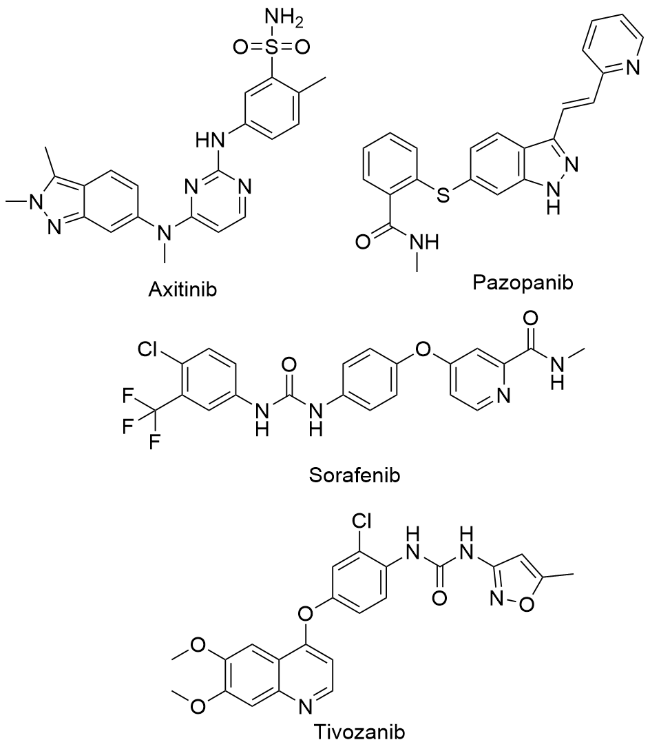
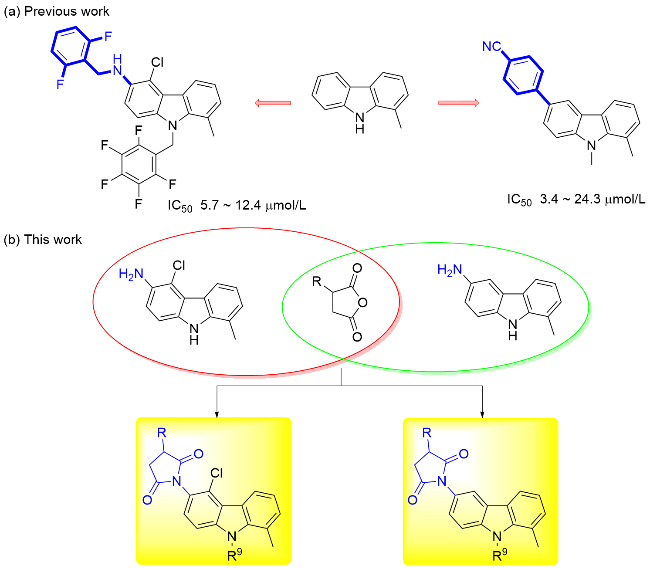
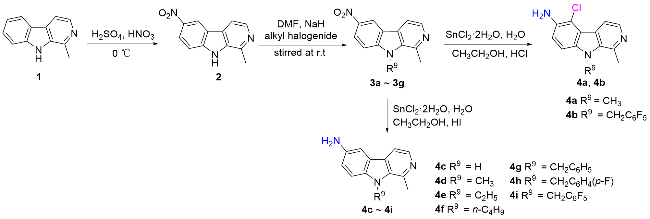
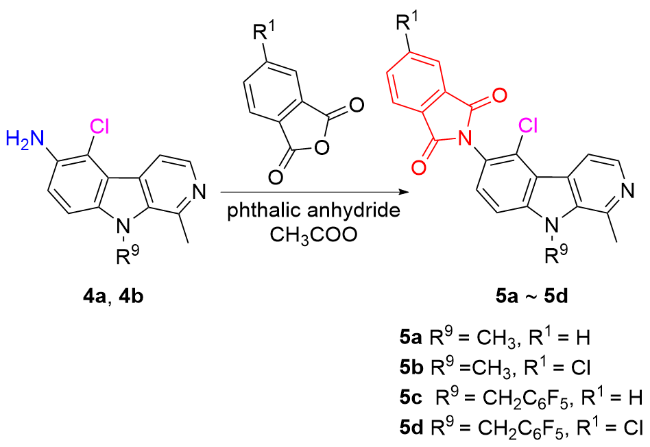
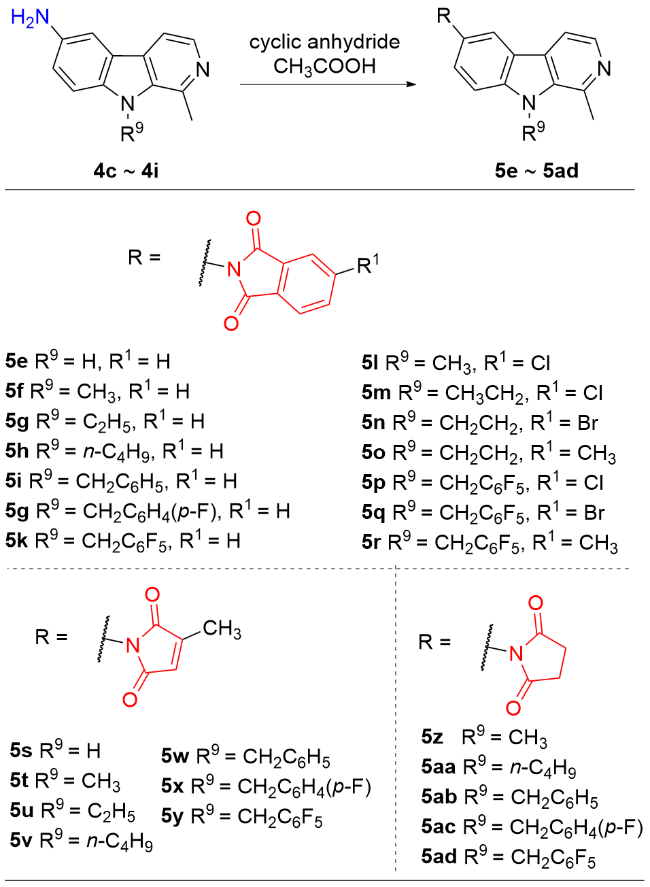
称取中间体4a (1 mmol)、邻苯二甲酸酐(1.5 mmol), 量取冰乙酸(6~8 mL)于100 mL圆底烧瓶中, 回流反应2~4 h, 薄层色谱法(TLC)监测反应进程. 反应完毕后, 将反应液冷却至室温, 用NaHCO3调节至pH大于8, 乙酸乙酯萃取(80 mL×3), 无水Na2SO4干燥, 减压浓缩, 柱层析纯化[V(二氯甲烷)∶V(甲醇)=200∶1~50∶1], 得目标产物5a. 化合物5b~5ad的合成方法与化合物5a类似.
6-(N-邻苯二甲酰亚胺基)-5-氯-1,9-二甲基-β-咔啉(5a): 白色固体, 产率88.2%. m.p. 288.2~289.9 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.42 (d, J=5.4 Hz, 1H), 8.39 (d, J=5.4 Hz, 1H), 8.02 (dd, J=5.4, 3.0 Hz, 2H), 7.85 (dd, J=5.2, 3.0 Hz, 2H), 7.58 (d, J=9.2 Hz, 1H), 7.54 (d, J=8.8 Hz, 1H), 4.23 (s, 3H), 3.213 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.23, 143.41, 141.44, 136.02, 134.56, 131.96, 129.63, 129.45, 128.70, 124.01, 121.93, 119.30, 115.55, 108.78, 32.84, 22.81. HRMS (ESI) calcd for C21H15ClN3O2 [M+H]+ 376.0847, found 376.0847.
6-(N-3-氯邻苯二甲酰亚胺基)-5-氯-1,9-二甲基-β-咔啉(5b): 白色固体, 产率86.9%. m.p. 205.4~207.7 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.37 (d, J=5.4 Hz, 1H), 8.28 (d, J=5.4 Hz, 1H), 7.97 (s, 1H), 7.94 (d, J=8.0 Hz, 1H), 7.79 (d, J=8.0 Hz, 1H), 7.51 (d, J=8.8 Hz, 1H), 7.47 (d, J=8.8 Hz, 1H), 4.15 (s, 3H), 3.10 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.36, 166.05, 142.95, 142.05, 141.28, 138.87, 136.13, 134.58, 133.61, 130.00, 128.86, 127.70, 125.21, 124.61, 124.38, 121.03, 119.46, 115.03, 108.63, 32.70, 23.81. HRMS (ESI) calcd for C21H14Cl2- N3O2 [M+H]+ 410.0457, found 410.0458.
