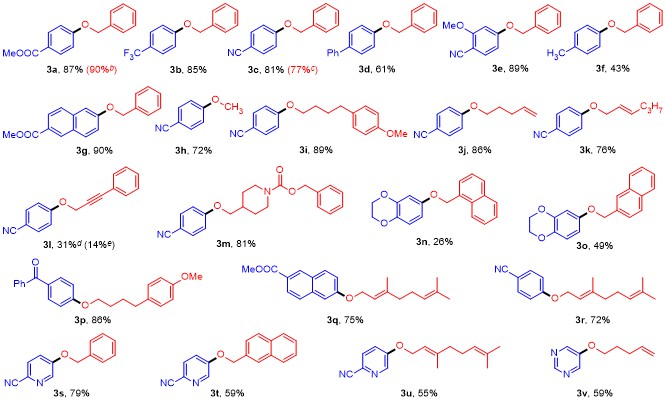
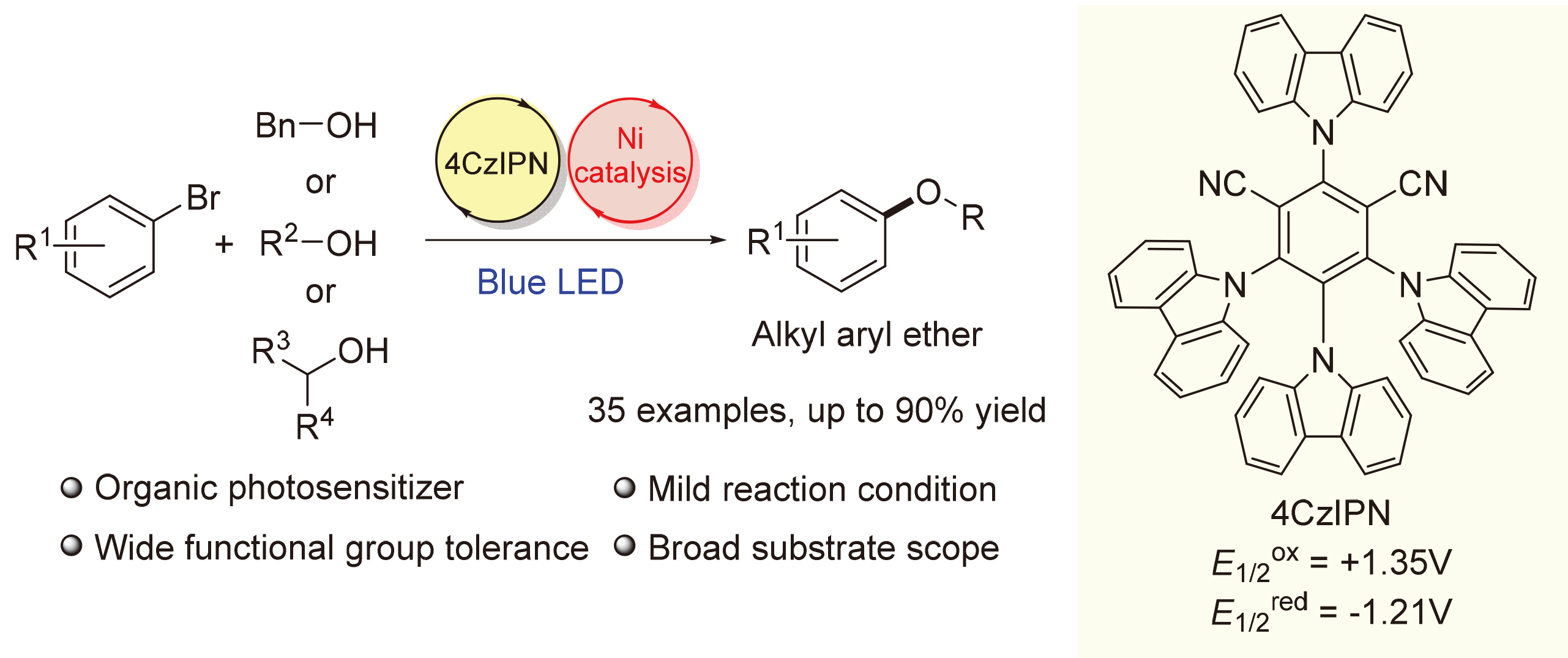
充满N
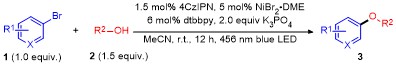
2的手套箱中, 向干燥的4 mL反应瓶中依次加入4CzIPN (3.6 mg, 1.5 mol%, 0.0045 mmol), NiBr
2• DME (4.6 mg, 5.0 mol%, 0.015 mmol), dtbbpy (4.8 mg, 6.0 mol%, 0.018 mmol), K
3PO
4 (127.4 mg, 2.0 equiv., 0.6 mmol), MeCN (3.0 mL), 4-溴苯甲酸甲酯(64.5 mg, 1.0 equiv., 0.3 mmol)以及苄醇(48.7 mg, 1.5 equiv., 0.45 mmol). 盖好瓶盖后, 移出手套箱置于2×40 W blue LED, 功率都设定为75% (0.08~0.09 W/cm
2), 光照条件下反应. 在室温下反应12 h后, TLC检测反应. 反应结束后, 通过铺有硅胶的漏斗进行抽滤, 并用乙酸乙酯冲洗, 所得滤液经旋转蒸发除去溶剂. 粗产品用硅胶柱层析[洗脱剂为
V(石油醚)∶
V(乙酸乙酯)=20∶1], 得到4-(苄氧基)苯甲酸甲酯(
3a) 63.3 mg, 产率87%. 白色固体, m.p. 96~97 ℃ (m.p.
[34a] 95~96 ℃);
1H NMR (400 MHz, CDCl
3)
δ: 7.99 (d,
J=9.2 Hz, 2H), 7.42~7.31 (m, 5H), 6.98 (d,
J=9.2 Hz, 2H), 5.09 (s, 2H), 3.86 (s, 3H);
13C NMR (100 MHz, CDCl
3)
δ: 166.7, 162.4, 136.2, 131.5, 128.6, 128.1, 127.4, 122.7, 114.4, 70.0, 51.8. 核磁数据与文献报道一致
[25].
当使用底物为4-氯苯甲酸甲酯代替4-溴苯甲酸甲酯时, 同样操作与后处理, 得到4-(苄氧基)苯甲酸甲酯(3a) 65.4 mg, 白色固体, 产率90%.
1-(苄氧基)-4-(三氟基甲基)苯(
