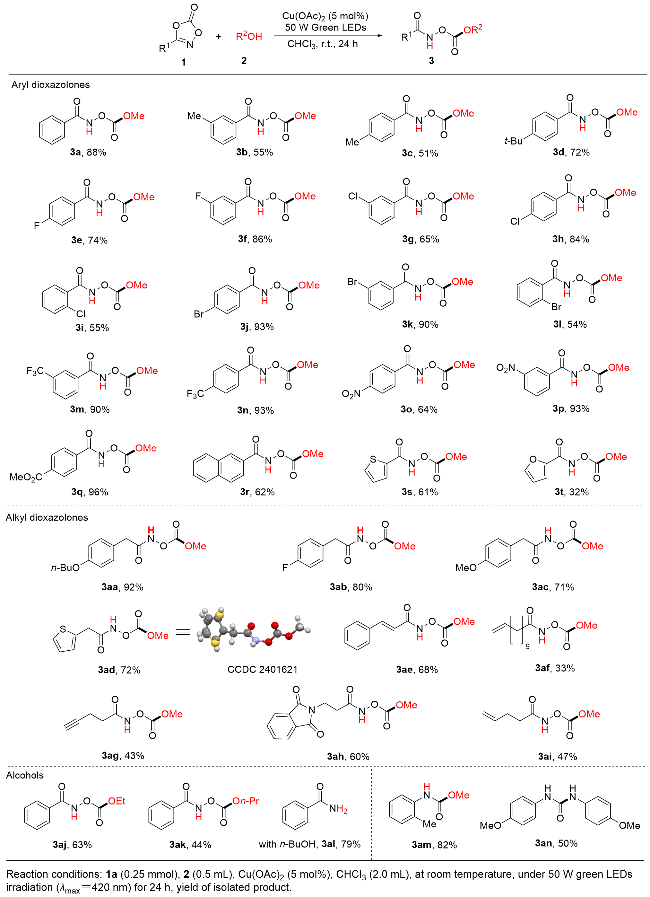
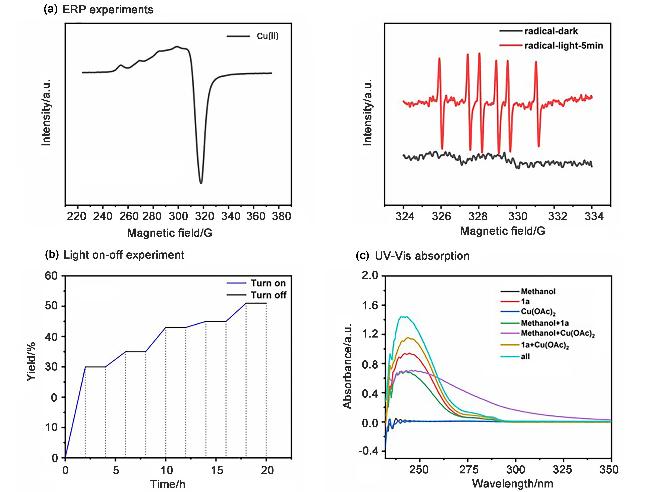
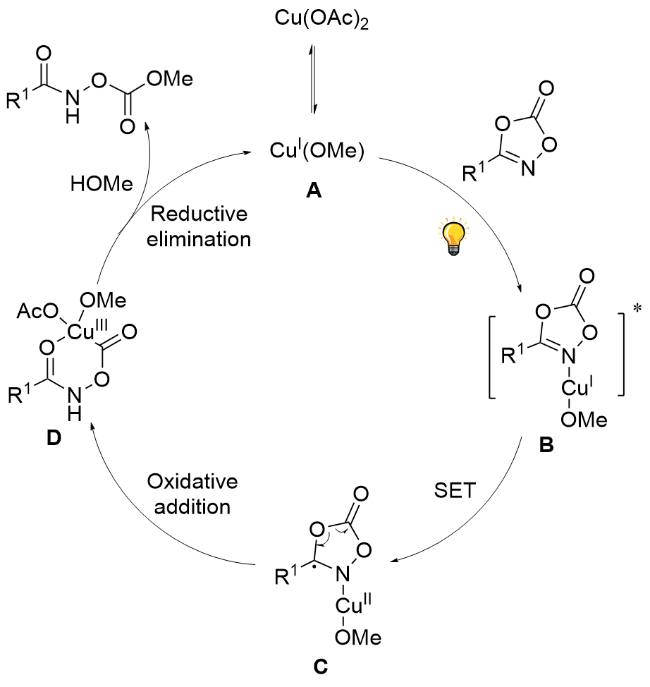
Compounds 1a~1s, 1aa~1ah (0.25 mmol) and Cu(OAc)2 (2.3 mg, 0.013 mmol, 0.05 equiv.) were dissolved in CHCl3 (2 mL), and 2 (0.5 mL) was added. The mixture was stirred at room temperature under irradiation with a 50 W green LEDs for 24 h. The solution was concentrated and purified by column chromatography.
