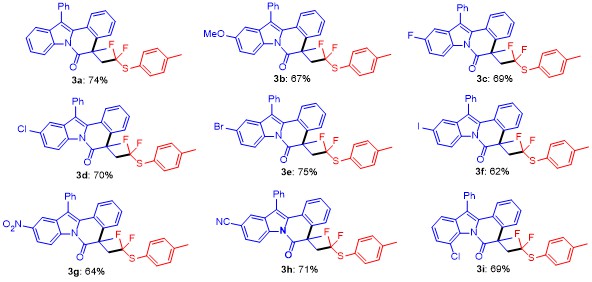
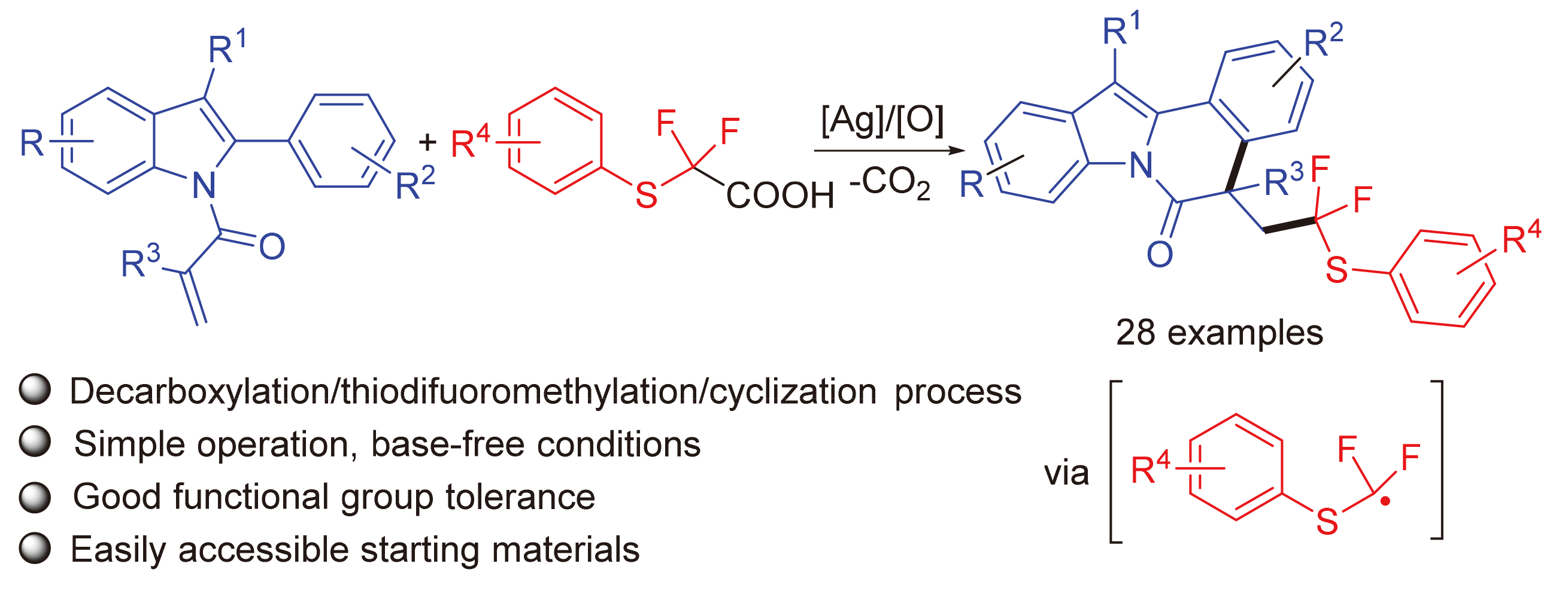
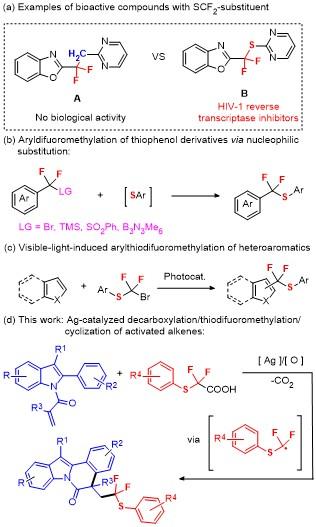
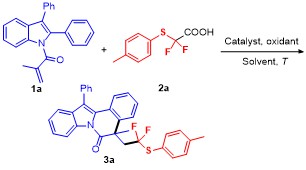
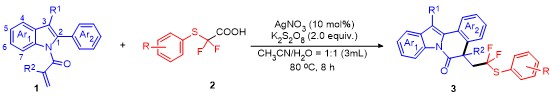
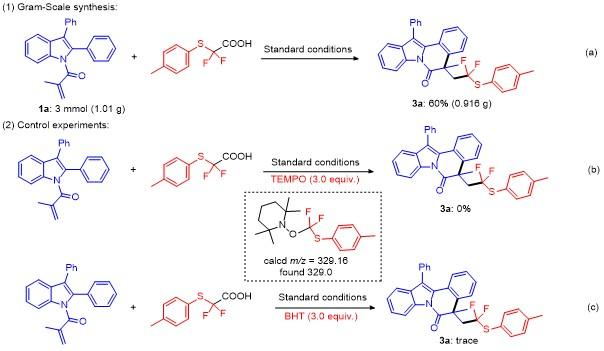
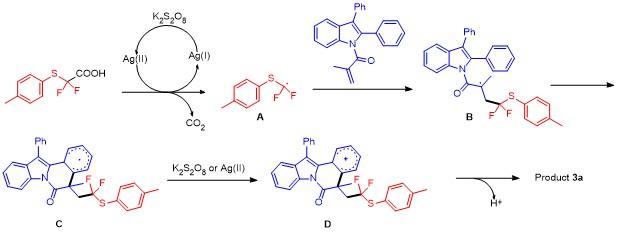
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-phen-ylindolo[2,1-a]isoquinolin-6(5H)-one (3a): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.67 (d, J=8.3 Hz, 1H), 7.60~7.45 (m, 5H), 7.39 (dd, J=14.3, 7.3 Hz, 3H), 7.26 (d, J=9.1 Hz, 5H), 7.01 (dd, J=22.5, 7.7 Hz, 3H), 3.51 (ddd, J=22.5, 15.1, 7.1 Hz, 1H), 3.09~2.85 (m, 1H), 2.27 (s, 3H), 1.74 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.68, 140.13, 136.81, 136.22, 134.30, 134.14, 132.40, 130.23, 129.66, 129.45, 129.21, 128.04, 127.93, 126.90, 126.86, 125.83, 125.28, 124.56, 122.75 (q, J=239 Hz), 119.37, 116.83, 48.86 (q, J=21 Hz), 45.45, 30.92, 26.90, 21.22; 19F NMR (376 MHz, CDCl3) δ: -67.90, -68.44, -70.04, -70.58. HRMS (ESI) calcd for C32H26F2NOS [M+H]+ 510.1703, found 510.1708.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-10-methoxy-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3b): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.57 (d, J=8.8 Hz, 1H), 7.97~7.19 (m, 11H), 7.00 (dd, J=27.7, 