4.1 General Information
Unless otherwise noted, all oxygen or moisture sensitive reactions were conducted in flame-dried glassware under nitrogen or argon atmosphere. Dry tetrahydrofuran (THF) and dichloromethane (DCM) were obtained from a solvent purification system on Innovative Technology. Other solvents and reagents were purchased from commercial sources and were used without further purification. An oil bath was used as heating source for reactions that require heating. Analytical thin layer chromatography (TLC) was performed on 0.2 mm thick silica gel 60-F254 plates (Merck) and visualized by exposure to ultraviolet light, or an ethanolic solution of phosphomolybdic acid. Column chromatographic purification of products was accomplished using forced-flow chromatography on 100~200 or 230~400 mesh silica gel. The 1H NMR spectra were recorded at 300, 400, or 500 MHz; and 13C NMR spectra were recorded at 75, 101, or 126 MHz on Bruker AV. Chemical shifts are given relative to TMS or the appropriate solvent peak. High resolution mass spectra (HRMS) were obtained on an IonSpec Ultima 7.0 T FT-ICR-MS (IonSpec, USA) using ESI or APCI as ionization method. Melting points were measured using a micro-melting point apparatus.
4.2 Deuterium labeling experiments for the isomerization of E-7 to Z-7
To a stirred solution of E-7 (38.3 mg, 0.100 mmol) in dimethyl sulfoxide (DMSO)/D2O (2 mL, V∶V=10∶1) was added BnMe3NOH [16.7 mg, 40% (w) in MeOH, 0.040 mmol, 0.4 equiv.] at room temperature under N2 atmosphere. The reaction mixture was stirred for 15 min at the same temperature and quenched by saturated aqueous NH4Cl. The solution was diluted with water (5 mL) and extracted with EtOAc (4 mL×4). The combined organic layers were washed successively with water (5 mL×3) and brine, then dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [eluent: V(petroleum ether)∶ V(EtOAc)=15∶1] afforded compound E-7-D (16.0 mg, 0.042 mmol, 42% yield) as a colorless oil and crude Michael/aldol product 9-D.
(4aS,8aS,E)-3-(((S)-3-Isopropyl-1-(2-oxopropyl)cyclo-pent-2-en-1-yl)methylene)-5,5,8a-trimethyl-3,4,4a,5,6,7,8,8a-octahydronaphthalene-1-carbaldehyde (E-7-D): 1H NMR (300 MHz, CDCl3) δ: 9.29 (s, 1H), 6.59 (s, 1H), 6.00 (d, J=2.2 Hz, 1H), 5.44~5.42 (m, 1H), 2.83 (d, J=15.7 Hz, 1H), 2.76~2.69 (m, 1.65H), 2.37~2.28 (m, 3H), 2.11 (s, 0.3H), 2.09~2.03 (m, 2H), 1.97~1.86 (m, 1H), 1.66~1.56 (m, 1H), 1.53~1.49 (m, 1H), 1.46~1.40 (m, 1H), 1.25~1.17 (m, 2H), 1.11 (s, 3H), 1.13~1.08 (m, 1H), 1.05 (d, J=6.7 Hz, 3H), 1.04 (d, J=6.5 Hz, 3H), 0.91 (s, 3H), 0.89 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 193.7, 152.3, 151.4, 148.3, 145.4 (×2), 136.6 (×2), 127.2, 51.8 (×2), 50.7, 41.9, 38.0 (broad), 37.3, 35.3, 33.6, 33.5, 32.0, 31.5, 29.9, 22.6, 21.6, 21.5, 21.4, 19.2, 18.8; HRMS (ESI) calcd for C26H36D3O2 [M+H]+ 386.3133, found 386.3129; calcd for C26H35D4O2 [M+H]+ 387.3196, found 387.3191; calcd for C26H34D5O2 [M+H]+ 388.3258, found 388.3249.
