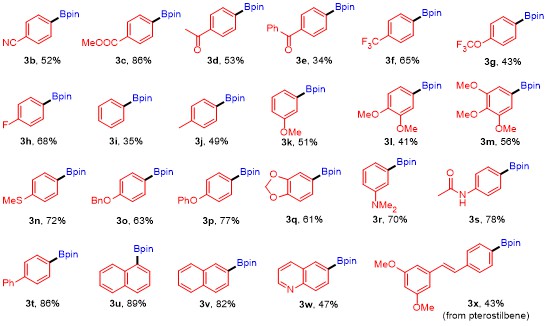
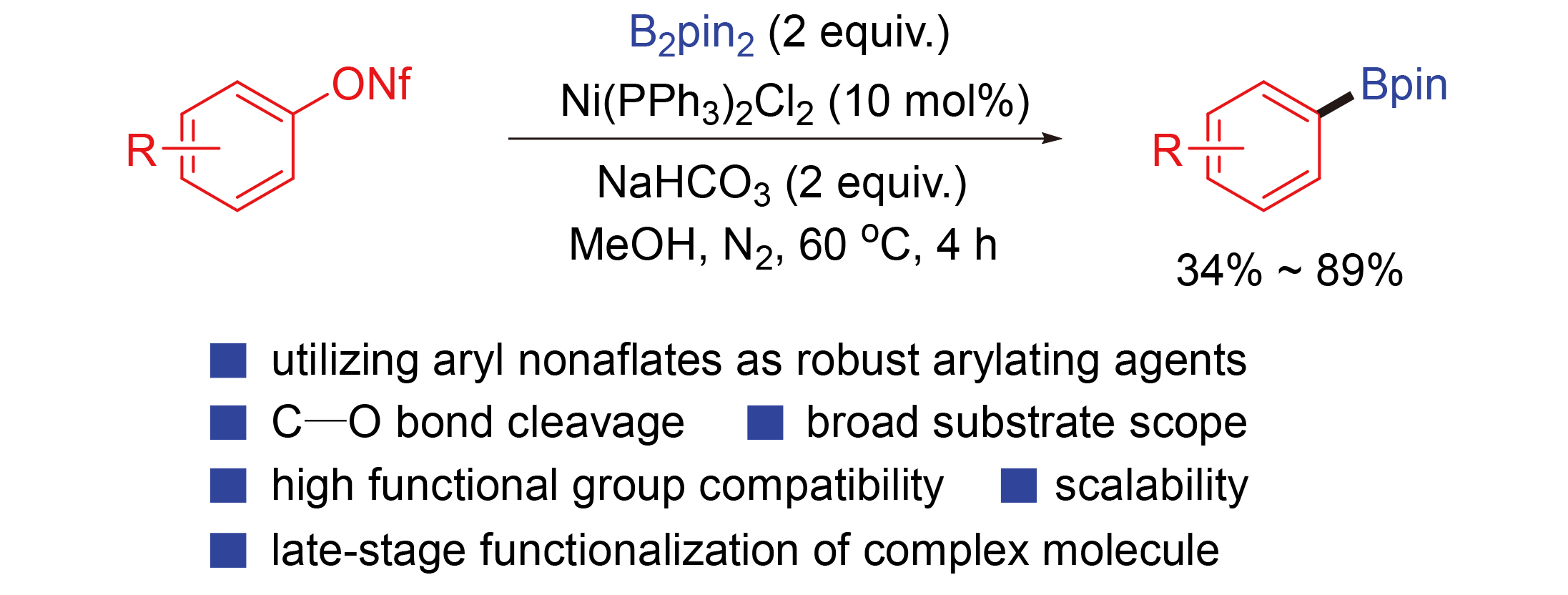With the successful establishment of optimal conditions for the present borylation, we continued to study substrate scope of the reaction by examining a wide array of aryl nonaflates
1 as starting materials. As illustrated in
Table 2, in most cases, the reactions proceeded well with structurally varied aryl nonaflates
1 to afford the corresponding arylboronates
3 in moderate to good yields. For examples, aryl nonaflates
1b~
1h bearing electron-withdrawing groups, including cyano, methoxycarbonyl, acetyl, benzoyl, trifluoromethyl, trifluoromethoxy, and fluoro, were amenable to the transformations, leading to the desired products
3b~
3h in 34%~86% yields. In addition, aryl nonaflates
1j~
1s containing electron-donating substituents, such as methyl, methoxy, methylthio, benzyloxy, phenoxy,
N,
N-dimethylamino, and acetamido, were also proven to be suited for the borylation, and the expected products of arylboronic esters
3j~
3s were furnished in 41%~78% yields. Analogously, the current protocol could also be applied to biphenyl- and naphthyl-substituted nonaflates
1t~
1v, giving rise to the target products
3t~
3v in excellent yields. Moreover, heteroaryl nonaflate
1w derived from quinoline also worked equally well under the optimized reaction conditions to generate the anticipated product
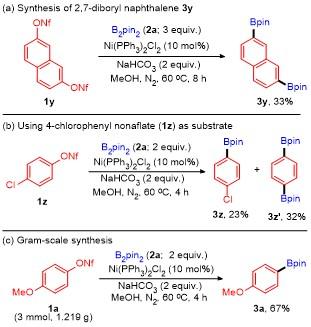
3w in moderate yield. Furthermore, the reaction could also be utilized in the late-stage functionalization of complex molecule, for the use of aryl nonaflate
1x derived from pterostilbene as substrate could also deliver the corresponding arylboronate
3x in 43% yield. However, no desired product was obtained when
ortho-substituted aryl nonaflate (
e.g., 2-methoxyphenyl nonaflate) was used as substrate, presumably owing to the presence of steric hindrance. In addition, the present protocol also could not be applied to the use of heteroaryl nonaflate (
e.g., 3-pyridyl nonaflate) as substrate. Notably, the present deoxygenative borylations proceeded with excellent functional group compatibility, as functionalities or substituents such as CN, COOMe, Ac, Bz, CF
3, OCF
3, F, Me, OMe, SMe, OBn, OPh, OCH
2O, NMe
2, NHAc, and Ph, could be well tolerated, which could potentially be subjected to late-stage modification.