7.9 Hz, 3H), 6.75 (d, J=36.9 Hz, 1H), 3.77 (s, 3H), 3.50 (d, J=33.5 Hz, 1H), 3.17~2.85 (m, 1H), 2.27 (s, 3H), 1.72 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.31, 157.35, 140.19, 137.05, 136.96, 136.30, 134.20, 133.55, 130.34, 130.27, 130.20, 129.72, 129.35, 128.97, 128.13, 127.95, 126.99, 126.88, 125.24, 122.79 (q, J=212 Hz), 117.76, 114.01, 102.20, 55.73, 48.88 (q, J=22 Hz), 45.29, 31.06,29.69, 21.30; 19F NMR (376 MHz, CDCl3) δ: -68.01, -68.55, -70.23, -70.77. HRMS (ESI) calcd for C33H28F2NO2S [M+H]+ 540.1809, found 540.1802.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-10-fluoro-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3c): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.61 (dd, J=14.4, 9.4 Hz, 1H), 7.79~7.18 (m, 11H), 7.15~6.74 (m, 4H), 3.68~3.33 (m, 1H), 3.17~2.84 (m, 1H), 2.26 (s, 3H), 1.73 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.60, 160.54 (d, J=240 Hz), 140.28, 137.08, 136.30, 133.84, 133.75, 133.67, 131.02, 130.62, 130.17, 129.76, 129.42, 128.36, 127.02, 125.43, 122.70 (q, J=239 Hz), 118.09, 118.00, 113.50, 113.26, 105.27, 105.03, 48.98 (q, J=21 Hz), 45.38, 30.99, 26.97, 21.30; 19F NMR (376 MHz, CDCl3) δ: -68.16, -68.70, -70.19, -70.74, -117.58. HRMS (ESI) calcd for C32H25F3NOS [M+H]+528.1609, found 528.1601.