To a stirred solution of Michael/aldol product 9-D in toluene (1 mL) was added p-TSA (p-methylbenzene- sulfonic acid, 17.2 mg, 0.100 mmol, 1.0 equiv.) at room temperature. The reaction mixture was stirred for 3 h at room temperature and quenched by saturated aqueous NaHCO3. The resulting mixture was extracted with EtOAc (5 mL×3). The combined organic layers were washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [eluent: V(petroleum ether)∶V(EtOAc)=20∶1] afforded compound 8-D (5 mg, 13.7 μmol, 14% yield over 2 steps, dr=ca. 3∶2) as a colorless oil.
(4R,6aS,10aR)-3'-Isopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-3H-spiro[acephenanthryl-ene-4,1'-cyclopentan]-2'-en-3-one (8-D): 1H NMR (300 MHz, CDCl3) δ: 7.05~7.02 (m, 0.6H, major), 6.91~6.88 (m, 0.4H, minor), 5.94 (d, J=10.2 Hz, 0.17H), 5.20~5.18 (m, 0.6H, major), 5.08 (s, 0.6H, major), 5.05 (s, 0.4H, minor), 4.81~4.79 (m, 0.4H, minor), 3.15~3.09 (m, 0.6H, major), 2.88~2.77 (m, 0.66H), 2.55~2.43 (m, 1H), 2.38~2.14 (m, 4H), 2.12~2.00 (m, 1.42H), 1.87~1.76 (m, 1.63H), 1.71~1.51 (m, 3H), 1.51~1.41 (m, 1H), 1.30~1.15 (m, 1H), 1.10~1.02 (m, 6H), 1.00~0.95 (m, 4H), 0.92~0.87 (m, 6H); 13C NMR (major isomer, 101 MHz, CDCl3) δ: 201.1, 151.8, 148.6, 148.5, 143.6, 131.3, 131.2, 130.7, 128.7, 65.7, 65.6, 54.0, 52.6, 49.7, 47.4 (×3), 41.8, 37.0, 35.9, 34.2, 34.1, 33.8, 33.0, 29.9, 25.3, 22.3, 22.0, 21.7, 21.6, 19.8; 13C NMR (minor isomer, 101 MHz, CDCl3) δ: 200.1, 152.9, 147.6, 147.5, 144.1, 132.8, 132.7, 128.2, 125.4, 65.1 (×2), 58.6, 56.7, 54.7, 48.4 (×3), 42.4, 39.3, 39.2, 37.8, 37.1, 34.0, 33.3, 31.9, 30.0, 25.9, 21.7, 21.6, 21.3, 18.7, 15.9; HRMS (ESI) calcd for C26H36DO [M+H]+ 366.2902, found 366.2898; HRMS (ESI) calcd for C26H35D2O [M+H]+ 367.2965, found 367.2956; HRMS (ESI) calcd for C26H34D3O [M+H]+ 368.3027, found 368.3010.
4.3 Cyclopropanation of 8a/b
According to our previously reported procedure,
[12] to a stirred solution of Et
2Zn (0.411 mL, 1.0 mol/L in hexane, 0.411 mmol, 1.5 equiv.) in dry DCM (2.0 mL) were added successively trifluoroacetic acid (TFA, 31 μL, 0.411 mmol, 1.5 equiv.) and CH
2I
2 (34 μL, 0.411 mmol, 1.5 equiv.) at 0 ℃ under N
2 atmosphere. The solution was stirred for 15 min at the same temperature. Then, the solution of
8a/b (100 mg, 0.274 mmol) in dry DCM (2.0 mL) was added dropwise. The mixture was stirred for 1 h at the same temperature and quenched with water (5 mL). The solution was extracted with DCM (5 mL×3). The combined organic layers were dried over Na
2SO
4, filtered, and concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [eluent:
V(petro- leum ether)∶
V(Et
2O)=80∶1] afforded compound
10 (34 mg, 0.090 mmol, 33% yield) as a colorless oil and compound
8a (54 mg, 0.145 mmol, 53% yield) as a colorless oil.
