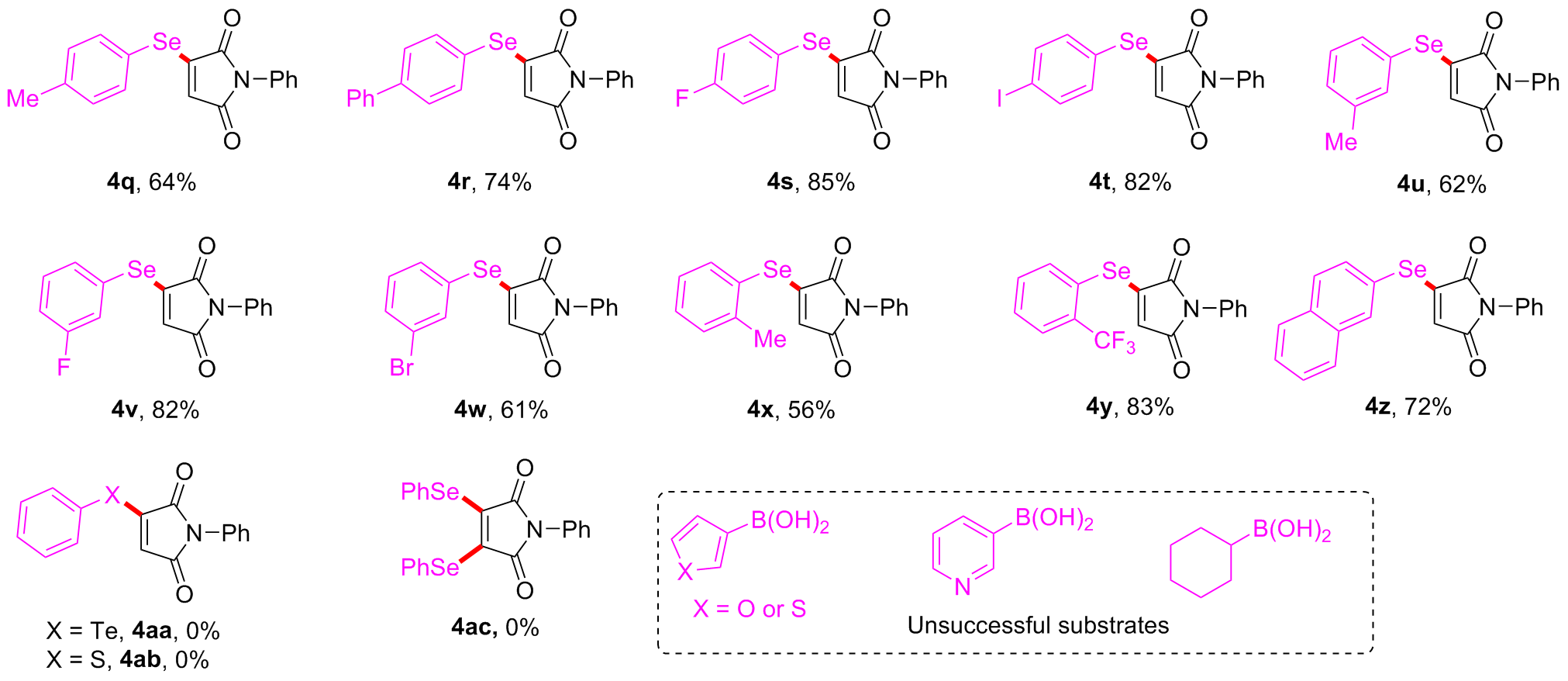
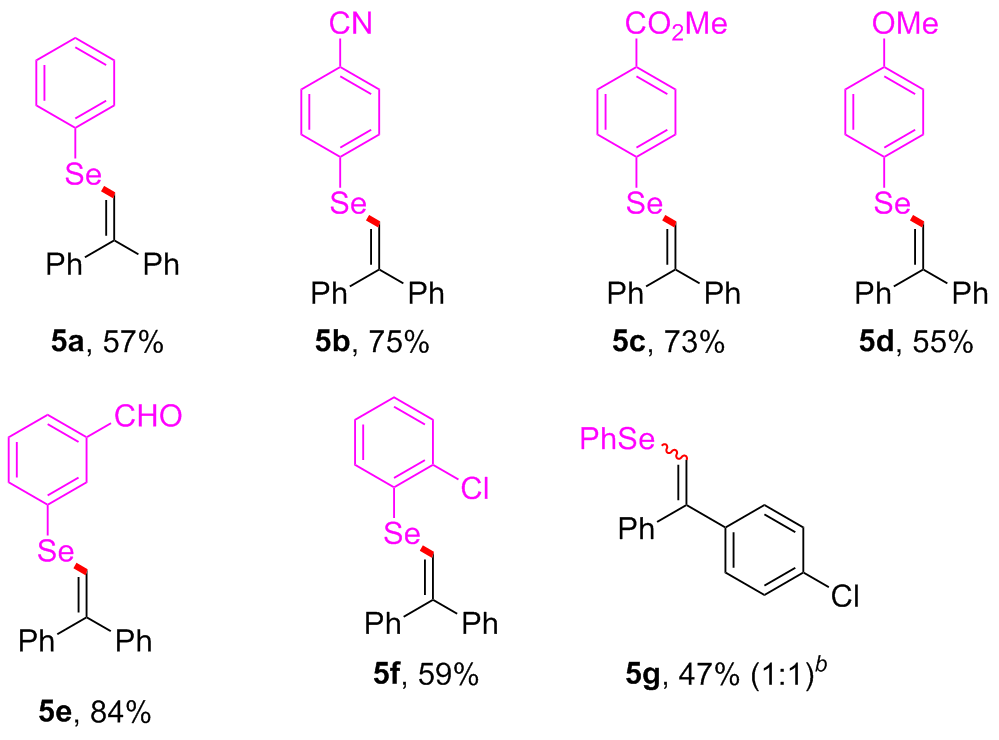
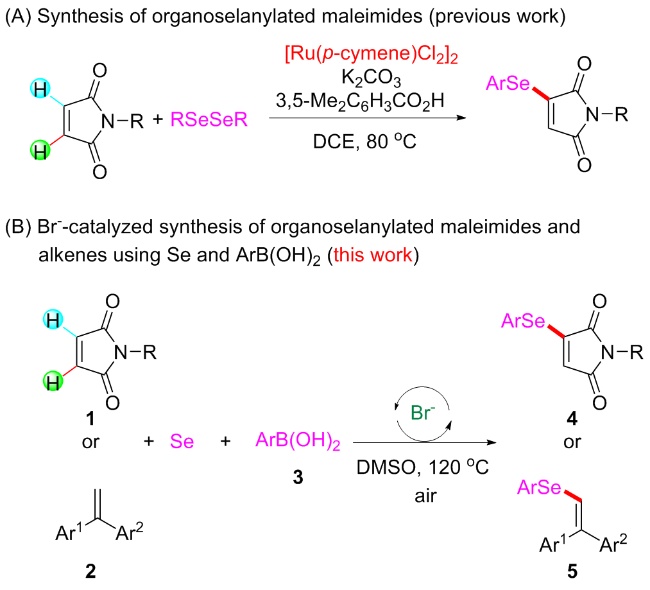
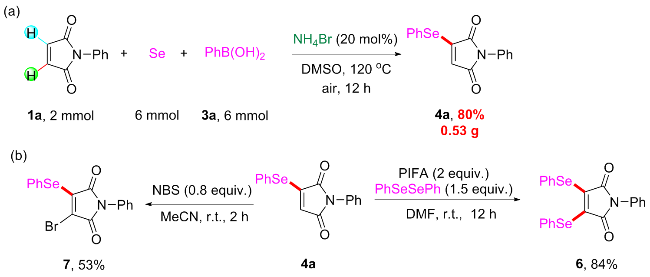
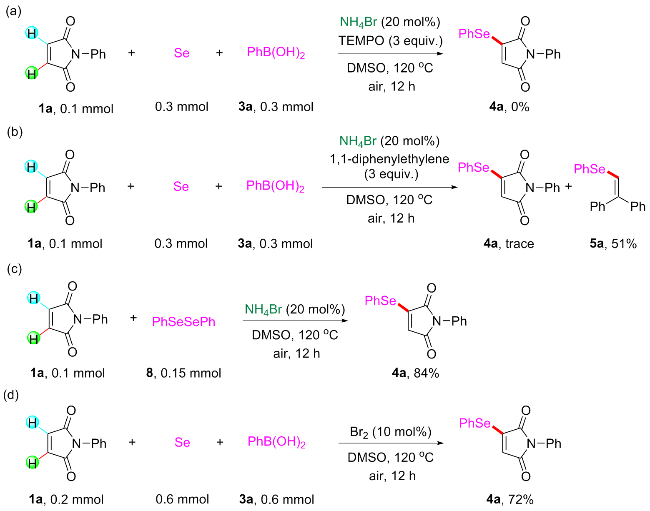
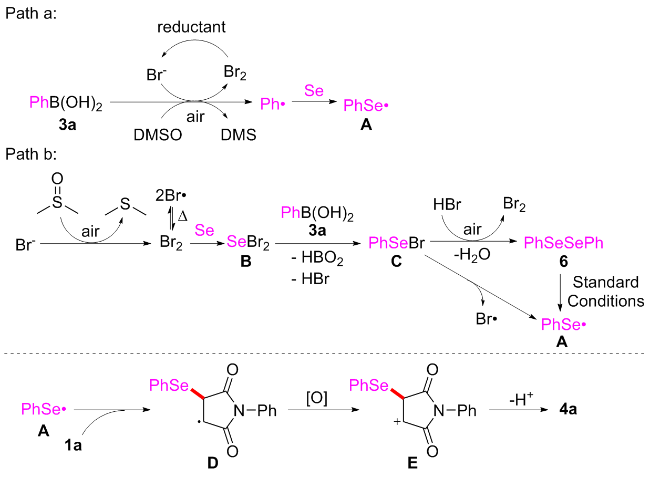
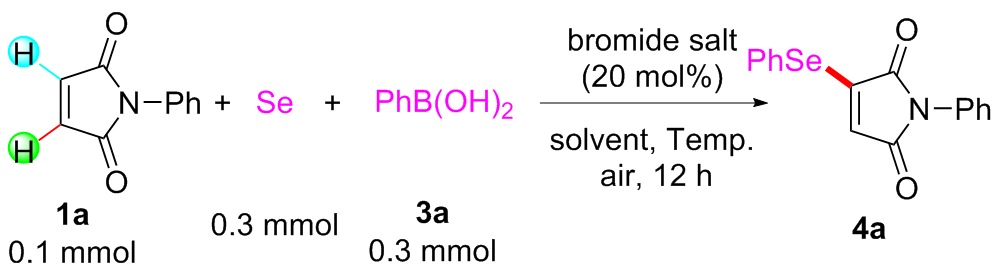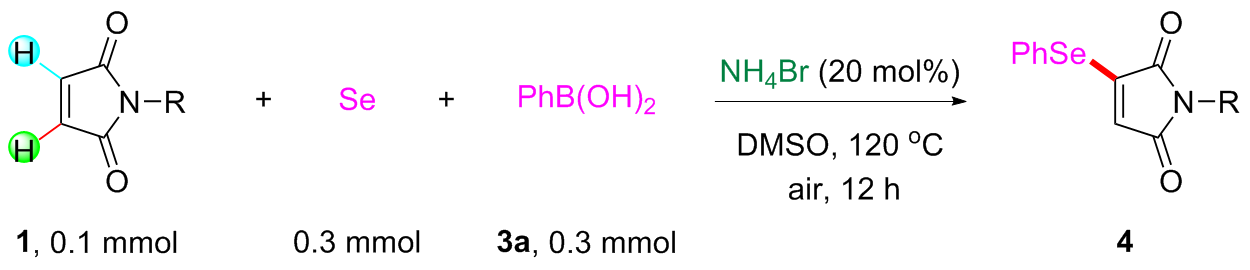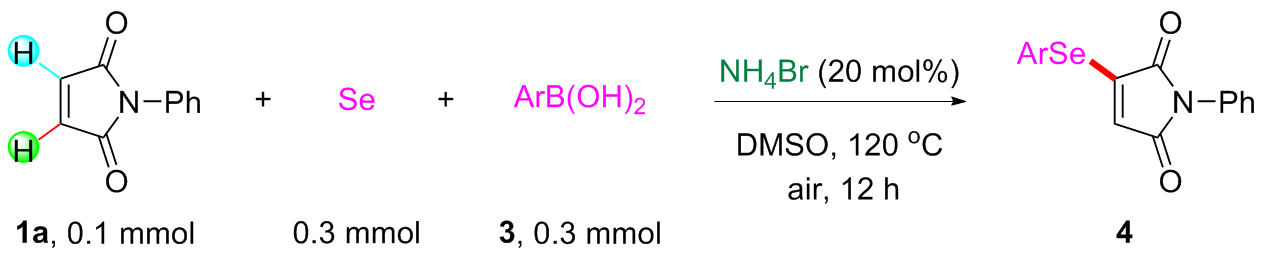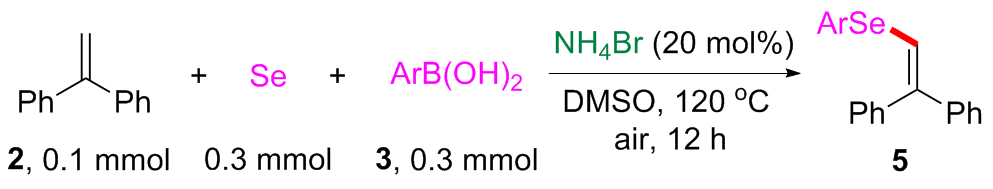Under air atmosphere, a 10 mL-Schlenk tube equipped with a stirring bar were charged with substrate 1a (0.1 mmol, 17.5 mg), PhB(OH)2 (3a, 0.3 mmol, 37.0 mg), Se (0.3 mmol, 24.9 mg) and DMSO (1 mL). The obtained mixture was stirred at 120 ℃ (oil bath) for 12 h. After the reaction was completed, the mixture was diluted with H2O (5 mL) and then extracted by ethyl acetate (EA, 5 mL×3), the combined organic phase was dried by anhydrous Na2SO4 and filtered. The filtrates were concentrated under reduced pressure to give a crude residue which was purified by flash column chromatography to provide compound 4a (29.9 mg) in 91% yield as a yellow solid. Compounds 4b~4z and 5a~5g were prepared using the same procedure.