10-Chloro-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3d): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.62 (dd, J=26.5, 9.0 Hz, 2H), 8.22~6.93 (m, 14H), 3.73~3.36 (m, 1H), 3.19~2.87 (m, 1H), 2.27 (s, 3H), 1.73 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.73, 140.29, 137.05, 136.29, 133.83, 133.49, 132.63, 130.79, 130.36, 130.23, 129.82, 129.77, 129.51, 129.45, 128.44, 128.40, 127.06, 125.89, 125.47, 124.53, 122.68 (q, J=239 Hz), 119.50, 119.04, 117.94, 49.05 (q, J=22 Hz), 45.45, 30.92, 21.31; 19F NMR (376 MHz, CDCl3) δ: -68.21, -68.75, -70.16, -70.70. HRMS (ESI) calcd for C32H25F2ClNOS [M+H]+ 544.1313, found 544.1307.
10-Bromo-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3e): White solid; m.p. 88~89 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.56 (dd, J=15.9, 8.6 Hz, 1H), 8.12~7.19 (m, 13H), 7.06 (dq, J=21.1, 13.5, 10.9 Hz, 2H), 3.50 (d, J=22.7 Hz, 1H), 3.00 (d, J=18.7 Hz, 1H), 2.27 (s, 3H), 1.73 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.71, 140.26, 137.03, 136.25, 134.26, 133.41, 132.95, 130.61, 130.19, 129.77, 129.73, 129.46, 129.42, 128.56, 128.42, 128.37, 127.02, 125.44, 122.61 (q, J=220 Hz), 122.06, 119.34, 118.26, 118.10, 49.05 (q, J=21 Hz), 45.45, 30.86, 26.94, 21.27; 19F NMR (376 MHz, CDCl3) δ: -68.27, -68.81, -70.21, -70.76. HRMS (ESI) calcd for C32H25F2BrNOS [M+H]+ 588.0808, found 588.0802.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-10-iodo-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3f): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.86~8.28 (m, 2H), 7.87~7.20 (m, 12H), 7.17~6.89 (m, 2H), 3.51 (d, J=32.7 Hz, 1H), 3.18~2.84 (m, 1H), 2.27 (s, 3H), 1.73 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.79, 140.30, 137.03, 136.30, 134.68, 134.34, 133.57, 133.45, 132.45, 130.30, 129.78, 129.51, 129.29, 128.45, 128.18, 127.07, 125.90, 125.48, 124.93, 124.64, 122.63 (q, J=222 Hz), 119.44, 118.67, 116.90, 89.09, 48.88 (q, J=22 Hz), 45.51, 30.88, 26.96, 21.32; 19F NMR (376 MHz, CDCl3) δ: -67.96, -68.22, -68.50, -68.77, -70.16, -70.71. HRMS (ESI) calcd for C32H25F2INOS [M+H]+ 636.0670, found 636.0662.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-5-methyl-10-nitro-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3g): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.94~8.71 (m, 1H), 8.62~8.06 (m, 3H), 7.99~6.93 (m, 12H), 3.48 (t, J=15.5 Hz, 1H), 3.03 (d, J=27.4 Hz, 1H), 2.27 (s, 3H), 1.77 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 172.18, 145.11, 140.43, 137.29, 137.01, 136.27, 132.73, 132.64, 132.52, 132.44, 130.13, 129.82, 129.70, 129.09, 128.83, 128.05, 127.31, 127.07, 125.75, 124.01, 122.47 (q, J=240 Hz), 120.97, 117.08, 115.50, 49.25 (q, J=22 Hz), 45.66, 30.79, 21.31; 19F NMR (376 MHz, CDCl3) δ: -68.52, -69.06, -70.05, -70.60. HRMS (ESI) calcd for C32H25F2N2O3S [M+H]+ 555.1554, found 555.1549.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-5-methyl-6-oxo-12-phenyl-5,6-dihydroindolo[2,1-a]isoquinoline-10-carbonitrile (3h): White solid, m.p. 197~198 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.77 (q, J=10.2, 9.4 Hz, 1H), 8.55~7.19 (m, 13H), 7.07 (dd, J=17.7, 8.1 Hz, 2H), 3.71~3.37 (m, 1H), 3.04 (s, 1H), 2.28 (s, 3H), 1.76 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 172.15, 140.43, 137.03, 137.00, 136.30, 136.26, 136.10, 132.80, 132.63, 131.64, 130.11, 129.79, 129.60, 128.98, 128.93, 128.86, 128.74, 127.26, 127.06, 125.70, 122.50 (q, J=220 Hz), 119.54, 117.63, 108.01, 49.17 (q, J=22 Hz), 45.65, 30.86, 29.78, 21.33; 19F NMR (376 MHz, CDCl3) δ: -68.48, -69.02, -70.12, -70.66. HRMS (ESI) calcd for C33H25F2N2OS [M+H]+ 535.1656, found 535.1650.