6-(N-邻苯二甲酰亚胺基)-5-氯-1-甲基-9-(2,3,4,5,6-五氟苄基)-β-咔啉(5c): 白色固体, 产率86.8%. m.p. 226.5~227.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.45 (d, J=5.4 Hz, 1H), 8.38 (d, J=5.4 Hz, 1H), 8.00 (dd, J=5.4, 3.0 Hz, 2H), 7.84 (dd, J=5.4, 3.0 Hz, 2H), 7.48 (d, J=8.8 Hz, 1H), 7.37 (d, J=8.8 Hz, 1H), 5.98 (s, 2H), 3.13 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.18, 146.45 (m), 144.01 (m), 142.62 (m), 141.86, 141.52, 140.07 (m), 139.24, 136.58 (m), 135.84, 134.60, 131.90, 129.53, 129.46, 128.40, 124.03, 122.65, 120.31, 115.40, 109.75 (m), 108.38, 38.09, 23.65; 19F NMR (376 MHz, CDCl3) δ: -142.04, -152.30, -160.10. HRMS (ESI) calcd for C27H14ClF5N3O2 [M+H]+ 542.0689, found 542.0689.
6-(N-3-氯邻苯二甲酰亚胺基)-5-氯-1-甲基-9-(2,3,4,5,6-五氟苄基)-β-咔啉(5d): 白色固体, 产率90.1%. m.p. 218.9~219.7 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.45 (d, J=5.4 Hz, 1H), 8.36 (d, J=5.4 Hz, 1H), 7.97 (s, 1H), 7.94 (d, J=8.0 Hz, 1H), 7.80 (d, J=8.0 Hz, 1H), 7.47 (d, J=8.8 Hz, 1H), 7.36 (d, J=8.8 Hz, 1H), 5.97 (s, 2H), 3.12 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.21, 165.90, 146.46 (m), 144.02 (m), 142.66 (m), 141.91, 141.67, 141.40, 139.63, 139.13 (m), 136.60 (m), 135.96, 134.66, 133.54, 129.92, 129.39, 129.28, 128.34, 125.28, 124.47, 122.25, 120.51, 115.36, 109.69 (m), 108.39, 38.14, 24.09; 19F NMR (376 MHz, CDCl3) δ: -141.99, -152.14, -160.01. HRMS (ESI) calcd for C27H13Cl2F5N3O2 [M+H]+ 576.0299, found 576.0299.
6-(N-邻苯二甲酰亚胺基)-1-甲基-β-咔啉(5e): 白色固体, 产率68.7%. m.p.>290 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.69 (s, 1H), 8.35 (s, 1H), 8.23~7.64 (m, 5H), 7.53 (s, 2H), 5.30 (s, 1H), 2.83 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.95, 142.06, 139.32, 139.13, 135.06, 134.50, 131.85, 128.03, 126.97, 123.81, 122.49, 120.69, 113.04, 112.13, 20.36. HRMS (ESI) calcd for C20H14N3O2 [M+H]+ 328.1080, found 328.1084.
6-(N-邻苯二甲酰亚胺基)-1,9-二甲基-β-咔啉(5f): 白色固体, 产率75.7%. m.p. 265.4~267.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.34 (d, J=5.4 Hz, 1H), 8.18 (d, J=1.6 Hz, 1H), 7.99 (dd, J=5.4, 3.0 Hz, 2H), 7.85 (d, J=5.2 Hz, 1H), 7.83 (dd, J=5.4, 3.0 Hz, 2H), 7.67 (dd, J=8.8, 1.6 Hz, 1H), 7.59 (d, J=8.8 Hz, 1H), 4.20 (s, 3H), 3.17 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.81, 141.69, 141.55, 136.19, 134.48, 131.83,129.23, 127.41, 123.93, 123.79, 121.23, 120.42, 113.47, 110.16, 32.55, 22.77. HRMS (ESI) calcd for C21H15N3O2 [M+H]+ 342.1237, found 342.1237.
6-(N-邻苯二甲酰亚胺基)-1-甲基-9-乙基-β-咔啉(5g): 白色固体, 产率70.5%. m.p. 260.2~261.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.33 (d, J=5.2 Hz, 1H), 8.16 (s, 1H), 7.98 (dd, J=5.4, 3.0 Hz, 2H), 7.87~7.74 (m, 3H), 7.61 (dd, J=8.8, 1.2 Hz, 1H), 7.57 (d, J=8.8 Hz, 1H), 4.63 (q, J=7.2 Hz, 2H), 3.07 (s, 3H), 1.48 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.88, 141.48, 140.42, 138.13, 135.36, 134.41, 131.85, 128.88, 126.92, 123.73, 123.56, 121.74, 120.40, 113.21, 110.07, 39.75, 23.31, 15.73. HRMS (ESI) calcd for C22H18N3O2 [M+H]+ 356.1393, found 356.1393.
