

Chinese Journal of Organic Chemistry >
Recent Advances on the Functionalization of Azlactones at Different Reactive Sites
† The authors contributed equally to this work
Received date: 2025-07-15
Revised date: 2025-08-19
Online published: 2025-09-12
Supported by
Natural Science Foundation of Hubei Province(2025AFB675)
2024 Shaanxi Higher Education Institutions Outstanding Young Talents Support Program(2025QJ-02(内))
Azlactones constitute a crucial precursor for the preparation of α,α-disubstituted amino acids, N,O-acetal derivatives, many heterocyclic compounds, finding increasingly widespread applications in organic chemistry. Recent advances in functionalization of azlactone have enabled the synthesis of many important organic compounds. Notably, asymmetric transformations of azlactone have facilitated the preparation of valuable chiral compounds, attracting growing interest among researchers in this field. Focusing on the multi-active-site nature of azlactone, this review classifies its participation in diverse organic reactions, including C-2, C-4, and C-5 functionalization and multi-site cycloaddition reactions. Meanwhile, our research group has recently developed a novel method for the in situ formation of azlactones via the intramolecular cyclization of N-hydroxyphthalimide esters, which yields a various of chiral unnatural amino acids. This strategy complements existing azlactone functionalization reactions, thereby promoting the robust development of this field. Finally, the challenges and opportunities in this field are critically assessed, providing insights to direct future research and innovation.
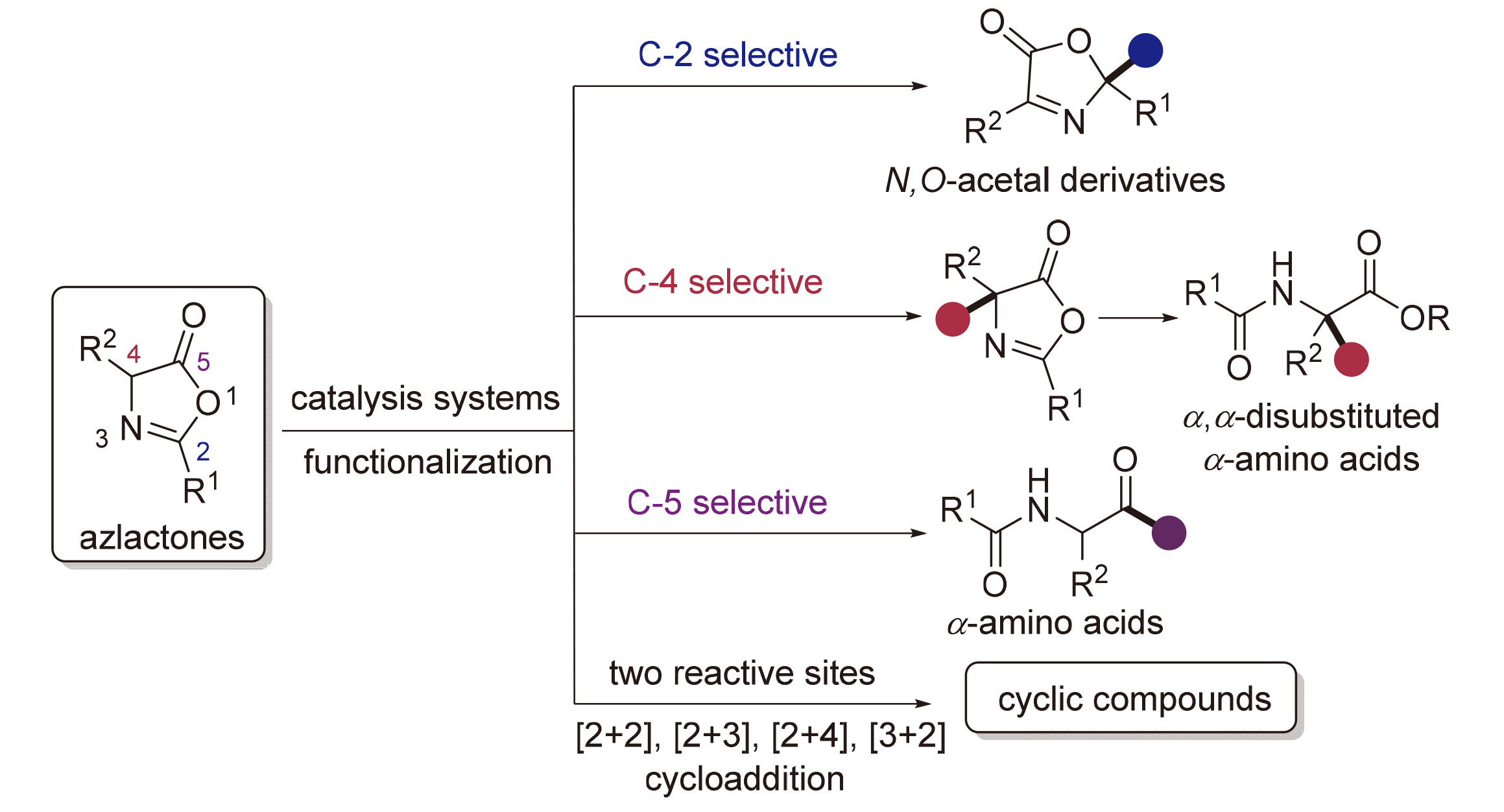
Key words: azlactones; reactive site; functionalization; amino acids; asymmetric transformation
Zhijie Jiang , Jinxiu Chen , Xiaofei Zhu , Xinhao Deng , Qiongjiao Yan , Wei Wang , Hui Zhou . Recent Advances on the Functionalization of Azlactones at Different Reactive Sites[J]. Chinese Journal of Organic Chemistry, 2026 , 46(1) : 1 -20 . DOI: 10.6023/cjoc202506007
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
/
| 〈 |
|
〉 |