8-Chloro-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3i): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.96~7.15 (m, 14H), 7.15~6.90 (m, 2H), 3.46 (d, J=23.1 Hz, 1H), 3.22~2.83 (m, 1H), 2.24 (s, 3H), 1.82 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.16, 139.90, 137.41,135.98, 135.93, 133.16, 132.61, 132.16, 130.09, 129.51, 129.47, 129.10, 128.22, 128.14, 128.08, 127.52, 126.80, 126.58, 125.56, 125.51, 125.25, 122.79, 122.07(q, J=227 Hz), 117.92, 48.28 (q, J=23 Hz), 46.44, 29.01, 21.04; 19F NMR (376 MHz, CDCl3) δ: -67.44, -67.99, -70.25, -70.80. HRMS (ESI) calcd for C32H25F2ClNOS [M+H]+ 544.1313, found 544.1307.
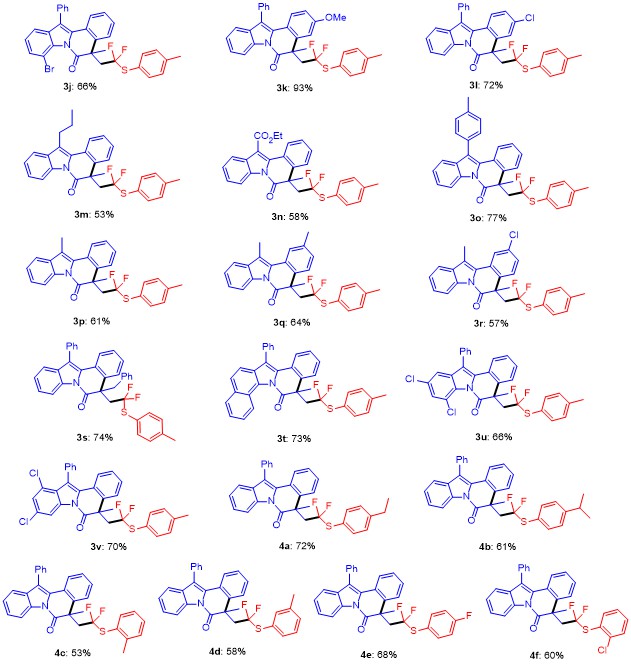
8-Bromo-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3j): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.34~7.20 (m, 14H), 7.17~6.94 (m, 2H), 3.75~3.37 (m, 1H), 2.96 (d, J=17.2 Hz, 1H), 2.26 (s, 3H), 1.82 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.10, 140.16, 137.51, 136.50, 136.25, 134.48, 133.32, 133.05, 131.04, 130.33, 129.72, 129.34, 129.09, 128.43, 128.34, 127.01, 125.85, 125.72, 125.58, 123.24 (q, J=211 Hz), 119.98, 118.76, 109.98, 48.21(q,J=21 Hz), 46.71, 30.33, 27.12, 21.30; 19F NMR (376 MHz, CDCl3) δ: -67.13, -67.68, -69.94, -70.49. HRMS (ESI) calcd for C32H25F2BrNOS [M+H]+ 588.0808, found 588.0801.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-3-methoxy-5-me-thyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3k): White solid, m.p. 104~105 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.62 (t, J=9.8 Hz, 1H), 7.69~7.17 (m, 11H), 7.14~6.75 (m, 3H), 6.73~6.48 (m, 1H), 3.77 (s, 3H), 3.47 (s, 1H), 2.93 (d, J=17.6 Hz, 1H), 2.27 (s, 3H), 1.72 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.58, 159.30, 138.86, 136.25, 134.35, 134.21, 132.64, 130.42, 129.81, 129.72, 129.33, 129.24, 127.97, 126.92, 125.38, 124.55, 122.90, 119.04 (q, J=229 Hz), 118.58, 118.04, 116.75, 112.75, 112.54, 55.34, 48.96 (q, J=21 Hz), 45.73, 31.02, 21.26; 19F NMR (376 MHz, CDCl3) δ: -67.81, -68.35, -69.90, -70.44. HRMS (ESI) calcd for C33H28F2NO2S [M+H]+ 540.1809, found 540.1803.