N-((Methoxycarbonyl)oxy)benzamide (3a): Yellow solid, yield 88% (42.1 mg), Rf=0.34 (CH2Cl2), m.p. 36~37 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.77 (s, 1H), 7.79~7.75 (m, 2H), 7.47 (t, J=7.3 Hz, 1H), 7.34 (t, J=7.7 Hz, 2H), 3.81 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.9, 155.2, 132.8, 130.3, 128.8, 127.7, 56.5. HRMS (ESI) calcd for C9H9NO4 [M+H]+ 195.0609, found 195.0610.
N-((Methoxycarbonyl)oxy)-3-methylbenzamide (3b): Yellow oil, yield 55% (25.9 mg), Rf=0.36 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.69 (s, 1H), 7.61 (s, 1H), 7.58 (d, J=7.5 Hz, 1H), 7.34 (d, J=7.7 Hz, 1H), 7.29 (t, J=7.5 Hz, 1H), 3.91 (s, 3H), 2.35 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.2, 155.5, 138.9, 133.7, 130.3, 128.8, 128.3, 124.6, 56.6, 21.4. HRMS (ESI) calcd for C10H11NO4 [M+H]+ 210.0766, found 210.0767.
N-((Methoxycarbonyl)oxy)-4-methylbenzamide (3c): Yellow oil, yield 51% (24.1 mg), Rf=0.35 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.64 (s, 1H), 7.69 (d, J=8.2 Hz, 2H), 7.21 (d, J=7.9 Hz, 2H), 3.91 (s, 3H), 2.38 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.1, 155.6, 143.7, 129.5, 127.6, 127.5, 56.6, 21.7. HRMS (ESI) calcd for C10H11NO4 [M+H]+ 210.0768, found 210.0769.
4-(tert-Butyl)-N-((methoxycarbonyl)oxy)benzamide (3d): Yellow solid, yield 72% (33.0 mg), Rf=0.41 (CH2Cl2), m.p. 91~92 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.48 (s, 1H), 7.75 (d, J=8.5 Hz, 2H), 7.45 (d, J=8.7 Hz, 2H), 3.93 (s, 3H), 1.32 (s, 9H); 13C NMR (100 MHz, CDCl3) δ: 167.1, 156.7, 155.6, 127.5, 125.9, 56.6, 35.2, 31.2. HRMS (ESI) calcd for C13H17NO4 [M+H]+ 252.1227, found 252.1252.
4-Fluoro-N-((methoxycarbonyl)oxy)benzamide (3e): Ye- llow solid, yield 74% (34.8 mg), Rf=0.32 (CH2Cl2), m.p. 45~46 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.02 (s, 1H), 7.81 (dd, J=8.8, 5.3 Hz, 2H), 7.08 (t, J=8.7 Hz, 2H), 3.91 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.0, 165.6 (d, J=235 Hz), 155.4, 130.2 (d, J=10 Hz), 126.5 (d, J=3 Hz), 116.1 (d, J=22 Hz), 56.7; 19F NMR (376 MHz, CDCl3) δ: -115.2 (s, 1F). HRMS (ESI) calcd for C9H8FNO4 [M+ H]+ 214.0515, found 214.0519.