3b): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=50∶1, 64.3 mg, 收率85%. 白色固体, m.p. 76~77 ℃ (m.p.
[34b] 73~75 ℃);
1H NMR (400 MHz, CDCl
3)
δ: 7.53 (d,
J=8.8 Hz, 2H), 7.43~7.33 (m, 5H), 7.02 (d,
J=8.4 Hz, 2H), 5.08 (s, 2H);
13C NMR (100 MHz, CDCl
3)
δ: 161.2, 136.2, 128.7, 128.3, 127.4, 126.9 (q,
3JC-F=3.9 Hz), 124.4 (q,
1JC-F=269.2 Hz), 123.1 (q,
2JC-F=32.5 Hz), 114.8, 70.1;
19F NMR (376 MHz, CDCl
3)
δ: -61.45 (s, 3F). 核磁数据与文献报道一致
[34c].
4-(苄氧基)苯腈(
3c): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=20∶1, 50.9 mg, 收率81%. 白色固体, m.p. 91~92 ℃ (m.p.
[34d] 87.6~89.2 ℃);
1H NMR (400 MHz, CDCl
3)
δ: 7.57 (d,
J=8.8 Hz, 2H), 7.42~7.34 (m, 5H), 7.01 (d,
J=8.8 Hz, 2H), 5.10 (s, 2H);
13C NMR (100 MHz, CDCl
3)
δ: 161.9, 135.6, 133.9, 128.7, 128.3, 127.4, 119.1, 115.5, 104.1, 70.2. 核磁数据与文献报道一致
[34e].
当使用底物为4-氯苯腈代替4-溴苯腈时, 同样操作与后处理, 得到4-(苄氧基)苯腈(3c) 48.3 mg, 白色固体, 产率77%.
4-(苄氧基)-1,1'-联苯(
3d): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=20∶1, 47.3 mg, 收率61%. 白色固体, m.p. 131~133 ℃ (m.p.