3-Chloro-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3l): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.67 (dd, J=17.3, 8.2 Hz, 1H), 8.32~7.18 (m, 13H), 7.04 (ddd, J=39.9, 17.2, 8.3 Hz, 2H), 3.73~3.37 (m, 1H), 2.95 (s, 1H), 2.29 (s, 3H), 1.73 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.98, 140.33, 138.71, 136.26, 134.34, 133.77, 132.27, 130.18, 129.81, 129.39, 128.59, 128.31, 127.43, 127.02, 126.57, 126.18, 123.54, 122.64 (q, J=215 Hz), 120.86, 119.54, 116.89, 48.82 (q, J=21 Hz), 45.48, 30.87, 21.32; 19F NMR (376 MHz, CDCl3) δ: -68.12, -68.66, -70.15, -70.70. HRMS (ESI) calcd for C32H25F2ClNOS [M+H]+ 544.1313, found 544.1307.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-prop-ylindolo[2,1-a]isoquinolin-6(5H)-one (3m): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.64 (d, J=7.8 Hz, 1H), 7.94 (d, J=7.7 Hz, 1H), 7.58 (d, J=7.8 Hz, 1H), 7.46~7.28 (m, 5H), 7.23 (d, J=8.0 Hz, 2H), 7.01 (d, J=7.6 Hz, 2H), 3.48 (ddd, J=22.6, 15.2, 7.2 Hz, 1H), 3.16~3.01 (m, 2H), 3.01~2.83 (m, 1H), 2.26 (s, 3H), 1.80 (dt, J=14.7, 7.4 Hz, 2H), 1.67 (d, J=6.0 Hz, 3H), 1.11 (t, J=7.3 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.38, 140.05, 136.65, 136.20, 134.42, 132.28, 129.60, 129.14, 127.98, 127.43, 127.39, 127.24, 125.85, 125.63, 124.64, 124.21, 122.80 (q, J=239 Hz), 119.84, 118.40, 116.98, 48.80 (q, J=21 Hz), 45.30, 31.14, 27.38, 22.27, 21.20, 14.49; 19F NMR (376 MHz, CDCl3) δ: -67.93, -68.48, -70.15, -70.66. HRMS (ESI) calcd for C29H28F2NOS [M+H]+ 476.1860, found 476.1866.
Ethyl 5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5-methyl-6-oxo-5,6-dihydroindolo[2,1-a]isoquinoline-12-carboxylate (3n): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.56 (ddd, J=69.8, 16.7, 7.9 Hz, 2H), 7.93 (dd, J=16.6, 7.5 Hz, 1H), 7.63~7.19 (m, 7H), 7.04 (dd, J=16.3, 7.6 Hz, 2H), 4.57 (dd, J=16.2, 7.3 Hz, 2H), 3.44 (d, J=15.8 Hz, 1H), 3.01 (d, J=18.3 Hz, 1H), 2.27 (s, 3H), 1.71 (s, 3H), 1.64~1.39 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 172.12, 165.84, 140.26, 137.77, 136.27, 134.25, 129.73, 129.05, 127.74, 127.34, 127.30, 126.65, 126.27, 126.23, 125.20, 123.31, 122.54, 122.0 (q, J=240 Hz), 120.91, 116.79, 110.63, 61.39, 48.89 (q, J=22 Hz), 45.85, 30.38, 21.28, 14.41; 19F NMR (376 MHz, CDCl3) δ: -67.99, -68.53, -69.79, -70.34. HRMS (ESI) calcd for C29- H26F2NO3S [M+H]+ 506.1601, found 506.1606.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-(p-tolyl)indolo[2,1-a]isoquinolin-6(5H)-one (3o): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.67 (d, J=8.1 Hz, 1H), 7.57~7.18 (m, 12H), 7.01 (t, J=8.0 Hz, 3H), 3.63~3.39 (m, 1H), 3.07~2.87 (m, 1H), 2.47 (s, 3H), 2.26 (s, 3H), 1.72 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.65, 140.10, 137.73, 136.70, 136.21, 134.27, 132.52, 130.94, 130.79, 130.02, 129.92, 129.64, 129.34, 127.99, 127.84, 126.85, 126.81, 125.76, 125.26, 124.50, 122.73 (q, J=225 Hz), 119.41, 116.79, 48.79 (q, J=22 Hz), 45.42, 30.92, 26.88, 21.43, 21.21; 9F NMR (376 MHz, CDCl3) δ: -67.89, -68.43, -70.09, -70.63. HRMS (ESI) calcd for C33H28F2NOS [M+H]+ 524.1860, found 524.1854.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-5,12-dimethyl-indolo[2,1-a]isoquinolin-6(5H)-one (3p): Pale yellow solid, m.p. 113~114 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.56 (dd, J=20.7, 10.2 Hz, 1H), 7.90~7.70 (m, 1H), 7.64~7.09 (m, 8H), 6.98 (dd, J=21.8, 8.2 Hz, 2H), 3.58~3.28 (m, 1H), 2.99~2.72 (m, 2H), 2.50 (s, 3H), 2.18 (s, 3H), 1.59 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.58, 140.27, 138.74, 136.23, 134.35, 132.35, 130.61, 130.30, 129.81, 129.76, 128.76, 126.41, 126.04, 124.47, 122.65 (q, J=238 Hz), 121.28, 118.49, 116.96, 115.15, 48.72 (q, J=23 Hz), 45.30, 31.14, 29.48, 21.30, 11.62; 19F NMR (376 MHz, CDCl3) δ: -68.22, -68.76, -70.32, -70.87. HRMS (ESI) calcd for C27H24F2NOS [M+H]+ 448.1547, found 448.1552.