3-Fluoro-N-((methoxycarbonyl)oxy)benzamide (3f): Ye- llow oil, yield 86% (40.1 mg), Rf=0.34 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 10.06 (s, 1H), 7.57 (d, J=7.6 Hz, 1H), 7.50 (d, J=9.3 Hz, 1H), 7.39 (dd, J=13.5, 7.9 Hz, 1H), 7.23 (t, J=8.3 Hz, 1H), 3.91 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 165.6, 162.7 (d, J=247 Hz), 155.2, 132.3, 130.7 (d, J=8 Hz), 123.2 (d, J=4 Hz), 120.0 (d, J=21 Hz), 115 (d, J=23 Hz), 56.7; 19F NMR (376 MHz, CDCl3) δ: -111.0 (s, 1F). HRMS (ESI) calcd for C9H8FNO4 [M+H]+ 214.0512, found 214.0516.
3-Chloro-N-((methoxycarbonyl)oxy)benzamide (3g): Yellow oil, yield 55% (25.6 mg), Rf=0.35 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.89 (s, 1H), 7.78 (s, 1H), 7.66 (d, J=7.8 Hz, 1H), 7.51 (d, J=8.0 Hz, 1H), 7.36 (t, J=7.9 Hz, 1H), 3.93 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 165.6, 155.3, 135.1, 133.1, 132.0, 130.2, 127.9, 125.7, 56.7. HRMS (ESI) calcd for C9H8ClNO4 [M+H]+ 230.0220, found 230.0221.
4-Chloro-N-((methoxycarbonyl)oxy)benzamide (3h): Yellow solid, yield 84% (39.1 mg), Rf=0.34 (CH2Cl2), m.p. 97~98 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.36 (s, 1H), 7.70 (d, J=8.6 Hz, 2H), 7.35 (d, J=8.5 Hz, 2H), 3.88 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.0, 155.3, 139.3, 129.1, 129.1, 128.6, 56.7. HRMS (ESI) calcd for C9H8Cl- NO4 [M+H]+ 230.0221, found 230.0222.
2-Chloro-N-((methoxycarbonyl)oxy)benzamide (3i): Brown oil, yield 55% (25.6 mg), Rf=0.35 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.54 (s, 1H), 7.70 (d, J=7.8 Hz, 1H), 7.44 (d, J=1.3 Hz, 1H), 7.42 (d, J=1.1 Hz, 1H), 7.35 (dd, J=7.7, 5.0 Hz, 1H), 3.95 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 164.7, 155.0, 132.7, 131.5, 130.9, 130.8, 130.6, 127.3, 56.7. HRMS (ESI) calcd for C9H8ClNO4 [M+H]+ 230.0221, found 230.0233.
4-Bromo-N-((methoxycarbonyl)oxy)benzamide (3j): Ye- llow solid, yield 93% (42.1 mg), Rf=0.40 (CH2Cl2), m.p. 90~91 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.75 (s, 1H), 7.65 (d, J=8.6 Hz, 2H), 7.57 (d, J=8.6 Hz, 2H), 3.93 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.1, 155.4, 132.2, 129.2, 129.1, 128.0, 56.7. HRMS (ESI) calcd for C9H8Br- NO4 [M+H]+ 273.9710, found 273.9714.
3-Bromo-N-((methoxycarbonyl)oxy)benzamide (3k): Yellow solid, yield 90% (40.8 mg), Rf=0.42 (CH2Cl2), m.p. 55~56 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.91 (s, 1H), 7.70 (d, J=7.8 Hz, 1H), 7.63 (d, J=8.1 Hz, 1H), 7.26 (t, J=8.0 Hz, 1H), 3.89 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 165.2, 155.1, 135.8, 132.1, 130.8, 130.4, 126.2, 122.9, 56.7. HRMS (ESI) calcd for C9H8BrNO4 [M+H]+ 273.9711, found 273.9715.