[34f] 131.5~134.8 ℃);
1H NMR (400 MHz, CDCl
3)
δ: 7.56~7.52 (m, 4H), 7.46 (d,
J=7.2 Hz, 2H), 7.43~7.38 (m, 4H), 7.35~7.28 (m, 2H), 7.05 (d,
J=8.8 Hz, 2H), 5.11 (s, 2H);
13C NMR (100 MHz, CDCl
3)
δ: 158.3, 140.8, 137.0, 134.0, 128.7, 128.6, 128.2, 128.0, 127.5, 126.73, 126.67, 115.1, 70.1. 核磁数据与文献报道一致
[34f].
4-(苄氧基)-2-甲氧基苯腈(3e): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=10∶1, 黄色固体, 64.2 mg, 收率89%, m.p. 82~83 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.46~7.35 (m, 6H), 6.59~6.54 (m, 2H), 5.09 (s, 2H), 3.86 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 163.6, 162.7, 135.6, 134.8, 128.7, 128.4, 127.5, 116.8, 106.4, 99.2, 94.0, 70.4, 55.9; IR (neat) ν: 2943, 2212, 1604, 1578, 1505, 1284, 1274, 1203, 1174, 1114, 1001, 820, 771, 728, 698 cm-1; HRMS (EI) calcd for C15H13NO2 239.0941, found 239.0937.
1-(苄氧基)-4-甲基苯(
3f): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=40∶1, 25.7 mg, 收率43%. 淡黄色固体, m.p. 42~44 ℃ (m.p.
[34g] 44~46 ℃);
1H NMR (400 MHz, CDCl
3)
δ: 7.43~7.31 (m, 5H), 7.09~7.07 (m, 2H), 6.88~6.86 (m, 2H), 5.03 (s, 2H), 2.28 (s, 3H);
13C NMR (100 MHz, CDCl
3)
δ: 156.6, 137.2, 130.1, 129.9, 128.5, 127.8, 127.4, 114.7, 70.0, 20.5. 核磁数据与文献报道一致
[34h].
6-(苄氧基)-2-萘酸甲酯(3g): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=20∶1, 白色固体, 79.0 mg, 收率90%, m.p. 150~151 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.51 (s, 1H), 8.01 (d, J=8.4 Hz, 1H), 7.82 (d, J=9.2 Hz, 1H), 7.71 (d, J=8.4 Hz, 1H), 7.48~7.32 (m, 5H), 7.27~7.20 (m, 2H), 5.15 (s, 2H), 3.94 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.3, 158.6, 137.0, 136.4, 130.9, 130.8, 128.6, 128.1, 127.9, 127.5, 126.8, 125.9, 125.2, 119.9, 106.9, 70.0, 52.0; IR (neat) ν: 2946, 1713, 1622, 1479, 1385, 1291, 1217, 1201, 1095, 1011, 865, 823, 755, 734, 695 cm-1; HRMS (EI) calcd for C19H16O3 292.1094, found 292.1095.
4-甲氧基苯腈(
3h): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=10∶1, 28.7 mg, 收率72%. 白色固体, m.p. 57~59 ℃ (m.p.
[34g] 60~62 ℃);
1H NMR (400 MHz, CDCl
3)
δ: 7.59 (d,
J=8.8 Hz, 2H), 6.95 (d,
J=9.2 Hz, 2H), 3.86 (s, 3H);
13C NMR (100 MHz, CDCl
3)
δ: 162.8, 133.9, 119.2, 114.7, 103.8, 55.5. 核磁数据与文献报道一致
[34i].
4-(4-(4-甲氧基苯基)丁氧基)苯腈(3i): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=10∶1, 淡黄色液体, 75.2 mg, 收率89%. 1H NMR (400 MHz, CDCl3) δ: 7.55 (d, J=8.8 Hz, 2H), 7.10 (d, J=8.4 Hz, 2H), 6.90 (d, J=9.2 Hz, 2H), 6.83 (d, J=8.4 Hz, 2H), 3.98 (t, J=6.4 Hz, 2H), 3.78 (s, 3H), 2.62 (t, J=7.2 Hz, 2H), 1.84~1.74 (m, 4H); 13C NMR (100 MHz, CDCl3) δ: 162.3, 157.7, 133.9, 133.8, 129.2, 119.2, 115.1, 113.7, 103.5, 68.1, 55.1, 34.5, 28.4, 27.8; IR (neat) ν: 2932, 2220, 1604, 1508, 1465, 1300, 1261, 1243, 1168, 1113, 1034, 947, 834, 810, 696 cm-1; HRMS (EI) calcd for C18H19NO2 281.1410, found 281.1420.