6-(N-邻苯二甲酰亚胺基)-1-甲基-9-正丁基-β-咔啉(5h): 白色固体, 产率60.3%. m.p. 152.3~153.2 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.35 (d, J=5.4 Hz, 1H), 8.17 (s, 1H), 7.99 (dd, J=5.4, 3.0 Hz, 2H), 7.90~7.72 (m, 3H), 7.62 (dd, J=8.8, 1.6 Hz, 1H), 7.57 (d, J=8.8 Hz, 1H), 4.70~4.45 (m, 2H), 3.08 (s, 3H), 1.90~1.81 (m, 2H), 1.52~1.43 (m, 2H), 1.00 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.88, 141.41, 140.93, 137.76, 135.61, 134.44, 131.85, 129.06, 126.96, 123.76, 123.59, 121.55, 120.36, 113.34, 110.35, 44.99, 32.93, 23.03, 20.22, 13.88. HRMS (ESI) calcd for C24H22N3O2 [M+H]+ 384.1706, found 384.1701.
6-(N-邻苯二甲酰亚胺基)-1-甲基-9-苄基-β-咔啉(5i): 白色固体, 产率62.8%. m.p. 177.4~178.6 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.35 (d, J=5.2 Hz, 1H), 8.19 (d, J=1.6 Hz,, 1H), 7.96 (dd, J=5.4, 3.0 Hz, 2H), 7.81 (d, J=5.4 Hz, 1H), 7.85~7.75 (m, 3H), 7.54 (dd, J=8.8, 1.8 Hz, 1H), 7.44 (d, J=8.8 Hz, 1H), 7.27 (q, J=6.8 Hz, 3H), 6.99 (d, J=6.8 Hz, 2H), 5.76 (s, 2H), 2.87 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.83, 141.82, 141.32, 138.22, 137.45, 136.02, 134.47, 131.82, 129.14, 127.76, 127.32, 125.42, 124.12, 123.79, 121.64, 120.36, 113.37, 110.53, 48.45, 22.82. HRMS (ESI) calcd for C27H20N3O2 [M+H]+ 418.1550, found 418.1552.
6-(N-邻苯二甲酰亚胺基)-1-甲基-9-(4-氟苄基)-β-咔啉(5j): 白色固体, 产率76.2%. m.p. 238.4~240.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.39 (d, J=5.4 Hz, 1H), 8.22 (d, J=1.6 Hz, 1H), 7.98 (dd, J=5.4, 3.0 Hz, 2H), 7.87 (d, J=5.4 Hz, 1H), 7.82 (dd, J=5.4, 3.0 Hz, 2H), 7.59 (dd, J=8.8, 1.6 Hz, 1H), 7.46 (d, J=8.8 Hz, 1H), 7.09~6.90 (m, 4H), 5.77 (s, 2H), 2.91 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.81, 163.49 (d, J=245.0 Hz), 141.55, 141.22, 138.10, 135.83, 134.51, 133.10 (d, J=3.1 Hz), 131.80, 129.36, 127.50, 127.09 (d, J=8.1 Hz), 124.32, 123.82, 121.64, 120.49, 116.13 (d, J=21.6 Hz), 113.50, 110.42, 47.82, 22.42. HRMS (ESI) calcd for C27H19FN3O2 [M+H]+ 436.1455, found 436.1455.
6-(N-邻苯二甲酰亚胺基)-1-甲基-9-(2,3,4,5,6-五氟苄基)-β-咔啉(5k): 白色固体, 产率78.3%. m.p.>290 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.41 (d, J=5.2 Hz, 1H), 8.17 (s, 1H), 7.99 (dd, J=5.4, 3.0 Hz, 2H), 7.92~7.69 (m, 3H), 7.58 (dd, J=8.8, 1.8 Hz, 1H), 7.44 (d, J=8.8 Hz, 1H), 5.98 (s, 2H), 3.12 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.72, 146.50 (m), 144.07 (m), 142.51 (m), 141.63, 140.18, 139.05, 136.54 (m), 136.12, 134.52, 131.77, 129.27, 127.27, 124.65, 123.83, 122.44, 120.43, 113.42, 110.11 (m), 109.89, 43.45, 38.02, 23.84. HRMS (ESI) calcd for C27H15F5N3O2 [M+H]+ 508.1078, found 508.1079.