(1'S,3aR,3a1S,4S,5'R,6aS,10aR,10bR)-5'-Isopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-3H-spiro[acephenanthrylene-4,2'-bicyclo[3.1.0]hexan]-3-one (10): $[\alpha]_{\mathrm{D}}^{20}$+93 (c 0.30, CHCl3); 1H NMR (400 MHz, CDCl3) δ: 7.02 (dd, J=10.2, 2.2 Hz, 1H), 5.90 (dd, J=10.1, 3.3 Hz, 1H), 5.23 (s, 1H), 3.18 (dd, J=11.7, 9.0 Hz, 1H), 3.09 (d, J=8.7 Hz, 1H), 2.55 (d, J=17.3 Hz, 1H), 2.24 (dd, J=17.3, 13.5 Hz, 1H), 2.00 (dt, J=12.1, 2.8 Hz, 1H), 1.76~1.53 (m, 6H), 1.48~1.41 (m, 1H), 1.39~1.34 (m, 1H), 1.32~1.25 (m, 2H), 1.22~1.11 (m, 1H), 1.02 (s, 3H), 1.00~0.89 (m, 14H), 0.89~0.80 (m, 1H), 0.35~0.30 (m, 1H); 13C NMR (75 MHz, CDCl3) δ: 200.5, 149.1, 142.3, 131.0, 130.0, 60.3, 52.1, 52.0, 49.5, 47.6, 41.7, 37.0, 36.0, 34.1, 34.0, 33.8, 32.6, 32.0, 30.4, 27.0, 25.3, 22.3, 22.0, 20.3, 19.9, 19.8, 12.0; HRMS (ESI) calcd for C27H39O [M+H]+ 379.2995, found 379.2987.
(3aS,3a1R,4R,6aS,10aR,10bS)-3'-Isopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-3H-spiro[acephenanthrylene-4,1'-cyclopentan]-2'-en-3-one(8a): $[\alpha]_{\mathrm{D}}^{20}$-113 (c 0.22, CHCl3); 1H NMR (300 MHz, CDCl3) δ: 6.90 (dd, J=10.1, 2.0 Hz, 1H), 5.93 (dd, J=10.1, 3.0 Hz, 1H), 5.05 (s, 1H), 4.81~4.79 (m, 1H), 2.89~2.80 (m, 2H), 2.46 (dd, J=12.7, 3.5 Hz, 1H), 2.38~2.14 (m, 4H), 2.12~2.00 (m, 2H), 1.87~1.76 (m, 2H), 1.73~1.62 (m, 1H), 1.56~1.41 (m, 2H), 1.26~1.16 (m, 1H), 1.12~1.02 (m, 2H), 0.98 (d, J=6.8 Hz, 6H), 0.95 (s, 3H), 0.91 (s, 3H), 0.89 (s, 3H); 13C NMR (75 MHz, CDCl3) δ: 200.1, 152.8, 147.7, 144.0, 132.6, 128.1, 125.4, 65.1, 58.5, 56.6, 54.6, 48.3, 42.3, 39.2, 37.7, 37.0, 34.0, 33.2, 31.9, 30.0, 25.9, 21.7, 21.5, 21.3, 18.6, 15.9; HRMS (ESI) calcd for C26H37O [M+H]+ 365.2839, found 365.2835.
4.4 Synthesis of (1'S,3aS,3a1R,4S,5'R,6aS,10aS, 10bR)-3,5'-diisopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-1H-spiro[acephenan-thrylene-4,2'-bicyclo[3.1.0]hexan]-1-one (11)
According to our previously reported procedure,
[12] to a stirred solution of
10 (12 mg, 31.7 μmol) in dry THF (2 mL) was added
i-PrLi (0.16 mL, 1.0 mol/L in hexane, 0.159 mmol, 5.0 equiv.) dropwise at -78 ℃ under N
2 atmosphere. The resulting solution was stirred for 3 h at -78 ℃ and then raised to 0 ℃. The reaction mixture was quenched by water (5 mL) at 0 ℃. The mixture was extracted with EtOAc (5 mL×3). The combined organic layers were washed with brine and dried over anhydrous Na
2SO
4, filtered, and then concentrated under reduced pressure to afford crude product which was used without purification.
