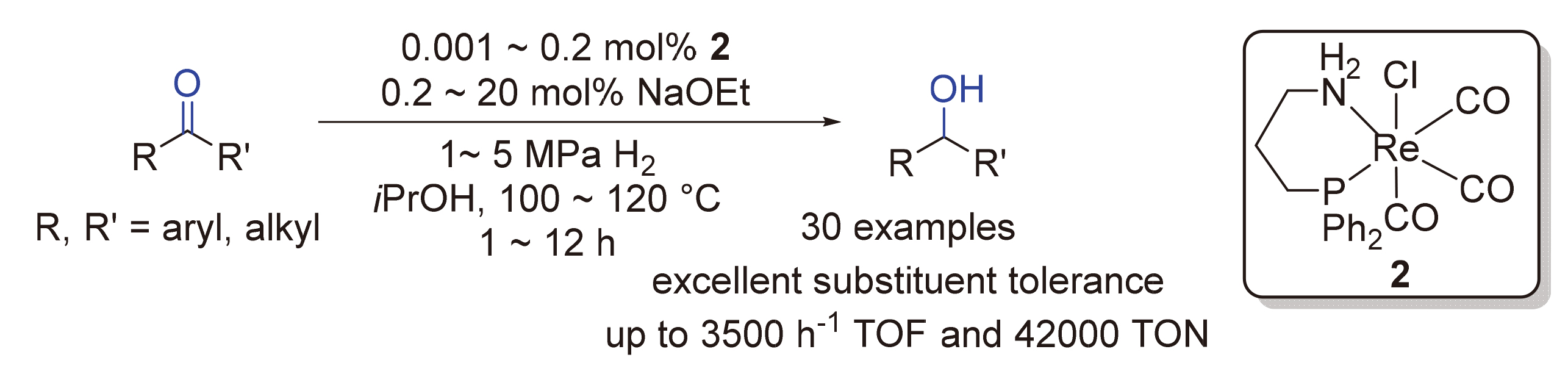
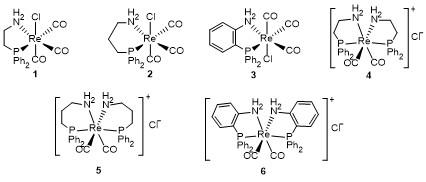
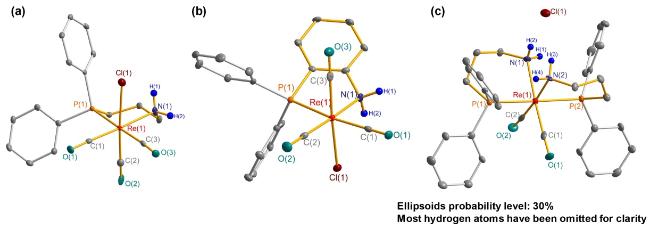
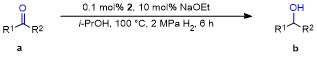
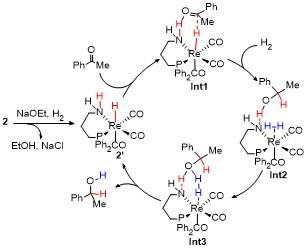
1 结果与讨论
1.1 铼配合物合成及结构表征
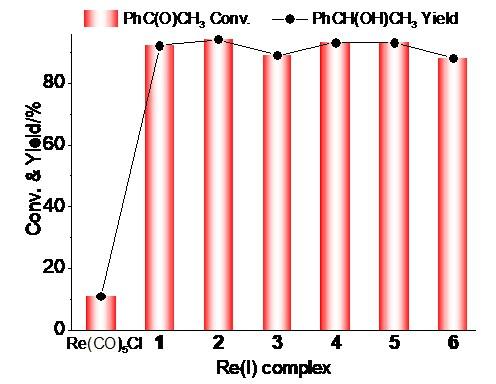
1.2 铼配合物活性评价
表1 铼(I)配合物2催化苯乙酮加氢制备1-苯乙醇aTable 1 Catalytic hydrogenation of acetophenone to 1-phenyl- ethanol by 2 |
| Entry | 2/mol% | Temp./ ℃ | p(H2)/ MPa | Time/h | Yieldb/ % | TOF/ h-1 | TON |
|---|---|---|---|---|---|---|---|
| 1 | 0.1 | 100 | 2 | 1 | 80 | 803 | 803 |
| 2 | 0.1 | 100 | 2 | 4 | 94 | 236 | 942 |
| 3 | 0.1 | 120 | 2 | 1 | >99 | 999 | 999 |
| 4 | 0.1 | 100 | 1 | 4 | 90 | 226 | 904 |
| 5 | 0.1 | 80 | 2 | 8 | 64 | 80 | 636 |
| 6 | 0.1 | 80 | 1 | 8 | 62 | 78 | 624 |
| 7c | 0.1 | 100 | 2 | 4 | 10 | 26 | 104 |
| 8d | 0.1 | 100 | 2 | 4 | 52 | 130 | 518 |
| 9e | 0.1 | 100 | 2 | 4 | 81 | 203 | 812 |
| 10f | 0.01 | 100 | 5 | 12 | >99 | 833 | 9990 |
| 11g | 0.002 | 120 | 5 | 12 | 50 | 2079 | 24950 |
| 12h | 0.001 | 120 | 5 | 12 | 42 | 3500 | 42000 |
a Reaction conditions: 2.0 mmol of acetophenone (0.5 mmol/mL i-PrOH solution), 10 mol% NaOEt. b Determined by GC. c 1 mol% NaOEt. d 2 mol%. e 5 mol%. f 5.0 mmol of acetophenone, 1 mol% NaOEt. g 12.0 mmol of acetophenone, 0.4 mol% NaOEt. h 24.0 mmol of acetophenone, 0.2 mol% NaOEt. |
1.3 底物范围的拓展
表2 铼(I)配合物2催化酮类加氢制醇aTable 2 Catalytic hydrogenation of ketones to alcohols by 2 |
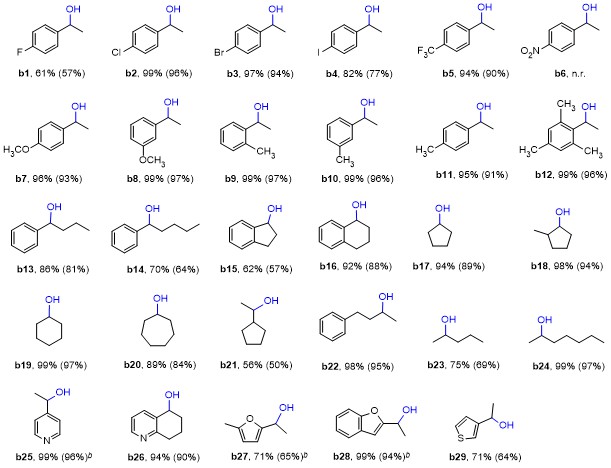 |
a Reaction condition: 2.0 mmol ketone, 4 mL iPrOH, 0.1 mol% 2, 10 mol% NaOEt, 2 MPa H2, 100 ℃, 6 h. Isolated yield in parentheses. b 0.2 mol% 2, 20 mol% NaOEt. |