2-Bromo-N-((methoxycarbonyl)oxy)benzamide (3l): Ye- llow oil, yield 60% (27.2 mg), Rf=0.41 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.33 (s, 1H), 7.63 (d, J=1.2 Hz, 1H), 7.61 (d, J=1.5 Hz, 1H), 7.41~7.32 (m, 2H), 3.95 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 165.5, 155.5, 133.8, 133.3, 132.7, 130.5, 127.7, 120.1, 56.7. HRMS (ESI) calcd for C9H8BrNO4 [M+H]+ 273.9714, found 273.9717.
N-((Methoxycarbonyl)oxy)-3-(trifluoromethyl)benzami-de (3m): Yellow solid, yield 90% (38.3 mg), Rf=0.30 (CH2Cl2), m.p. 58~59 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.16 (s, 1H), 8.06 (s, 1H), 7.98 (d, J=7.8 Hz, 1H), 7.78 (d, J=7.8 Hz, 1H), 7.56 (t, J=7.8 Hz, 1H), 3.92 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 164.3, 155.2, 131.3 (t, J=33 Hz), 130.9, 129.6, 129.5 (d, J=3 Hz), 124.9, 124.7, 122.2 (d, J=3 Hz), 56.9; 19F NMR (376 MHz, CDCl3) δ: -62.9 (s, 3F). HRMS (ESI) calcd for C10H8F3NO4 [M+H]+ 264.0481, found 264.0483.
N-((Methoxycarbonyl)oxy)-4-(trifluoromethyl)benzami-de (3n): White solid, yield 93% (39.6 mg), Rf=0.35 (CH2Cl2), m.p. 110~111 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.01 (d, J=8.2 Hz, 2H), 7.92 (d, J=8.2 Hz, 2H), 3.89 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 164.0, 155.0, 134.9, 132.6 (d, J=32 Hz), 128.8, 126.3 (d, J=4 Hz), 124.2 (d, J=271 Hz), 56.9; 19F NMR (376 MHz, CDCl3) δ: -63.1 (s, 3F). HRMS (ESI) calcd for C10H8F3- NO4 [M+H]+ 264.0483, found 264.0486.
N-((Methoxycarbonyl)oxy)-4-nitrobenzamide (3o): Ye- llow solid, yield 64% (29.5 mg), Rf=0.29 (CH2Cl2), m.p. 89~90 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.38 (d, J=9.1 Hz, 2H), 8.05 (d, J=8.9 Hz, 2H), 3.90 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 163.6, 155.0, 150.3, 136.7, 129.5, 124.5, 57.0. HRMS (ESI) calcd for C9H8N2O6 [M+H]+ 241.0461, found 241.0467.
N-((Methoxycarbonyl)oxy)-3-nitrobenzamide (3p): Whi- te solid, yield 93% (42.9 mg), Rf=0.35 (CH2Cl2), m.p. 142~143 ℃; 1H NMR (400 MHz, Acetone-d6) δ: 11.99 (s, 1H), 8.69 (s, 1H), 8.50 (d, J=8.2 Hz, 1H), 8.32 (d, J=7.8 Hz, 1H), 7.88 (t, J=8.0 Hz, 1H), 3.94 (s, 3H); 13C NMR (100 MHz, Acetone-d6) δ: 163.3, 154.9, 148.5, 133.6, 132.7, 130.6, 127.0, 122.2, 56.0. HRMS (ESI) calcd for C9H8N2O6 [M+H]+ 241.0464, found 241.0468.
Methyl 4-((methoxycarbonyl)oxy)carbamoyl)benzoate (3q): Yellow solid, yield 96% (43.9 mg), Rf=0.30 (CH2Cl2), m.p. 118~119 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.89 (s, 1H), 8.06 (d, J=8.8 Hz, 2H), 7.85 (d, J=8.3 Hz, 2H), 3.93 (s, 6H); 13C NMR (100 MHz, CDCl3) δ: 166.3, 166.0, 155.3, 134.3, 133.9, 130.1, 127.8, 56.8, 52.7. HRMS (ESI) calcd for C11H11NO6 [M+H]+ 254.0665, found 254.0666.