5-(2,2-Difluoro-2-(p-tolylthio)ethyl)-2,5,12-trimethyl-indolo[2,1-a]isoquinolin-6(5H)-one (3q): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.75~8.58 (m, 1H), 7.82 (d, J=12.9 Hz, 1H), 7.68~7.14 (m, 8H), 7.13~6.96 (m, 2H), 3.66~3.38 (m, 1H), 3.07~2.87 (m, 1H), 2.67 (s, 3H), 2.42 (s, 3H), 2.29 (s, 3H), 1.69 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.54, 140.03, 136.79, 136.23, 134.28, 133.76, 132.50, 130.83, 129.62, 128.43, 127.08, 125.79, 125.54, 125.36, 124.16, 122.76 (q, J=240 Hz), 118.23, 116.88, 114.18, 48.61 (q, J=21 Hz), 45.08, 31.31, 21.47, 21.21, 11.64; 19F NMR (376 MHz, CDCl3) δ: -68.04, -68.58, -70.67, -71.21. HRMS (ESI) calcd for C28H26F2NOS [M+H]+ 462.1703, found 462.1708.
2-Chloro-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5,12-dimethylindolo[2,1-a]isoquinolin-6(5H)-one (3r): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.83~8.55 (m, 1H), 8.54~7.16 (m, 9H), 7.15~6.94 (m, 1H), 3.76~3.40 (m, 1H), 3.07~2.83 (m, 1H), 2.62 (s, 3H), 2.26 (s, 3H), 1.65 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.87, 140.28, 136.28, 135.04, 134.37, 133.30, 132.23, 129.74, 128.77, 128.00, 127.59, 127.41, 126.21, 125.05, 124.64, 124.48, 122.59 (q, J=240 Hz), 118.62, 116.99, 115.66, 48.67 (q, J=22 Hz), 45.17, 31.21, 21.29, 11.60; 19F NMR (376 MHz, CDCl3) δ: -67.94, -68.48, -70.59, -71.13. HRMS (ESI) calcd for C27H23F2ClNOS [M+H]+482.1157, found 482.1160.
5-Benzyl-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-12-phen-ylindolo[2,1-a]isoquinolin-6(5H)-one (3s): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.61 (d, J=8.2 Hz, 1H), 7.54~7.11 (m, 11H), 7.09~6.95 (m, 4H), 6.86 (q, J=7.4 Hz, 2H), 6.70 (t, J=7.6 Hz, 2H), 6.44 (d, J=7.5 Hz, 2H), 3.86~3.65 (m, 1H), 3.44 (d, J=12.4 Hz, 1H), 3.23~3.06 (m, 1H), 2.98 (d, J=12.4 Hz, 1H), 2.21 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.38, 140.17, 136.31, 134.06, 133.96, 133.93, 132.05, 130.92, 130.06, 129.69, 129.49, 129.07, 129.02, 128.13, 127.85, 127.50, 127.35, 127.33, 126.98, 126.85, 126.58, 125.63, 125.04, 124.38, 122.96 (q, J=238 Hz), 119.90, 119.14, 116.69, 51.81, 51.08, 46.66 (q, J=22 Hz), 21.26; 19F NMR (376 MHz, CDCl3) δ: -65.26, -65.80, -68.52, -69.06. HRMS (ESI) calcd for C38H30F2NOS [M+H]+ 586.2016, found 586.2013.
5-(2,2-Difluoro-2-(
p-tolylthio)ethyl)-5-methyl-14-phen-ylbenzo[
6,
7]indolo[2,1-
a]isoquinolin-6(5
H)-one (
3t): Pale yellow oil.