4-(戊-4-烯-1-基氧基)苯腈(
3j): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=10∶1, 黄色液体, 48.4 mg, 收率86%.
1H NMR (400 MHz, CDCl
3)
δ: 7.57 (d,
J=8.8 Hz, 2H), 6.94 (d,
J=8.8 Hz, 2H), 5.87~5.79 (m, 1H), 5.09~5.01 (m, 2H), 4.01 (t,
J=6.4 Hz, 2H), 2.24 (q,
J=6.8 Hz, 2H), 1.94~1.87 (m, 2H);
13C NMR (100 MHz, CDCl
3)
δ: 162.3, 137.3, 133.9, 119.2, 115.5, 115.1, 103.6, 67.4, 29.8, 28.0. 核磁数据与文献报道一致
[34j].
(E)-4-(己-2-烯-1-基氧基)苯腈(3k): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=10∶1, 黄色液体, 45.8 mg, 收率76%. 1H NMR (400 MHz, CDCl3) δ: 7.57 (d, J=8.0 Hz, 2H), 6.95 (d, J=8.0 Hz, 2H), 5.89~5.82 (m, 1H), 5.70~5.63 (m, 1H), 4.52 (d, J=6.0 Hz, 2H), 2.08 (q, J=6.8 Hz, 2H), 1.48~1.39 (m. 2H), 0.91 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 161.9, 136.5, 133.8, 123.7, 119.2, 115.4, 103.7, 69.0, 34.3, 22.0, 13.6; IR (neat) ν: 3332, 2962, 2224, 1604, 1506, 1300, 1252, 1170, 970, 832 cm-1; HRMS (EI) calcd for C13H15NO 201.1148, found 201.1147.
4-((3-苯基丙-2-炔-1-基)氧基)苯腈(
3l): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=20∶1, 黄色固体, m.p. 87~89 ℃ (m.p.
[34k] 90.8~91.8 ℃); 21.8 mg, 收率31%.
1H NMR (400 MHz, CDCl
3)
δ: 7.63~7.60 (m, 2H), 7.43~7.41 (m, 2H), 7.34~7.31 (m, 3H), 7.11~7.07 (m. 2H), 4.97 (s, 2H);
13C NMR (100 MHz, CDCl
3)
δ: 160.9, 133.9, 131.8, 129.0. 128.4, 121.7, 119.0, 115.7, 104.7, 88.2, 82.4, 56.8. 核磁数据与文献报道一致
[34k].
4-((4-氰基苯氧基)甲基)哌啶-1-羧酸苄酯(3m): 三乙胺(φ=10%)润柱, 洗脱剂: V(石油醚)∶V(乙酸乙 酯)=2∶1, 白色固体, 84.8 mg, 收率81%, m.p. 96~97 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.57 (d, J=8.8 Hz, 2H), 7.37~7.36 (m, 4H), 7.34~7.29 (m, 1H), 6.92 (d, J=8.8 Hz, 2H), 5.14 (s, 2H), 4.27 (br, 2H), 3.84 (d, J=6.4 Hz, 2H), 2.83 (br, 2H), 2.04~1.96 (m, 1H), 1.85~1.82 (m, 2H), 1.32~1.29 (m, 2H); 13C NMR (100 MHz, CDCl3) δ: 162.1, 155.1, 136.7, 133.9, 128.4, 127.9, 127.8, 119.1, 115.0, 103.9, 72.3, 67.0, 43.6, 35.8, 28.6; IR (neat) ν: 2928, 2218, 1697, 1605, 1505, 1448, 1280, 1260, 1220, 1173, 1148, 1029, 839, 732, 697 cm-1; HRMS (ESI) calcd for C21H22N2O3Na [M+Na]+ 373.1523, found 373.1516.
6-(萘-1-基甲氧基)-2,3-二氢苯并[
b][
1,
4]二噁嘧啶(
3n): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=20∶1, 淡黄色液体, 22.4 mg, 收率26%.