1-Phenyl-3-(phenylselanyl)-1
H-pyrrole-2,5-dione (
4a): Yellow solid, 29.9 mg, 91% yield. m.p. 145~147 ℃ (lit.
[12] 145~146 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 7.72~7.65 (m, 2H), 7.52~7.44 (m, 5H), 7.39~7.33 (m, 3H), 6.03 (s, 1H);
13C NMR (125 MHz, CDCl
3)
δ: 168.5, 167.9, 151.3, 136.1, 131.5, 130.6, 130.4, 129.2, 128.0, 126.0, 124.6, 123.8; HRMS (ESI
+) calcd for C
16H
12NO
2Se [M+H]
+ 330.0028, found 330.0030.
3-(Phenylselanyl)-1-(p-tolyl)-1H-pyrrole-2,5-dione (4b): Yellow solid, 28.5 mg, 83% yield. m.p. 159~161 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.71~7.66 (m, 2H), 7.54~7.43 (m, 3H), 7.28~ 7.24 (m, 2H), 7.23~7.20 (m, 2H), 6.01 (s, 1H), 2.38 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 168.7, 168.0, 151.1, 138.0, 136.1, 130.5, 130.3, 129.9, 128.8, 126.0, 124.6, 123.9, 21.3; HRMS (ESI+) calcd for C17H14NO2Se [M+H]+ 344.0184, found 344.0183.
(4-Ethylphenyl)-3-(phenylselanyl)-1
H-pyrrole-2,5-dione (
4c): Yellow solid, 27.1 mg, 76% yield. m.p. 133~134 ℃(lit.
[12] 133~134 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 7.70~7.65 (m, 2H), 7.53~7.44 (m, 3H), 7.30~7.27 (m, 2H), 7.26~7.22 (m, 2H), 6.01 (s, 1H), 2.68 (q,
J=7.6 Hz, 2H), 1.25 (t,
J=7.6 Hz, 3H);
13C NMR (125 MHz, CDCl
3)
δ: 168.7, 168.0, 151.2, 144.2, 136.1, 130.5, 130.3, 129.0, 128.7, 126.0, 124.6, 123.9, 28.7, 15.6; HRMS (ESI
+) calcd for C
18H
16NO
2Se [M+H]
+ 358.0341, found 358.0344.
(2,5-Dioxo-3-(phenylselanyl)-2,5-dihydro-1H-pyrrol-1-yl)benzonitrile (4d): Yellow solid, 15.2 mg, 43% yield. m.p. 190~192 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.74 (d, J=8.5 Hz, 2H), 7.67 (d, J=7.1 Hz, 2H), 7.60 (d, J=8.5 Hz, 2H), 7.55~7.45 (m, 3H), 6.05 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 167.5, 167.2, 152.1, 136.0, 135.8, 133.1, 130.7, 130.6, 125.5, 124.7, 123.5, 118.4, 111.0; HRMS (ESI+) calcd for C17H11N2O2Se [M+H]+ 354.9980, found 354.9981.
1-(4-Methoxyphenyl)-3-(phenylselanyl)-1H-pyrrole-2,5-dione (4e): Yellow solid, 26.6 mg, 74% yield. m.p. 152~154 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.68 (d, J=6.9 Hz, 2H), 7.52~7.42 (m, 3H), 7.25~7.21 (m, 2H), 6.99~6.95 (m, 2H), 6.00 (s, 1H), 3.82 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 168.9, 168.1, 159.2, 151.1, 136.1, 130.5, 130.3, 127.6, 124.6, 124.2, 123.9, 114.6, 55.6; HRMS (ESI+) calcd for C17H14NO3Se [M+H]+ 360.0133, found 360.0135.
1-(4-Fluorophenyl)-3-(phenylselanyl)-1H-pyrrole-2,5-dione (4f): Yellow solid, 24.2 mg, 70% yield. m.p. 166~168 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.74~7.65 (m, 2H), 7.56~7.44 (m, 3H), 7.37~7.29 (m, 2H), 7.20~7.10 (m, 2H), 6.02 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.4, 167.8, 161.9 (d, J=246.3 Hz), 151.4, 136.1, 130.6, 130.4, 127.9 (d, J=8.9 Hz), 127.5 (d, J=3.3 Hz), 124.6, 123.8, 116.2 (d, J=22.5 Hz); 19F NMR (471 MHz, CDCl3) δ: 113.31; HRMS (ESI+) calcd for C16H11FNO2Se [M+H]+ 347.9934, found 347.9934.
1-(3-Methoxyphenyl)-3-(phenylselanyl)-1H-pyrrole-2,5-dione (4g): Yellow solid, 22.3 mg, 62% yield. m.p. 106~108 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.72~7.64 (m, 2H), 7.54~7.44 (m, 3H), 7.38~7.33 (m, 1H), 6.97~6.88 (m, 3H), 6.01 (s, 1H), 3.81 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 168.5, 167.8, 160.1, 151.3, 136.1, 132.5, 130.5, 130.4, 129.9, 124.7, 123.8, 118.3, 113.9, 111.8, 55.5; HRMS (ESI+) calcd for C17H14NO3Se [M+H]+ 360.0133, found 360.0131.