6-(N-3-氯邻苯二甲酰亚胺基)-1,9-二甲基-β-咔啉(5l): 白色固体, 产率73.6%. m.p. 278.9~279.4 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.33 (d, J=5.2 Hz, 1H), 8.14 (s, 1H), 7.95 (s, 1H), 7.92 (d, J=8.0 Hz, 1H), 7.79 (t, J=6.4 Hz, 2H), 7.61 (d, J=7.8 Hz, 1H), 7.56 (d, J=8.8 Hz, 1H), 4.18 (s, 3H), 3.12 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.90, 166.58, 142.04, 141.46, 141.17, 138.25 136.36, 134.51, 133.51, 129.89, 128.60, 126.77, 125.00, 124.17, 123.28, 121.45, 120.22, 113.18, 110.08, 32.51, 23.54. HRMS (ESI) calcd for C21H15ClN3O2 [M+H]+ 376.0847, found 376.0848.
6-(N-3-氯邻苯二甲酰亚胺基)-1-甲基-9-乙基-β-咔啉(5m): 白色固体, 产率71.1%. m.p. 227.6~228.9 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.34 (d, J=5.4 Hz, 1H), 8.15 (s, 1H), 7.95 (d, J=1.4 Hz, 1H), 7.92 (d, J=8.0 Hz, 1H), 7.81 (d, J=5.2 Hz, 1H), 7.78 (dd, J=8.0, 1.6 Hz, 1H), 7.58 (s, 2H), 4.65 (q, J=7.2 Hz, 2H), 3.08 (s, 3H), 1.49 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.92, 166.60, 141.55, 141.16, 140.49, 138.22, 135.41, 134.50, 133.51, 129.89, 128.86, 126.74, 125.00, 124.17, 123.26, 121.78, 120.33, 113.22, 110.14, 39.79, 23.32, 15.73. HRMS (ESI) calcd for C22H17ClN3O2 [M+H]+ 390.1003, found 390.1006.
6-(N-3-溴邻苯二甲酰亚胺基)-1-甲基-9-乙基-β-咔啉(5n): 白色固体, 产率79.0%. m.p. 225.7~226.4 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.36 (d, J=5.4 Hz, 1H), 8.15 (s, 1H), 8.10 (s, 1H), 7.94 (d, J=8.0 Hz, 1H), 7.82 (dd, J=9.2, 6.8 Hz, 2H), 7.58 (t, J=6.2 Hz, 2H), 4.63 (q, J=7.2 Hz, 2H), 3.09 (s, 3H), 1.48 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.04, 166.51, 141.28, 140.63, 137.61, 137.48, 135.31, 133.44, 130.33, 129.39, 129.13, 127.08, 126.97, 125.12, 123.38, 121.63, 120.41, 113.37, 110.19, 39.82, 22.78, 15.75. HRMS (ESI) calcd for C22H17BrN3O2 [M+H]+ 434.0498, found 434.0499.
6-(N-3-甲基邻苯二甲酰亚胺基)-1-甲基-9-乙基-β-咔啉(5o): 白色固体, 产率80.6%. m.p. 255.6~256.7 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.35 (d, J=5.4 Hz, 1H), 8.16 (s, 1H), 7.87 (d, J=7.6 Hz, 1H), 7.83 (d, J=5.2 Hz, 1H), 7.79(s, 1H), 7.66~7.54 (m, 3H), 4.66 (q, J=7.2 Hz, 2H), 3.09 (s, 3H), 2.57 (s, 3H), 1.49 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 168.09, 167.97, 145.80, 141.45, 140.45, 138.04, 135.40, 135.00, 132.24, 129.26, 129.01, 127.00, 124.27, 123.74, 123.68, 121.75, 120.42, 113.29, 110.07, 39.79, 23.23, 22.11, 15.74. HRMS (ESI) calcd for C23H20N3O2 [M+H]+ 370.1550, found 370.1551.
6-(N-3-氯邻苯二甲酰亚胺基)-1-甲基-9-(2,3,4,5,6-五氟苄基)-β-咔啉(5p): 白色固体, 产率79.3%. m.p. 274.3~275.6 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.41 (d, J=5.2 Hz, 1H), 8.16 (s, 1H), 7.96 (s, 1H), 7.92 (d, J=8.0 Hz, 1H), 7.85 (d, J=5.2 Hz, 1H), 7.79 (dd, J=8.0, 1.6 Hz, 1H), 7.56 (d, J=8.8 Hz, 1H), 7.45(d, J=8.8 Hz, 1H), 5.99 (s, 2H), 3.12 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.75, 166.44, 146.54 (m), 141.65, 141.30, 140.29, 139.05 (m), 136.60 (m), 136.14, 134.61, 133.42, 129.79, 129.29, 127.15, 125.09, 124.38, 124.26, 122.45, 120.36, 113.45, 109.97, 38.03, 23.79; 19F NMR (376 MHz, CDCl3) δ: -142.12, -152.54, -160.43. HRMS (ESI) calcd for C27H14ClF5N3O2 [M+H]+ 542.0689, found 542.0688.