1H NMR (400 MHz, CDCl
3)
δ: 8.04 (d,
J=8.8 Hz, 1H), 7.89~7.83 (m, 2H), 7.58~7.43 (m, 4H), 6.80 (d,
J=8.8 Hz, 1H), 6.62~6.54 (m, 2H), 5.40 (s, 2H), 4.25~4.19 (m, 4H);
13C NMR (100 MHz, CDCl
3)
δ: 153.5, 143.8, 137.9, 133.8, 132.4, 131.5, 128.9, 128.6, 126.5, 126.4, 125.9, 125.3, 123.7, 117.4, 108.3, 104.0, 69.3, 64.6, 64.1; IR (neat)
ν: 2873, 1596, 1501, 1458, 1272, 1240, 1197, 1157, 1065, 1012, 921, 886, 829, 792, 770 cm
-1; HRMS (EI) calcd for C
19H
16O
3 292.1094, found 292.1100.
6-(萘-2-基甲氧基)-2,3-二氢苯并[
b][
1,
4]二噁嘧啶(
3o): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=20∶1, 白色固体, 42.9 mg, 收率49%, m.p. 89~90 ℃;
1H NMR (400 MHz, CDCl
3)
δ: 7.85~7.81 (m, 4H), 7.52~7.50 (m, 1H), 7.48~7.46 (m, 2H), 6.77 (d,
J=8.8 Hz, 2H), 6.57~6.51 (m, 2H), 5.13 (s, 2H), 4.22~4.17 (m, 4H);
13C NMR (100 MHz, CDCl
3)
δ: 153.4, 143.8, 137.9, 134.6, 133.3, 133.0, 128.3, 127.9, 127.7, 126.24, 126.15, 126.0, 125.3, 117.4, 108.3, 103.9, 70.7, 64.5, 64.1; IR (neat)
ν: 2933, 2870, 1596, 1500, 1465, 1318, 1274, 1213, 1201, 1163, 1066, 1010, 918, 837, 743 cm
-1; HRMS (FI) calcd for C
19H
16O
3 292.1094, found 292.1100.
(4-(4-(4-甲氧基苯基)丁氧基)苯基)(苯基)甲烷酮(3p): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=10∶1, 白色固体, 92.9 mg, 收率86%, m.p. 81~82 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.72 (d, J=8.8 Hz, 2H), 7.66 (d, J=7.2 Hz, 2H), 7.46 (t, J=7.6 Hz, 1H), 7.37 (t, J=6.8 Hz, 2H), 7.02 (d, J=8.8 Hz, 2H), 6.84 (d, J=8.8 Hz, 2H), 6.74 (d, J=8.8 Hz, 2H), 3.94 (t, J=6.0 Hz, 2H), 3.69 (s, 3H), 2.55 (t, J=6.8 Hz, 2H), 1.77~1.69 (m, 4H); 13C NMR (100 MHz, CDCl3) δ: 195.5, 162.7, 157.7, 138.3, 134.0, 132.5, 131.8, 129.9, 129.6, 129.2, 128.1, 113.9, 113.7, 68.0, 55.2, 34.5, 28.5, 27.9; IR (neat) ν: 2933, 2829, 1642, 1598, 1510, 1286, 1263, 1242, 1182, 1173, 859, 851, 742, 707, 700 cm-1; HRMS (ESI) calcd for C24H24O3Na [M+Na]+383.1618, found 383.1614.
(E)-6-((3,7-二甲基辛-2,6-二烯-1-基)氧基)-2-萘甲酸甲酯(3q): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=50∶1, 白色固体, 75.8 mg, 收率75%, m.p. 77~78 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.51 (s, 1H), 8.01 (dd, J=8.4, 1.2 Hz, 1H), 7.82 (d, J=9.2 Hz, 1H), 7.72 (d, J=8.4 Hz, 1H), 7.20 (dd, J=9.2, 2.4 Hz, 1H), 7.15 (d, J=2.0 Hz, 1H), 5.56~5.53 (m, 1H), 5.13~5.08 (m, 1H), 4.66 (d, J=6.4 Hz, 2H), 3.95 (s, 3H), 2.16~2.09 (m, 4H), 1.78 (s, 3H), 1.66 (s, 3H), 1.60 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 167.3, 158.7, 141.7, 137.1, 131.8, 130.8, 127.8, 126.8, 125.8, 125.0, 123.7, 120.0, 119.0, 106.5, 64.9, 52.0, 39.5, 26.2, 25.6, 17.7, 16.7; IR (neat) ν: 2920, 1720, 1626, 1480, 1435, 1291, 1270, 1254, 1199, 1095, 1004, 865, 815, 767, 753 cm-1; HRMS (ESI) calcd for C22H26O3Na [M+Na]+ 361.1774, found 361.1771.