N-((Methoxycarbonyl)oxy)-2-naphthamide (3r): Yellow solid, yield 62% (28.5 mg), Rf=0.35 (CH2Cl2), m.p. 84~85 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.04 (s, 1H), 8.31 (s, 1H), 7.82 (d, J=6.9 Hz, 2H), 7.79 (s, 2H), 7.52 (dd, J=16.9, 8.2 Hz, 2H), 3.91 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.2, 155.6, 135.4, 132.4, 129.2, 128.8, 128.8, 128.4, 127.9, 127.5, 127.0, 123.5, 56.6. HRMS (ESI) calcd for C13H11NO4 [M+H]+ 246.0728, found 246.0734.
N-((Methoxycarbonyl)oxy)thiophene-2-carboxamide(3s): Brown oil, yield 61% (29.0 mg), Rf=0.55 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.95 (s, 1H), 7.69 (d, J=2.4 Hz, 1H), 7.56 (d, J=3.7 Hz, 1H), 7.09~7.05 (m, 1H), 3.90 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 162.0, 155.5, 133.4, 132.2, 130.7, 128.1, 56.7. HRMS (ESI) calcd for C7H7NO4S [M+H]+ 201.0128, found 201.0122.
N-((Methoxycarbonyl)oxy)furan-2-carboxamide (3t): Brown oil, yield 32% (15.5 mg), Rf=0.45 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.61 (s, 1H), 7.49 (dd, J=1.7, 0.8 Hz, 1H), 7.25 (dd, J=3.6, 0.8 Hz, 1H), 6.53 (dd, J=3.6, 1.7 Hz, 1H), 3.93 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 157.2, 155.3, 145.4, 144.7, 117.3, 112.4, 56.7. HRMS (ESI) calcd for C7H7NO5 [M+H]+ 186.0402, found 186.0403.
2-(4-Butoxyphenyl)-N-((methoxycarbonyl)oxyacetami-de (3aa): Yellow solid, yield 92% (41.8 mg), Rf=0.55 (CH2Cl2), m.p. 86~87 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.52 (s, 1H), 7.20 (d, J=8.6 Hz, 2H), 6.88 (d, J=8.7 Hz, 2H), 3.94 (t, J=6.6 Hz, 2H), 3.89 (s, 3H), 3.60 (s, 2H), 1.79~1.72 (m, 2H), 1.48 (dd, J=15.1, 7.4 Hz, 2H), 0.97 (t, J=7.4 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 158.9, 155.1, 130.6, 124.3, 115.2, 67.8, 56.6, 40.0, 31.4, 19.3, 13.9. HRMS (ESI) calcd for C14H19NO5 [M+H]+ 287.1754, found 287.1750.
2-(4-Fluorophenyl)-N-((methoxycarbonyl)oxy)acetami-de (3ab): White solid, yield 80% (37.3 mg), Rf=0.35 (CH2Cl2), m.p. 42~43 ℃; 1H NMR (400 MHz, Acetone-d6) δ: 11.10 (s, 1H), 7.36 (dd, J=8.6, 5.5 Hz, 2H), 7.08 (t, J=8.9 Hz, 2H), 3.84 (s, 3H), 3.57 (s, 2H); 13C NMR (100MHz, Acetone-d6) δ: 168.4, 162.0 (d, J=242 Hz), 155.1, 131.2 (d, J=8 Hz), 115.1 (d, J=21 Hz), 55.8, 38.1. 19F NMR (376 MHz, CDCl3) δ: -117.5 (s, 3F). HRMS (ESI) calcd for C14H19NO5 [M+H]+ 227.1904, found 227.1902.