1-(3-Chlorophenyl)-3-(phenylselanyl)-1H-pyrrole-2,5-dione (4h): Yellow solid, 24.6 mg, 68% yield. m.p. 118~120 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.70~7.65 (m, 2H), 7.53~7.45 (m, 3H), 7.43~7.37 (m, 2H), 7.34~7.31 (m, 1H), 7.30~7.27 (m, 1H), 6.02 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.0, 167.5, 151.6, 136.1, 134.7, 132.7, 130.6, 130.4, 130.1, 128.0, 126.0, 124.6, 123.9, 123.7; HRMS (ESI+) calcd for C16H11ClNO2Se [M+H]+ 363.9638, found 363.9636.
3-(Phenylselanyl)-1-(o-tolyl)-1H-pyrrole-2,5-dione (4i): Yellow solid, 26.8 mg, 78% yield. m.p. 78~80 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.73~7.68 (m, 2H), 7.54~7.44 (m, 3H), 7.36~7.26 (m, 3H), 7.12 (d, J=7.7 Hz, 1H), 6.04 (s, 1H), 2.19 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 168.7, 167.9, 151.2, 136.7, 136.1, 131.3, 130.5, 130.34, 130.28, 129.5, 128.8, 127.0, 124.8, 123.9, 18.1; HRMS (ESI+) calcd for C17H14NO2Se [M+H]+ 344.0184, found 344.0187.
1-(2-(tert-Butyl)phenyl)-3-(phenylselanyl)-1H-pyrrole-2,5-dione (4j): Yellow solid, 35.4 mg, 92% yield. m.p. 104~106 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.74~7.68 (m, 2H), 7.58 (dd, J=8.1, 1.3 Hz, 1H), 7.54~7.45 (m, 3H), 7.41~7.37 (m, 1H), 7.28 (dd, J=7.6, 1.4 Hz, 1H), 6.93 (dd, J=7.7, 1.4 Hz, 1H), 6.08 (s, 1H), 1.32 (s, 9H); 13C NMR (125 MHz, CDCl3) δ: 169.9, 169.0, 151.7, 149.7, 136.0, 131.5, 130.5, 130.3, 129.9, 129.6, 128.7, 127.4, 125.4, 123.9, 35.6, 31.7; HRMS (ESI+) calcd for C20H20NO2Se [M+H]+ 386.0654, found 386.0653.
1-(3,4-Dimethylphenyl)-3-(phenylselanyl)-1H-pyrrole-2,5-dione (4k): Yellow solid, 20.3 mg, 57% yield. m.p. 95~97 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.71~7.66 (m, 2H), 7.53~7.44 (m, 3H), 7.21 (d, J=8.0 Hz, 1H), 7.09 (d, J=1.7 Hz, 1H), 7.04 (dd, J=8.0, 2.1 Hz, 1H), 6.01 (s, 1H), 2.28 (s, 3H), 2.28 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 168.8, 168.1, 151.1, 137.7, 136.9, 136.1, 130.5, 130.3, 130.0, 129.0, 127.3, 124.6, 123.9, 123.7, 20.0, 19.6; HRMS (ESI+) calcd for C18H16NO2Se [M+H]+ 358.0341, found 358.0339.
1-(3,5-Difluorophenyl)-3-(phenylselanyl)-1H-pyrrole-2,5-dione (4l): Yellow solid, 22.2 mg, 61% yield. m.p. 173~174 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.70~7.64 (m, 2H), 7.55~7.45 (m, 3H), 7.09~7.02 (m, 2H), 6.80 (tt, J=8.8, 2.3 Hz, 1H), 6.02 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 167.5, 167.2, 162.9 (dd, J=246.3, 13.8 Hz), 151.9, 136.1, 133.7 (dd, J=1.4, 0.8 Hz), 130.63, 130.55, 124.7, 123.6, 108.7 (dd, J=22.5, 7.5 Hz), 103.2 (dd, J=25.0, 0.8 Hz); 19F NMR (471 MHz, CDCl3) δ: 108.22; HRMS (ESI+) calcd for C16H10F2NO2Se [M+H]+ 365.9839, found 365.9843.
1-Methyl-3-(phenylselanyl)-1H-pyrrole-2,5-dione (4m): Yellow solid, 9.1 mg, 34% yield. m.p. 104~105 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.65~7.60 (m, 2H), 7.51~7.41 (m, 3H), 5.88 (s, 1H), 3.02 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 169.9, 169.2, 150.6, 136.0, 130.4, 130.2, 124.7, 124.0, 24.2; HRMS (ESI+) calcd for C11H10NO2Se [M+H]+ 267.9871, found 267.9872.