(
E)-4-((3,7-二甲基辛-2,6-二烯-1-基)氧基)苯甲腈(
3r): 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=20∶1, 黄色液体, 55.4 mg, 收率72%.
1H NMR (400 MHz, CDCl
3)
δ: 7.57 (d,
J=8.8 Hz, 2H), 6.95 (d,
J=9.2 Hz, 2H), 5.47~5.43 (m, 1H), 5.09~5.06 (m, 1H), 4.59 (d,
J=6.4 Hz, 2H), 2.14~2.07 (m, 4H), 1.74 (s, 3H), 1.67 (s, 3H), 1.60 (s, 3H);
13C NMR (100 MHz, CDCl
3)
δ: 162.1, 142.2, 133.8, 131.9, 123.5, 119.2, 118.3, 115.4, 103.6, 65.2, 39.4, 26.1, 25.6, 17.6, 16.6. 核磁数据与文献报道一致
[34l].
2-氰基-5-苄氧基吡啶(
3s): 三乙胺(
φ=10%)润柱, 洗脱剂:
V(石油醚)∶
V(乙酸乙酯)=5∶1, 49.6 mg, 收率79%. 淡黄色固体, m.p. 54~55 ℃;
1H NMR (400 MHz, CDCl
3)
δ: 8.44 (d,
J=2.4 Hz, 1H), 7.63 (d,
J=8.8 Hz, 1H), 7.42~7.37 (m, 5H), 7.30 (d,
J=8.8 Hz, 1H), 5.17 (s, 2H);
13C NMR (100 MHz, CDCl
3)
δ: 156.9, 140.6, 134.7, 129.5, 128.8, 128.7, 127.5, 125.3, 120.7, 117.4, 70.7. 核磁数据与文献报道一致
[34m].
5-(萘-2-基甲氧基)吡啶-2-甲腈(3t): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=5∶1, 黄色固体, 45.7 mg, 收率59%, m.p. 91~92 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.46 (d, J=2.8 Hz, 1H), 7.89~7.83 (m, 4H), 7.59 (d, J=8.4 Hz, 1H), 7.53~7.48 (m, 3H), 7.30~7.28 (m, 1H), 5.30 (s, 2H); 13C NMR (100 MHz, CDCl3) δ: 156.9, 140.6, 133.2, 133.1, 132.1, 129.5, 128.8, 127.9, 127.7, 126.7, 126.6, 126.5, 125.4, 124.8, 120.8, 117.4, 70.8; IR (neat) ν: 3272, 3040, 2228, 1580, 1567, 1483, 1459, 1276, 1255, 1036, 1008, 904, 830, 767, 743 cm-1; HRMS (ESI) calcd for C17H13N2O [M+H]+ 261.1022, found 261.1016.
(E)-5-((3,7-二甲基-2,6-二烯-1-基)氧)吡啶-2-甲腈(3u): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=5∶1, 淡黄色液体, 42.6 mg, 收率55%. 1H NMR (400 MHz, CDCl3) δ: 8.36 (d, J=2.8 Hz, 1H), 7.63 (d, J=8.8 Hz, 1H), 7.23 (dd, J=8.8, 3.2 Hz, 1H), 5.46~5.42 (m, 1H). 5.08~5.04 (m, 1H), 4.66 (d, J=6.8 Hz, 2H), 2.14~2.08 (m, 4H), 1.76 (s, 3H), 1.67 (s, 3H), 1.60 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 157.2, 143.4, 140.5, 132.0, 129.4, 124.9, 123.4, 120.6, 117.6, 117.5, 65.7, 39.4, 26.1, 25.6, 17.6, 16.7; IR (neat) ν: 3421, 2928, 2232, 1720, 1570, 1462, 1383, 1305, 1275, 1251, 1197, 1135, 1011, 974, 839 cm-1; HRMS (ESI) calcd for C16H21N2O [M+H]+ 257.1648, found 257.1652.