1H NMR (400 MHz, CDCl
3)
δ: 8.93 (dd,
J=16.8, 8.9 Hz, 1H), 8.53~7.50 (m, 9H), 7.47~6.61 (m, 9H), 3.56 (d,
J=6.9 Hz, 1H), 3.02 (t,
J=9.3 Hz, 1H), 2.25 (s, 3H), 1.76 (s, 3H);
13C NMR (101 MHz, CDCl
3)
δ: 172.58, 140.19, 136.83, 136.40, 136.24, 131.79, 130.50, 130.27, 130.01, 129.92, 129.68, 129.02, 128.53, 128.49, 127.46, 127.00, 126.85, 126.75, 125.92, 125.60, 125.21, 124.83, 124.68, 123.61, 122.7 (q,
J=230 Hz), 121.98, 116.53, 49.00 (q,
J=26 Hz), 45.66, 30.98, 21.24;
19F NMR (376 MHz, CDCl
3)
δ: -68.11, -68.65, -70.03, -70.57. HRMS (ESI) calcd for C
36H
28F
2NOS [M+H]
+ 560.1860, found 560.1867.
8,10-Dichloro-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3u): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 7.60~7.16 (m, 12H), 7.00 (d, J=7.9 Hz, 3H), 3.50~3.27 (m, 1H), 3.05~2.82 (m, 1H), 2.24 (s, 3H), 1.81 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.30, 140.23, 137.83, 136.90, 136.18, 134.13, 132.76, 131.00, 130.60, 130.22, 129.73, 129.52, 128.88, 128.61, 128.08, 127.29, 127.16, 126.78, 125.91, 125.28, 122.96, 122.85 (q, J=225 Hz), 119.31, 117.87, 48.70 (q, J=22 Hz), 46.71, 28.95, 21.28; 19F NMR (376 MHz, CDCl3) δ: -67.71, -68.26, -70.25, -70.80. HRMS (ESI) calcd for C32H24F2Cl2NOS [M+H]+ 578.0924, found 578.0920.
9,11-Dichloro-5-(2,2-difluoro-2-(p-tolylthio)ethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (3v): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.94~8.68 (m, 1H), 8.65~6.79 (m, 14H), 3.68~3.33 (m, 1H), 3.01 (s, 1H), 2.28 (s, 3H), 1.71 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.96, 140.39, 136.84, 136.32, 135.42, 134.26, 131.47, 131.28, 130.95, 130.76, 129.82, 128.99, 128.90, 128.54, 128.43, 127.18, 126.99, 126.85, 126.24, 125.64, 124.29, 122.60 (q, J=240 Hz), 119.44, 115.91, 49.12 (q, J=22 Hz), 45.56, 31.04, 21.34; 19F NMR (376 MHz, CDCl3) δ: -68.39, -68.93, -70.05, -70.59. HRMS (ESI) calcd for C32H24F2Cl2NOS [M+H]+ 578.0924, found 578.0920.
5-(2-((4-Ethylphenyl)thio)-2,2-difluoroethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (4a): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.66 (dd, J=12.8, 8.3 Hz, 1H), 7.71~6.89 (m, 16H), 3.69~3.41 (m, 1H), 2.99 (s, 1H), 2.76~2.47 (m, 2H), 1.74 (s, 3H), 1.28~1.07 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.71, 146.32, 138.14, 136.86, 136.32, 134.34, 134.19, 132.45, 130.27, 129.24, 129.22, 128.50, 128.47, 128.07, 127.97, 126.92, 125.87, 125.33, 124.94, 124.60, 123.01 (q, J=238 Hz), 120.41, 119.40, 116.88, 48.90 (q, J=21 Hz), 45.53, 31.02, 28.56, 15.22; 19F NMR (376 MHz, CDCl3) δ: -67.85, -68.36, -70.06, -70.61. HRMS (ESI) calcd for C33H28F2NOS [M+H]+ 524.1860, found 524.1855.
5-(2,2-Difluoro-2-((4-isopropylphenyl)thio)ethyl)-5-me- thyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (4b): Pale yellow solid, m.p. 88~89 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.63 (dd, J=22.8, 8.8 Hz, 2H), 8.14~6.83 (m, 15H), 3.48 (d, J=22.5 Hz, 1H), 3.11~2.65 (m, 2H), 1.66 (s, 3H), 1.12 (d, J=23.6 Hz, 6H); 13C NMR (101 MHz, CDCl3) δ: 171.76, 150.91, 136.84, 136.40, 134.34, 134.18, 132.47, 130.33, 129.51, 129.34, 129.28, 128.14, 128.00, 127.17, 127.12, 127.00, 126.93, 125.91, 125.36, 124.91, 124.65, 123.04 (q, J=240 Hz), 119.44, 116.92, 48.83(q, J=22 Hz), 45.54, 33.89, 31.17, 23.79; 19F NMR (376 MHz, CDCl3) δ: -67.78, -68.32, -70.11, -70.65. HRMS (ESI) calcd for C34H30F2NOS [M+H]+ 538.2016, found 538.2012.