N-((Methoxycarbonyl)oxy)-2-(4-methoxyphenyl)aceta-mide (3ac): Brown solid, yield 72% (33.8 mg), Rf=0.43 (CH2Cl2), m.p. 95~96 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.72 (s, 1H), 7.22 (d, J=5.2 Hz, 1H), 6.97 (m, 1H), 6.95 (d, J=3.3 Hz, 1H), 3.86 (s, 3H), 3.80 (s, 2H); 13C NMR (100 MHz, CDCl3) δ: 168.2, 154.9, 134.0, 127.7, 127.4, 125.8, 56.6, 34.2. HRMS (ESI) calcd for C8H9NO4S [M+H]+ 216.0311, found 216.0305.
N-((Methoxycarbonyl)oxy)-2-(thiophen-2-yl)acetamide(3ad): White solid, yield 71% (41.6 mg), Rf=0.50 (CH2Cl2/ EtOAc, V∶V=10∶1), m.p. 59~60 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.19 (s, 1H), 7.20 (d, J=8.7 Hz, 2H), 6.86 (d, J=8.7 Hz, 2H), 3.86 (s, 3H), 3.78 (s, 3H), 3.54 (s, 2H); 13C NMR (100 MHz, CDCl3) δ: 169.8, 159.1, 155.1, 130.6, 124.9, 114.5, 56.5, 55.4, 39.5. HRMS (ESI) calcd for C11H13NO5 [M+H]+ 239.2311, found 239.2310.
N-((Methoxycarbonyl)oxy)cinnamamide (3ae): Yellow solid, yield 68% (31.8 mg), Rf=0.53 (CH2Cl2), m.p. 120~121 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.23 (s, 1H), 7.75 (d, J=15.6 Hz, 1H), 7.49~7.46 (m, 2H), 7.32 (d, J=6.7 Hz, 3H), 6.56 (d, J=15.5 Hz, 1H), 3.87 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 165.7, 155.3, 144.2, 134.3, 130.5, 129.0, 128.3, 115.3, 56.6. HRMS (ESI) calcd for C11H11N- O4 [M+H]+ 222.0766, found 222.0769.
N-((Methoxycarbonyl)oxy)dodec-11-enamide (3af): Ye- llow oil, yield 33% (15.1 mg), Rf=0.52 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.08 (s, 1H), 5.85~5.74 (m, 1H), 5.00~4.90 (m, 2H), 3.90 (s, 3H), 2.23 (t, J=7.4 Hz, 2H), 2.05~1.99 (m, 2H), 1.70~1.62 (m, 2H), 1.37~1.24 (m, 10H); 13C NMR (100 MHz, CDCl3) δ: 172.0, 155.3, 139.3, 114.3, 56.5, 33.9, 32.8, 29.3, 29.3, 29.1, 29.1, 29.0, 25.1. HRMS (ESI) calcd for C13H23NO4 [M+H]+ 258.1741, found 258.1738.
N-((Methoxycarbonyl)oxy)pent-4-ynamide (3ag): Yellow solid, yield 21% (10.3 mg), Rf=0.53 (CH2Cl2), m.p. 83~84 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.38 (s, 1H), 3.92 (s, 3H), 2.56 (t, J=3.2 Hz, 2H), 2.50 (t, J=6.8 Hz, 2H), 2.04 (s, 1H); 13C NMR (100 MHz, CDCl3) δ: 169.8, 155.1, 82.2, 70.0, 56.6, 32.0, 14.4. HRMS (ESI) calcd for C7H9NO4 [M+H]+ 172.0698, found 172.0601.
3-(1,3-Dioxoisoindolin-2-yl)-N-((methoxycarbonyl)-oxy)propanamide (3ah): Yellow solid, yield 60% (27.0 mg), Rf=0.50 (EtOAc/CH2Cl2, V∶V=1∶4), m.p. 130~131 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.63 (s, 1H), 7.82 (dd, J=5.7, 3.1 Hz, 2H), 7.70 (dd, J=5.4, 3.0 Hz, 2H), 4.04 (t, J=7.1 Hz, 2H), 3.86 (s, 3H), 2.74 (t, J=7.2 Hz, 2H); 13C NMR (100 MHz, CDCl3) δ: 168.3, 155.0, 134.3, 132.0, 123.5, 56.6, 33.7, 317, 29.8. HRMS (ESI) calcd for C13H12- N2O6 [M+H]+ 293.0771, found 293.0774.