5-(戊-4-烯-1-基氧基)嘧啶(3v): 三乙胺(φ=10%)润板, 薄层色谱层析: V(石油醚)∶V(乙酸乙酯)=3∶1, 黄色液体, 29.3 mg, 收率59%. 1H NMR (400 MHz, CDCl3) δ: 8.84 (s, 1H), 8.41 (s, 2H), 5.89~5.79 (m, 1H), 5.11~5.02 (m, 2H), 4.08 (t, J=6.4 Hz, 2H), 2.26 (q, J=7.1 Hz, 2H), 1.97~1.90 (m, 2H); 13C NMR (100 MHz, CDCl3) δ: 153.0, 151.3, 143.5, 137.1, 115.7, 67.8, 29.7, 28.0; IR (neat) ν: 2942, 1641, 1558, 1438, 1416, 1341, 1271, 1181, 1113, 1036, 997, 914, 886, 721 cm-1; HRMS (ESI) calcd for C9H13N2O [M+H]+ 165.1022, found 165.1025.
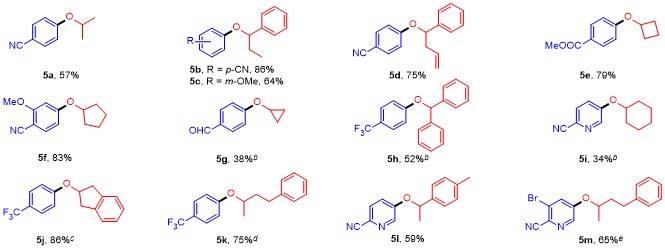
充满N
2的手套箱中, 向干燥的4 mL反应瓶中依次加入4CzIPN (3.6 mg, 1.5 mol%, 0.0045 mmol), NiBr
2• DME (4.6 mg, 5.0 mol%, 0.015 mmol), dtbbpy (4.8 mg, 6.0 mol%, 0.018 mmol), K
3PO
4 (127.4 mg, 2.0 equiv., 0.6 mmol), MeCN (3.0 mL), 4-溴苯腈(54.6 mg, 1.0 equiv., 0.3 mmol)以及异丙醇(27.0 mg, 1.5 equiv., 0.45 mmol). 盖好瓶盖后移出手套箱置于2×40 W blue LED, 功率都设定为75% (0.08~0.09 W/cm
2), 光照条件下反应. 在室温下反应24 h后, TLC检测反应. 反应结束后, 短硅胶柱过滤并用乙酸乙酯冲洗, 旋蒸浓缩, 粗产品用硅胶柱层析[洗脱剂为
V(石油醚)∶
V(乙酸乙酯)=20∶1], 得到4-异丙氧基苯腈(
5a) 27.6 mg, 无色油状液体, 收率57%.
1H NMR (400 MHz, CDCl
3)
δ: 7.56 (d,
J=8.8 Hz, 2H), 6.91 (d,
J=8.8 Hz, 2H), 4.66~4.57 (m, 1H), 1.36 (d,
J=6.0 Hz, 6H);
13C NMR (100 MHz, CDCl
3)
δ: 161.3, 133.9, 119.3, 116.0, 103.3, 70.3, 21.7. 核磁数据与文献报道一致
[34n].
4-(1-苯基丙氧基)苯腈(5b): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=20∶1, 无色油状液体, 61.1 mg, 收率86%. 1H NMR (400 MHz, CDCl3) δ: 7.45 (d, J=8.4 Hz, 2H), 7.35~7.28 (m, 5H), 6.88 (d, J=8.8 Hz, 2H), 5.08~5.05 (m, 1H), 2.08~1.98 (m, 1H), 1.95~1.84 (m, 1H), 0.99 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 161.6, 140.5, 133.7, 128.6, 127.8, 125.8, 119.2, 116.4, 103.6, 81.9, 31.4, 9.9; IR (neat) ν: 2969, 2933, 2223, 1603, 1574, 1505, 1452, 1298, 1249, 1170, 997, 973, 832, 753, 700 cm-1; HRMS (FI) calcd for C16H15NO 237.1148, found 237.1152.