1-Cyclohexyl-3-(phenylselanyl)-1
H-pyrrole-2,5-dione (
4n): Yellow solid, 17.4 mg, 52% yield. m.p. 77~78 ℃(lit.
[17] 76~77 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 7.66~7.60 (m, 2H), 7.50~7.40 (m, 3H), 5.81 (s, 1H), 3.88 (tt,
J=12.4, 3.9 Hz, 1H), 2.09~1.97 (m, 2H), 1.85~1.79 (m, 2H), 1.70~1.61 (m, 3H), 1.38~1.15 (m, 3H);
13C NMR (125 MHz, CDCl
3)
δ: 170.0, 169.1, 150.4, 136.0, 130.4, 130.1, 124.5, 124.0, 51.2, 30.1, 26.1, 25.2; HRMS (ESI
+) calcd for C
16H
18NO
2Se [M+H]
+ 336.0497, found 336.0497.
1-Benzyl-3-(phenylselanyl)-1
H-pyrrole-2,5-dione (
4o): Yellow solid, 15.4 mg, 45% yield. m.p. 125~127 ℃ (lit.
[17] 125~126 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 7.64~7.60 (m, 2H), 7.50~7.40 (m, 3H), 7.38~7.27 (m, 5H), 5.88 (s, 1H), 4.67 (s, 2H);
13C NMR (125 MHz, CDCl
3)
δ: 169.4, 168.8, 150.7, 136.4, 136.0, 130.4, 130.2, 128.8, 128.7, 128.0, 124.7, 123.9, 41.9; HRMS (ESI
+) calcd for C
17H
14NO
2Se [M+H]
+ 344.0184, found 344.0187.
3-(Phenylselanyl)-1H-pyrrole-2,5-dione (4p): Yellow solid, 5.6 mg, 22% yield. m.p. 126~128 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.76 (s, 1H), 7.67~7.61 (m, 2H), 7.51~7.41 (m, 3H), 5.87 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 169.4, 168.9, 151.7, 136.0, 130.5, 130.3, 125.7, 123.9; HRMS (ESI+) calcd for C10H8NO2Se [M+H]+ 253.9715, found 253.9717.
1-Phenyl-3-(
p-tolylselanyl)-1
H-pyrrole-2,5-dione (
4q): Yellow solid, 22.0 mg, 64% yield. m.p. 120~122 ℃(lit.
[17] 120~121 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 7.55 (d,
J=8.0 Hz, 2H), 7.45 (dd,
J=14.3, 6.6 Hz, 2H), 7.38~7.32 (m, 3H), 7.26 (d,
J=8.0 Hz, 2H), 6.01 (s, 1H), 2.41 (s, 3H);
13C NMR (125 MHz, CDCl
3)
δ: 168.6, 167.9, 151.7, 140.8, 136.0, 131.6, 131.3, 129.2, 127.9, 126.0, 124.6, 120.2, 21.5; HRMS (ESI
+) calcd for C
17H
14NO
2Se [M+H]
+ 344.0184, found 344.0185.
3-([1'-Biphenyl]-4-ylselanyl)-1-phenyl-1H-pyrrole-2,5-dione (4r): Yellow solid, 30.0 mg, 74% yield. m.p. 146~147 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.75 (d, J=8.2 Hz, 2H), 7.68 (d, J=8.2 Hz, 2H), 7.61 (d, J=7.4 Hz, 2H), 7.46~7.40 (m, 5H), 7.39~7.33 (m, 3H), 6.12 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.6, 167.9, 151.2, 143.5, 139.8, 136.5, 131.6, 129.3, 129.2, 128.3, 128.0, 127.3, 126.1, 124.8, 122.5; HRMS (ESI+) calcd for C22H16NO2Se [M+H]+ 406.0341, found 406.0342.
3-((4-Fluorophenyl)selanyl)-1-phenyl-1H-pyrrole-2,5-dione (4s): Yellow solid, 29.4 mg, 85% yield. m.p. 134~135 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.70~7.64 (m, 2H), 7.48~7.43 (m, 2H), 7.38~7.32 (m, 3H), 7.21~7.14 (m, 2H), 6.00 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.3, 167.7, 164.1 (d, J=250.0 Hz), 151.0, 138.3 (d, J=8.6 Hz), 131.5, 129.3, 128.0, 126.0, 124.8, 118.5 (d, J=3.6 Hz), 118.0 (d, J=21.3 Hz); 19F NMR (471 MHz, CDCl3) δ: 109.27; HRMS (ESI+) calcd for C16H11FNO2Se [M+ H]+ 347.9934, found 347.9934.
3-((4-Iodophenyl)selanyl)-1-phenyl-1H-pyrrole-2,5-dione (4t): Yellow solid, 37.2 mg, 82% yield. m.p. 173~174 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.85~7.76 (m, 2H), 7.50~7.44 (m, 2H), 7.43~7.39 (m, 2H), 7.38~7.32 (m, 3H), 6.05 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.3, 167.7, 150.3, 139.7, 137.7, 131.5, 129.3, 128.0, 126.0, 124.9, 123.5, 97.1; HRMS (ESI+) calcd for C16H11INO2Se [M+H]+ 455.8994, found 455.8992.