6-(N-3-溴邻苯二甲酰亚胺基)-1-甲基-9-(2,3,4,5,6-五氟苄基)-β-咔啉(5q): 白色固体, 产率68.2%. m.p. 289.4~290.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.41 (d, J=5.4 Hz, 1H), 8.18 (d, J=1.6 Hz, 1H), 7.94~7.88 (m, 1H), 7.85 (d, J=5.2 Hz, 1H), 7.74 (d, J=3.8 Hz, 2H), 7.53 (dd, J=8.8, 1.6 Hz, 1H), 7.45 (d, J=8.8 Hz, 1H), 5.98 (s, 2H), 3.14 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.90, 166.37, 146.49 (m), 144.05 (m), 142.46 (m), 141.72, 140.20, 139.23, 137.55, 136.61 (m), 136.13, 133.37, 130.26, 129.48, 129.13, 127.16, 127.04, 125.17, 124.28, 122.48, 120.33, 113.37, 110.10 (m), 109.93, 38.01, 23.93; 19F NMR (376 MHz, CDCl3) δ: -142.13, -152.61, -160.32. HRMS (ESI) calcd for C27H14BrF5N3O2 [M+H]+ 587.0262, no found.
6-(N-3-甲基邻苯二甲酰亚胺基)-1-甲基-9-(2,3,4,5,6-五氟苄基)-β-咔啉(5r): 白色固体, 产率70.6%. m.p. 284.4~285.2 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.42 (d, J=5.4 Hz, 1H), 8.19 (s, 1H), 7.86 (d, J=7.8 Hz, 2H), 7.79 (s, 1H), 7.61 (d, J=7.6 Hz, 2H), 7.45 (d, J=8.8 Hz, 1H), 5.99 (s, 2H), 3.16 (s, 3H), 2.57 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.91, 167.79, 146.53 (m), 145.90, 144.05 (m), 142.43 (m), 141.60, 140.06, 139.05, 139.04 (m), 136.63 (m), 136.05, 135.06, 132.14, 129.16, 127.26, 124.76, 124.29, 123.71, 122.38, 120.40, 113.36, 110.18 (m), 109.82, 37.95, 23.82, 22.10; 19F NMR (376 MHz, CDCl3) δ: -142.15, -152.81, -160.43. HRMS (ESI) calcd for C28H17F5N3O2 [M+H]+ 446.1299, no found.
6-(N-衣康酰亚胺基)-1-甲基-β-咔啉(5s): 白色固体, 产率51.4%. m.p. >290 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.79 (s, 1H), 8.24 (d, J=5.2 Hz, 1H), 8.15 (s, 1H), 7.94 (d, J=5.2 Hz, 1H), 7.68 (d, J=8.6 Hz, 1H), 7.45 (dd, J=8.8, 1.6 Hz, 1H), 6.84 (d, J=1.2 Hz, 1H), 2.79 (s, 3H), 2.12 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.27, 170.26, 145.96, 141.06, 139.97, 136.41, 134.95, 128.56, 127.52, 127.18, 123.96, 121.80, 120.43, 113.15, 112.45, 19.02, 11.29. HRMS (ESI) calcd for C17H14N3O2 [M+H]+ 292.1080, found 292.1088.
6-(N-衣康酰亚胺基)-1,9-二甲基-β-咔啉(5t): 白色固体, 产率58.7%. m.p. 239.2~240.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.32 (d, J=5.2 Hz, 1H), 8.04 (s, 1H), 7.77 (d, J=5.2 Hz, 1H), 7.53~7.48 (m, 2H), 6.53 (d, J=1.6 Hz, 1H), 4.15 (s, 3H), 3.10 (s, 3H), 2.22 (d, J=1.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.20, 170.17, 145.85, 142.01, 141.15, 138.25, 136.32, 128.51, 127.46, 126.52, 123.54, 121.39, 119.77, 113.12, 109.96, 32.44, 23.60, 11.26. HRMS (ESI) calcd for C21H15ClN3O2 [M+H]+ 306.1237, found 306.1231.
6-(N-衣康酰亚胺基)-1-甲基-9-乙基-β-咔啉(5u): 白色固体, 产率55.1%. m.p. 195.6~196.8 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.33 (d, J=5.2 Hz, 1H), 8.05 (s, 1H), 7.79 (d, J=5.2 Hz, 1H), 7.52 (d, J=8.8 Hz, 1H), 7.49(d, J=8.8 Hz, 1H), 6.57~6.46 (m, 1H), 4.61 (q, J=7.2 Hz, 2H), 3.06 (s, 3H), 2.21 (s, 3H), 1.45 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.22, 170.21, 145.85, 141.56, 140.18, 138.29, 135.39, 128.83, 127.46, 126.49, 123.50, 121.75, 119.91, 113.15, 110.02, 39.72, 23.42, 15.69, 11.26. HRMS (ESI) calcd for C19H18N3O2 [M+H]+ 320.1393, found 320.1392.