1-甲氧基-3-(1-苯基丙氧基)苯(5c): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=50∶1, 白色固体, 46.5 mg, 收率64%, m.p. 72~73 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.34~7.28 (m, 4H), 7.24~7.21 (m, 1H), 7.05 (t, J=8.8 Hz, 1H), 6.44~6.40 (m, 3H), 4.99 (t, J=6.0 Hz, 1H), 3.70 (s, 1H), 2.05~1.80 (m, 2H), 0.99 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 160.6, 159.6, 141.9, 129.6, 128.4, 127.4, 126.0, 108.1, 106.1, 102.3, 81.4, 55.1, 31.6, 10.2; IR (neat) ν: 2969, 2834, 1591, 1490, 1450, 1283, 1263, 1197, 1169, 1148, 1041, 1000, 753, 699, 685 cm-1; HRMS (EI) calcd for C16H18O2 242.1301, found 242.1304.
4-((1-苯基丁-3-烯-1-基)氧基)苯腈(5d): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=20∶1, 黄色油状液体, 56.3 mg, 收率75%. 1H NMR (400 MHz, CDCl3) δ: 7.46 (d, J=8.4 Hz, 2H), 7.36~7.27 (m, 5H), 6.88 (d, J=8.8 Hz, 2H), 5.87~5.77 (m, 1H), 5.20~5.08 (m, 3H), 2.81~2.74 (m, 1H), 2.64~2.58 (m, 1H); 13C NMR (100 MHz, CDCl3) δ: 161.3, 140.0, 133.8, 133.3, 128.7, 128.0, 125.8, 119.1, 118.1, 116.5, 103.8, 80.2, 42.6; IR (neat) ν: 3058, 2918, 2224, 1603, 1505, 1299, 1248, 1170, 989, 919, 832, 760, 699 cm-1; HRMS (FI) calcd for C17H15NO 249.1148, found 249.1151.
4-环丁氧基苯甲酸甲酯(5e): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=20∶1, 白色固体, 48.6 mg, 收率79%, m.p. 55~56 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.96 (d, J=8.8 Hz, 2H), 6.81 (d, J=8.8 Hz, 2H), 4.72~4.65 (m, 1H), 3.87 (s, 3H), 2.50~2.43 (m, 2H), 2.23~2.13 (m, 2H), 1.92~1.84 (m, 1H), 1.76~1.67 (m, 1H); 13C NMR (100 MHz, CDCl3) δ: 166.8, 161.4, 131.5, 122.3, 114.5, 71.7. 51.8, 30.4, 13.2; IR (neat) ν: 2933, 1709, 1608, 1436, 1267, 1166, 1103, 1077, 964, 849, 773, 696 cm-1; HRMS (ESI) calcd for C12H15O3 [M+H]+ 207.1016, found 207.1011.
4-(环戊氧基)-2-甲氧基苯腈(5f): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=10∶1, 淡黄色液体, 54.1 mg, 收率83%. 1H NMR (400 MHz, CDCl3) δ: 7.44 (d, J=8.4 Hz, 1H), 6.49~6.42 (m, 2H), 4.82~4.79 (m, 1H), 3.88 (s, 3H), 1.98~1.89 (m, 2H), 1.85~1.78 (m, 4H), 1.67~1.63 (m, 2H); 13C NMR (100 MHz, CDCl3) δ: 163.2, 162.7, 134.6, 117.0, 107.0, 99.7, 93.2, 80.0, 55.8, 32.7, 23.9; IR (neat) ν: 2960, 2219, 1604, 1503, 1454, 1283, 1270, 1204, 1165, 1123, 1111, 1027, 989, 835, 803 cm-1; HRMS (EI) calcd for C13H15NO2 217.1097, found 217.1096.
5-(1-(对甲苯基)乙氧基)吡啶-2-甲腈(5l): 洗脱剂: V(石油醚)∶V(乙酸乙酯)=20∶1, 黄色液体, 42.3 mg, 收率59%. 1H NMR (400 MHz, CDCl3) δ: 8.34 (d, J=3.2 Hz, 1H), 7.49 (d, J=8.8 Hz, 1H), 7.22~7.10 (m, 5H), 5.35 (q, J=6.4 Hz, 1H), 2.32 (s, 3H), 1.68 (d, J=6.4 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 156.4, 141.3, 138.1, 137.8, 129.7, 129.3, 125.3, 124.8, 121.6, 117.5, 77.3, 24.3, 21.1; IR (neat) ν: 2980, 2925, 2231, 1568, 1480, 1461, 1301, 1270, 1250, 1182, 1065, 1009, 923, 816, 653 cm-1; HRMS (ESI) calcd for C15H15N2O [M+H]+ 239.1179, found 239.1183.