5-(2,2-Difluoro-2-(o-tolylthio)ethyl)-5-methyl-12-phen- ylindolo[2,1-a]isoquinolin-6(5H)-one (4c): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.97~8.57 (m, 1H), 8.53~6.88 (m, 16H), 3.83~3.49 (m, 1H), 3.02 (t, J= 12.1 Hz, 1H), 2.20 (s, 3H), 1.74 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.74, 143.68, 138.20, 136.74, 134.28, 134.16, 132.46, 130.49, 130.26, 130.22, 129.45, 129.27, 128.10, 127.99, 127.05, 126.94, 126.22, 125.90, 125.57, 125.26, 124.95, 124.63, 120.41 (q, J=240 Hz), 119.42, 116.87, 49.01 (q, J=22 Hz), 45.51, 31.32, 21.22; 19F NMR (376 MHz, CDCl3) δ: -66.66, -67.20, -68.11, -68.65. HRMS (ESI) calcd for C32H26F2NOS [M+H]+ 510.1703, found 510.1709.
5-(2,2-Difluoro-2-(m-tolylthio)ethyl)-5-methyl-12-phen- ylindolo[2,1-a]isoquinolin-6(5H)-one (4d): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 3.48 (d, J=14.8 Hz, 1H), 2.93 (d, J=18.7 Hz, 1H), 2.17 (s, 3H), 1.67 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.68, 138.72, 136.76, 136.68, 134.29, 134.10, 133.11, 132.41, 130.59, 130.22, 129.45, 129.22, 128.64, 128.06, 127.95, 126.93, 126.88, 125.93, 125.85, 125.27, 124.87, 124.59, 120.37 (q, J=238 Hz), 119.39, 116.86, 48.85 (q, J=21 Hz), 45.45, 31.04, 26.90, 21.12; 19F NMR (376 MHz, CDCl3) δ: -67.50, -68.04, -69.71, -70.25. HRMS (ESI) calcd for C32H26- F2NOS [M+H]+ 510.1703, found 510.1709.
5-(2,2-Difluoro-2-((4-fluorophenyl)thio)ethyl)-5-meth- yl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (4e): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.67 (d, J=8.3 Hz, 1H), 7.73~7.20 (m, 13H), 6.96 (dt, J=33.4, 8.1 Hz, 3H), 3.51 (ddd, J=22.6, 15.2, 7.8 Hz, 1H), 3.11~2.88 (m, 1H), 1.74 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 171.60, 163.89 (d, J=249 Hz), 138.47, 138.39, 136.67, 134.30, 134.08, 132.41, 130.21, 129.24, 128.10, 127.97, 126.95, 126.88, 125.90, 125.32, 124.92, 124.64, 121.52 (q, J=236 Hz), 120.48, 119.43, 116.83, 116.18, 115.96, 48.75 (q, J=21 Hz), 45.48, 31.01; 19F NMR (376 MHz, CDCl3) δ: -67.75, -68.29, -69.99, -70.53, -110.50. HRMS (ESI) calcd for C31H23F3NOS [M+H]+ 514.1452, found 514.1455.
5-(2-((2-Chlorophenyl)thio)-2,2-difluoroethyl)-5-methyl-12-phenylindolo[2,1-a]isoquinolin-6(5H)-one (4f): Pale yellow oil. 1H NMR (400 MHz, CDCl3) δ: 8.60 (d, J=8.2 Hz, 1H), 7.53~7.09 (m, 14H), 6.97 (dt, J=37.0, 7.7 Hz, 2H), 3.65~3.42 (m, 1H), 3.00 (d, J=11.8 Hz, 1H), 1.66 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 170.54, 138.42, 137.26, 135.50, 133.24, 133.07, 131.40 (q, J=232 Hz), 130.05, 129.18, 129.02, 128.40, 128.20, 127.17, 127.04, 126.97, 125.94, 124.84, 124.27, 123.92, 123.57, 119.41, 118.37, 115.79, 47.87 (q, J=21 Hz), 44.44, 30.18; 19F NMR (376 MHz, CDCl3) δ: -67.05, -67.59, -67.89, -68.43. HRMS (ESI) calcd for C31H23F2ClNOS [M+H]+ 530.1157, found 530.1151.
Supporting Information Copies of
1H NMR and
13C NMR spectra of the products
3a~
4f. The Supporting Information is available free of charge
via the Internet at
http://siocjournal.cn/.