6-(N-衣康酰亚胺基)-1-甲基-9-正丁基-β-咔啉(5v): 白色固体, 产率44.8%. m.p. 129.3~130.9 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.33 (d, J=5.2 Hz, 1H), 8.04 (s, 1H), 7.79 (d, J=5.2 Hz, 1H), 7.56~7.44 (m, 2H), 6.61~6.44 (m, 1H), 4.60~4.46 (m, 2H), 3.05 (s, 3H), 2.28~2.14 (m, 3H), 1.86~1.78 (m, 2H), 1.50~1.40 (m, 2H), 0.98 (t, J=7.4 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 170.19, 169.18, 144.82, 140.54, 139.56, 137.15, 134.58, 127.71, 126.44, 125.40, 122.44, 120.55, 118.78, 112.11, 109.24, 43.88, 31.88, 22.47, 19.17, 12.84, 10.23. HRMS (ESI) calcd for C21H22N3O2 [M+H]+ 348.1706, found 348.1706.
6-(N-衣康酰亚胺基)-1-甲基-9-苄基-β-咔啉(5w): 白色固体, 产率52.7%. m.p. 119.3~120.7 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.13 (d, J=1.6 Hz, 1H), 7.90 (d, J=5.4 Hz, 1H), 7.51 (dd, J=8.8, 1.6 Hz, 1H), 7.45 (d, J=8.8 Hz, 1H), 7.30~7.23 (m, 4H), 6.96 (d, J=6.4 Hz, 2H), 6.54 (d, J=1.6 Hz, 1H), 5.77 (s, 2H), 2.96 (s, 3H), 2.22 (d, J=1.6Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.06, 170.03, 145.95, 141.60, 137.09, 135.83, 129.94, 129.58, 129.19, 127.85, 127.52, 125.34, 124.55, 121.15, 120.06, 113.73, 110.67, 48.49, 21.91, 11.28. HRMS (ESI) calcd for C24H20N3O2 [M+H]+ 382.1550, found 382.1557.
6-(N-衣康酰亚胺基)-1-甲基-9-(4-氟苄基)-β-咔啉(5x): 白色固体, 产率50.4%. m.p. 205.2~206.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.38 (d, J=5.2 Hz, 1H), 8.11 (s, 1H), 7.87 (d, J=5.2 Hz, 1H), 7.48 (d, J=8.8 Hz, 1H), 7.41 (d, J=8.8 Hz, 1H), 7.03~6.90 (m, 4H), 6.54 (s, 1H), 5.76 (s, 2H), 2.90 (s, 3H), 2.22 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.12, 170.10, 162.26 (d, J=246.7 Hz), 145.94, 141.56, 141.01, 138.07, 135.82, 133.10 (d, J=3.0 Hz), 129.32, 127.52, 127.12, 127.02 (d, J=8.1 Hz), 124.32, 121.60, 119.95, 116.12 (d, J=21.7 Hz), 113.47, 110.40, 47.81, 22.48, 11.28. HRMS (ESI) calcd for C27H19FN3O2 [M+H]+ 436.1455, found 436.1455.
6-(N-衣康酰亚胺基)-1-甲基-9-(2,3,4,5,6-五氟苄基)-β-咔啉(5y): 白色固体, 产率66.1%. m.p.>290 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.39 (d, J=5.2 Hz, 1H), 8.06 (s, 1H), 7.81 (d, J=5.2 Hz, 1H), 7.47 (d, J=8.8 Hz, 1H), 7.38 (d, J=8.8 Hz, 1H), 6.52 (d, J=1.2Hz, 1H), 5.95 (s, 2H), 3.10 (s, 3H), 2.21 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.04, 170.01, 146.52 (m), 145.94, 143.99 (m), 142.50 (m), 141.64, 139.94, 139.07, 136.53 (m), 136.11, 129.19, 127.51, 126.84, 124.63, 122.41, 119.89, 113.36, 110.15 (m), 109.85, 37.99, 23.87, 11.27. HRMS (ESI) calcd for C24H15F5N3O2 [M+H]+ 472.1078, found 472.1076.