N-((Methoxycarbonyl)oxy)pent-4-enamide (3ai): Yellow oil, yield 42% (20.6 mg), Rf=0.42 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.44 (s, 1H), 5.87~5.77 (m, 1H), 5.04 (t, J=9.0 Hz, 2H), 3.90 (s, 3H), 2.44~2.39 (m, 2H), 2.34 (t, J=7.2 Hz, 2H); 13C NMR (100 MHz, CDCl3) δ: 171.1, 155.2, 136.3, 116.2, 56.5, 32.1, 28.9. HRMS (ESI) calcd for C7H11NO4 [M+H]+ 173.0702, found 173.0703.
N-((Ethoxycarbonyl)oxy)benzamide (3aj): Brown oil, yield 63% (32.3 mg), Rf=0.50 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.50 (s, 1H), 7.81~7.79 (m, 2H), 7.54 (t, J=7.3 Hz, 1H), 7.45~7.41 (m, 2H), 4.33 (q, J=7.2 Hz, 2H), 1.36 (t, J=7.1 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 166.9, 154.8, 132.9, 130.4, 128.9, 127.6, 66.5, 14.2. HRMS (ESI) calcd for C10H11NO4 [M+H]+ 210.0795, found 210.0701.
N-((Propoxycarbonyl)oxy)benzamide (3ak): Yellow oil, yield 44% (24.1 mg), Rf=0.35 (CH2Cl2); 1H NMR (400 MHz, CDCl3) δ: 9.49 (s, 1H), 7.81~7.78 (m, 2H), 7.55 (t, J=7.5 Hz, 1H), 7.43 (t, J=7.9 Hz, 2H), 4.24 (t, J=6.7 Hz, 2H), 1.80~1.71 (m, 2H), 0.98 (t, J=7.4 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.0, 155.0, 133.0, 130.5, 128.9, 127.6, 72.0, 22.0, 10.1. HRMS (ESI) calcd for C11H13NO4 [M+H]+ 224.0923, found 224.0924.
Benzamide (3al): White solid, yield 79% (23.6 mg), Rf=0.50 (EtOAc/CH2Cl2, V∶V=1∶1), m.p. 129~130 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.84~7.80 (m, 2H), 7.52 (t, J=7.4 Hz, 1H), 7.44 (t, J=7.4 Hz, 2H), 6.32 (s, 2H); 13C NMR (100 MHz, CDCl3) δ: 169.9, 133.5, 132.1, 128.7, 127.5. HRMS (ESI) calcd for C7H7NO [M+H]+ 121.0598, found 121.1200.
Methyl o-tolylcarbamate (3am): Yellow solid, yield 82% (42.3 mg), Rf=0.35 (CH2Cl2), m.p. 45~46 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.76 (s, 1H), 7.23~7.16 (m, 2H), 7.04 (t, J=6.3 Hz, 1H), 6.49 (s, 1H), 3.78 (s, 3H), 2.25 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 154.6, 135.9, 130.5, 127.0, 124.4, 52.5, 17.7. HRMS (ESI) calcd for C10H11NO4 [M+H]+ 210.0767, found 210.0768.
1,3-Bis(4-methoxyphenyl)urea (3an): Yellow solid, yield 50% (24.1 mg), Rf=0.35 (CH2Cl2), m.p. 64~65 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.38 (s, 2H), 7.34 (d, J=9.2 Hz, 4H), 6.86 (d, J=9.2 Hz, 4H), 3.71 (s, 6H); 13C NMR (100 MHz, DMSO-d6) δ: 154.8, 153.5, 133.5, 120.4, 114.5, 55.7. HRMS (ESI) calcd for C15H16N2O3 [M+H]+ 272.1298, found 272.3001.
Supporting Information Crystallographic data,
1H NMR and
13C NMR spectra of compounds
3a~
3t and
3aa~
3an. The Supporting Information is available free of charge via the Internet at
http://sioc-journal.cn.