1-Phenyl-3-(
m-tolylselanyl)-1
H-pyrrole-2,5-dione (
4u): Yellow solid, 21.3 mg, 62% yield. m.p. 106~108 ℃ (lit.
[13] 107~108 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 7.53~7.43 (m, 4H), 7.38~7.28 (m, 5H), 6.04 (s, 1H), 2.40 (s, 3H);
13C NMR (125 MHz, CDCl
3)
δ: 168.6, 167.9, 151.4, 140.6, 136.5, 133.0, 131.6, 131.1, 130.3, 129.2, 127.9, 126.0, 124.7, 123.6, 21.4; HRMS (ESI
+) calcd for C
17H
14NO
2Se [M+H]
+ 344.0184, found 344.0182.
3-((3-Fluorophenyl)selanyl)-1-phenyl-1H-pyrrole-2,5-dione (4v): Yellow solid, 28.4 mg, 82% yield. m.p. 106~108 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.52~7.41 (m, 5H), 7.39~7.32 (m, 3H), 7.25~7.19 (m, 1H), 6.08 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.3, 167.7, 163.1 (d, J=251.3 Hz), 150.2, 131.9, 131.82 (d, J=3.8 Hz), 131.77, 131.5, 129.3, 128.0, 126.0, 124.9, 123.0 (d, J=22.5 Hz), 117.7 (d, J=20.5 Hz); 19F NMR (471 MHz, CDCl3) δ: 109.30; HRMS (ESI+) calcd for C16H11FNO2Se [M+H]+ 347.9934, found 347.9936.
3-((3-Bromophenyl)selanyl)-1-phenyl-1H-pyrrole-2,5-dione (4w): Yellow solid, 24.8 mg, 61% yield. m.p. 137~139 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.86 (t, J=1.7 Hz, 1H), 7.68~7.60 (m, 2H), 7.48~7.44 (m, 2H), 7.39~7.33 (m, 4H), 6.08 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.3, 167.7, 150.2, 138.5, 134.6, 133.6, 131.8, 131.5, 129.3, 128.1, 126.0, 125.4, 125.0, 123.9; HRMS (ESI+) calcd for C16H11BrNO2Se [M+H]+ 407.9133, found 407.9134.
1-Phenyl-3-(o-tolylselanyl)-1H-pyrrole-2,5-dione (4x): Yellow solid, 19.2 mg, 56% yield. m.p. 114~114 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.69 (d, J=7.6 Hz, 1H), 7.49~7.40 (m, 4H), 7.38~7.32 (m, 3H), 7.29~7.22 (m, 1H), 5.88 (s, 1H), 2.52 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 168.6, 167.9, 150.6, 142.6, 137.5, 131.6, 131.4, 131.1, 129.2, 127.9, 127.8, 126.0, 124.8, 124.5, 22.8; HRMS (ESI+) calcd for C17H14NO2Se [M+H]+ 344.0184, found 344.0185.
1-Phenyl-3-((2-(trifluoromethyl)phenyl)selanyl)-1H-pyrrole-2,5-dione (4y): Yellow solid, 32.9 mg, 83% yield. m.p. 100~102 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.94~7.86 (m, 2H), 7.74~ 7.58 (m, 2H), 7.48~7.43 (m, 2H), 7.38~7.33 (m, 3H), 5.91 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.3, 167.6, 150.1, 139.9, 134.5 (q, J=30.0 Hz), 133.4, 131.4, 130.8, 129.2, 128.0, 127.9 (q, J=5.0 Hz), 126.0, 125.1, 123.2 (q, J=272.5 Hz), 122.1; 19F NMR (471 MHz, CDCl3) δ: 60.34; HRMS (ESI+) calcd for C17H11F3NO2Se [M+H]+ 397.9902, found 397.9906.
3-(Naphthalen-2-ylselanyl)-1-phenyl-1H-pyrrole-2,5-dione (4z): Yellow solid, 27.3 mg, 72% yield. m.p. 144~145 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.23 (s, 1H), 7.92 (t, J=7.8 Hz, 2H), 7.89~7.83 (m, 1H), 7.68 (dd, J=8.5, 1.5 Hz, 1H), 7.65~7.55 (m, 2H), 7.51~7.43 (m, 2H), 7.41~7.32 (m, 3H), 6.03 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 168.5, 167.9, 151.2, 136.4, 134.3, 133.7, 131.7, 131.6, 130.3, 129.2, 128.1, 128.0, 127.9, 127.4, 126.1, 124.9, 120.9; HRMS (ESI+) calcd for C20H14NO2Se [M+H]+ 380.0184, found 380.0185.
(2,2-Diphenylvinyl)(phenyl)selane (
5a): Yellow solid, 19.2 mg, 57% yield. m.p. 80~82℃ (lit.