6-(N-丁二酰亚胺基)-1,9-二甲基-β-咔啉(5z): 白色固体, 产率64.5%. m.p. 88.7~89.6 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.32 (d, J=5.2 Hz, 1H), 8.02 (s, 1H), 7.77 (d, J=5.2 Hz, 1H), 7.53 (d, J=8.8 Hz, 1H), 7.47 (d, J=8.4 Hz, 1H), 4.16 (s, 3H), 3.11 (s, 3H), 2.96 (s, 4H); 13C NMR (100 MHz, CDCl3) δ: 176.72, 141.98, 141.60, 138.12, 136.31, 128.62, 126.53, 123.83, 121.42, 120.08, 113.18, 110.13, 32.48, 28.49, 23.43. HRMS (ESI) calcd for C17H16N3O2 [M+H]+ 294.1237, found 294.1238.
6-(N-丁二酰亚胺基)-1-甲基-9-正丁基-β-咔啉(5aa): 白色固体, 产率58.9%. m.p. 184.6~185.7 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.34 (d, J=5.2 Hz, 1H), 8.03 (d, J=1.6 Hz, 1H), 7.80 (d, J=5.2 Hz, 1H), 7.54 (d, J=8.8 Hz, 1H), 7.45 (dd, J=8.8, 1.8 Hz, 1H), 4.63~4.47 (m, 2H), 3.06 (s, 3H), 2.97 (s, 4H), 1.87~1.79 (m, 2H), 1.51~1.41 (m, 2H), 0.99 (t, J=7.4 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 176.77, 141.60, 141.02, 138.17, 135.63, 128.79, 126.38, 123.73, 121.65, 120.09, 113.19, 110.43, 44.97, 32.90, 28.48, 23.45, 20.20, 13.86. HRMS (ESI) calcd for C20H22N3O2 [M+H]+ 336.1706, found 336.1707.
6-(N-丁二酰亚胺基)-1-甲基-9-苄基-β-咔啉(5ab): 白色固体, 产率54.0%. m.p. 196.2~197.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.36 (d, J=5.2 Hz, 1H), 8.07 (s, 1H), 7.83 (d, J=5.2 Hz, 1H), 7.43 (d, J=8.6 Hz, 1H), 7.39 (d, J=8.6 Hz, 1H), 7.28~7.26 (m, 3H), 6.99 (d, J=7.2Hz, 2H), 5.78 (s, 2H), 2.95 (s, 4H), 2.88 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 176.76, 142.04, 141.36, 138.76, 137.49, 136.05, 128.95, 127.71, 126.69, 125.42, 124.20, 121.74, 120.10, 113.22, 110.58, 48.39, 28.49, 23.12. HRMS (ESI) calcd for C23H20N3O2 [M+H]+ 370.1550, found 370.1551.
6-(N-丁二酰亚胺基)-1-甲基-9-(4-氟苄基)-β-咔啉(5ac): 白色固体, 产率69.2%. m.p. 277.0~278.4 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.38 (d, J=5.2 Hz, 1H), 8.09 (s, 1H), 7.85 (d, J=5.2 Hz, 1H), 7.43 (s, 2H), 7.06~6.89 (m, 4H), 5.76 (s, 2H), 2.97 (s, 4H), 2.89 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 176.70, 162.25 (d, J=245 Hz), 141.67, 141.36, 138.33, 135.84, 133.03 (d, J=3.1 Hz), 129.17, 127.10 (d, J=8.1 Hz), 127.02, 124.46, 121.68, 120.25, 116.11 (d, J=21.8 Hz), 113.41, 110.50, 47.80, 28.48, 22.58. HRMS (ESI) calcd for C23H19FN3O2 [M+H]+ 388.1455, found 388.1455.
6-(N-丁二酰亚胺基)-1-甲基-9-(2,3,4,5,6-五氟苄基)-β-咔啉(5ad): 白色固体, 产率71.8%. m.p. 257.6~259.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.39 (d, J=5.6 Hz, 1H), 8.03 (s, 1H), 7.80 (d, J=4.4 Hz, 1H), 7.47~7.36 (m, 2H), 5.94 (s, 2H), 3.09 (s, 3H), 3.00~2.92 (m, 4H); 13C NMR (100 MHz, CDCl3) δ: 176.60, 146.48 (m), 144.05(m), 142.58 (m), 141.73, 140.32, 139.26, 136.54 (m), 136.10, 129.05, 126.80, 124.80, 122.49, 120.22, 113.33, 110.22 (m), 109.96, 37.96, 28.47, 23.96. HRMS (ESI) calcd for C23H15F5N3O2 [M+H]+ 460.1078, found 460.1076.