[17] 81.5~82 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 7.67~7.61 (m, 2H), 7.51~7.45 (m, 2H), 7.45~7.26 (m, 11H), 7.20~7.17 (m, 1H);
13C NMR (125 MHz, CDCl
3)
δ: 143.1, 141.7, 140.4, 132.6, 131.7, 129.43, 129.41, 128.6, 128.4, 128.0, 127.5, 127.33, 127.25, 122.7; HRMS (ESI
+) calcd for C
20H
17Se [M+H]
+ 337.0490, found 337.0492.
4-((2,2-Diphenylvinyl)selanyl)benzonitrile (5b): Yellow solid, 27.1 mg, 75% yield. m.p. 99~100 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.63~7.59 (m, 2H), 7.58~7.54 (m, 2H), 7.46~7.38 (m, 3H), 7.36~7.27 (m, 7H), 7.10 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 146.5, 141.1, 140.0, 139.7, 132.6, 131.3, 129.3, 128.7, 128.6, 128.4, 128.0, 127.4, 118.8, 118.5, 110.4; HRMS (ESI+) calcd for C21H16NSe [M+H]+ 362.0442, found 362.0440.
Methyl 4-((2,2-diphenylvinyl)selanyl)benzoate (5c): Yellow solid, 28.8 mg, 73% yield. m.p. 91~92 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.95 (d, J=8.4 Hz, 2H), 7.58 (d, J=8.4 Hz, 2H), 7.45~7.36 (m, 3H), 7.34~7.24 (m, 7H), 7.14 (s, 1H), 3.91 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 166.9, 145.3, 141.5, 140.3, 138.9, 131.0, 130.3, 129.4, 128.8, 128.7, 128.5, 128.3, 127.8, 127.4, 120.0, 52.3; HRMS (ESI+) calcd for C22H19O2Se [M+H]+ 395.0545, found 395.0542.
(2,2-Diphenylvinyl)(4-methoxyphenyl)selane (5d): Yellow solid, 20.1 mg, 55% yield. m.p. 85~86 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.54~7.50 (m, 2H), 7.42 (t, J=7.6 Hz, 2H), 7.38~7.31 (m, 3H), 7.26~7.18 (m, 5H), 7.03 (s, 1H), 6.86 (d, J=8.6 Hz, 2H), 3.80 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 159.8, 142.1, 141.8, 140.5, 135.2, 129.5, 128.7, 128.4, 128.0, 127.23, 127.21, 124.4, 121.7, 115.2, 55.5; HRMS (ESI+) calcd for C21H19OSe [M+H]+ 367.0596, found 367.0597.
3-((2,2-Diphenylvinyl)selanyl)benzaldehyde (5e): Yellow solid, 30.6 mg, 84% yield. m.p. 103~104 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.99 (s, 1H), 8.06 (t, J=1.4 Hz, 1H), 7.82~7.77 (m, 2H), 7.51~7.36 (m, 5H), 7.34~7.25 (m, 6H), 7.09 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 191.7, 144.7, 141.4, 140.2, 138.0, 137.3, 133.3, 133.1, 130.0, 129.4, 128.7, 128.5, 128.3, 127.7, 127.3, 120.7; HRMS (ESI+) calcd for C21H17OSe [M+H]+ 365.0439, found 365.0440.
(2-Chlorophenyl)(2,2-diphenylvinyl)selane (5f): Yellow solid, 21.8 mg, 59% yield. m.p. 93~94 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.59 (dd, J=7.6, 1.5 Hz, 1H), 7.48~7.36 (m, 4H), 7.36~7.27 (m, 7H), 7.27~7.18 (m, 2H), 7.12 (s, 1H); 13C NMR (125 MHz, CDCl3) δ: 145.8, 141.7, 140.2, 135.2, 132.7, 131.9, 129.8, 129.5, 128.7, 128.5, 128.3, 128.1, 127.8, 127.6, 127.5, 119.6; HRMS (ESI+) calcd for C20H16ClSe [M+H]+ 371.0100, found 371.0103.
(2-(4-Chlorophenyl)-2-phenylvinyl)(phenyl)selane (5g): Yellow solid, 17.4 mg, 47% yield. m.p. 86~87 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.61~7.56 (m, 2H), 7.47~7.38 (m, 3H), 7.34~7.31 (m, 3H), 7.30~7.25 (m, 3H), 7.25~7.20 (m, 2H), 7.18~7.15 (m, 1H), 7.12 (d, J=5.6 Hz, 1H); 13C NMR (125 MHz, CDCl3) δ: 142.0, 141.8, 141.4, 140.1, 140.0, 138.8, 133.9, 133.2, 132.8, 132.7, 131.43, 131.35, 130.9, 129.5, 129.4, 128.9, 128.8, 128.6, 128.51, 128.48, 128.3, 127.7, 127.6, 127.2, 123.6, 123.4; HRMS (ESI+) calcd for C20H16ClSe [M+H]+ 371.0100, found 371.0100.