To a stirred solution of the above crude product in DCM (2 mL) was added PCC (13.7 mg, 63.4 μmol, 2.0 equiv.) at 0 ℃. The reaction mixture was stirred for 24 h at room temperature and diluted with water (5 mL). The mixture was extracted with DCM (5 mL×3). The combined organic layers were dried over anhydrous Na2SO4, filtered, and then concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [eluent: V(petroleum ether)∶V(DCM)=5∶1] afforded compound 11 (9.6 mg, 22.8 μmol, 72% yield over 2 steps) as a white solid. m.p. 120~121 ℃; $[\alpha]_{\mathrm{D}}^{20}$-14 (c 0.10, CHCl3); 1H NMR (500 MHz, CDCl3) δ: 5.78 (s, 1H), 5.21 (s, 1H), 3.21 (dd, J=14.0, 7.5 Hz, 1H), 3.09 (d, J=7.6 Hz, 1H), 2.62 (sept, J=6.8 Hz, 1H), 2.58~2.53 (m, 1H), 2.54~2.48 (m, 1H), 2.19 (dd, J=17.3, 13.7 Hz, 1H), 1.95 (d, J=14.0 Hz, 1H), 1.71~1.61 (m, 2H), 1.65 (dd, J=13.7, 4.3 Hz, 1H), 1.58~1.53 (m, 1H), 1.53~1.49 (m, 1H), 1.46~1.40 (m, 2H), 1.19~1.03 (m, 4H), 1.13 (s, 3H), 1.10 (d, J=7.1 Hz, 3H), 1.09 (d, J=7.1 Hz, 3H), 0.99~0.96 (m, 1H), 0.98 (d, J=6.8 Hz, 3H), 0.91 (d, J=6.8 Hz, 3H), 0.90 (s, 3H), 0.89 (s, 3H), 0.43~0.39 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 198.6, 168.9, 144.3, 129.8, 125.8, 60.7, 58.9, 49.7, 48.0, 46.5, 42.0, 38.6, 34.6, 34.3, 34.0, 33.4, 33.1, 32.6, 31.7, 31.4, 27.1, 25.1, 23.8, 22.2, 21.1, 20.3, 20.2, 19.9 (×2), 13.5; HRMS (ESI) calcd for C30H45O [M+H]+ 421.3465, found 421.3457.
4.5 Synthesis of (E/Z)-1-(2,4-dinitrophenyl)-2- ((3aS,3a1R,4R,6aS,10aR,10bS)-3'-isopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-3H-spiro[acephenanthrylene-4,1'-cyclopentan]-2'-en-3-ylidene)hydrazine (13)
To a stirred solution of recovered 8a (18.2 mg, 0.050 mmol) in MeOH (2 mL) were added 2,4-dinitrophenyl- hydrazine 12 (49.5 mg, 0.250 mmol, 5.0 equiv.) and con. H2SO4 (9.8 mg, 0.10 0 mmol, 2.0 equiv.) at 0 ℃. The resulting solution was raised to 50 ℃ and stirred for 18 h at the same temperature. The reaction was quenched by the addition of water (5 mL). The mixture was extracted with EtOAc (5 mL×3), and the combined organic layers were washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [V(petroleum ether)∶V(Et2O)=40∶1] afforded compound 13 as a E/Z mixture with ca. 10∶1 ratio (17.4 mg, 0.032 mmol, 64% yield) as a reddish brown solid. 1H NMR for major isomer (500 MHz, CDCl3) δ: 11.40 (s, 1H), 9.12 (d, J=2.5 Hz, 1H), 8.27 (dd, J=9.6, 2.4 Hz, 1H), 7.87 (d, J=9.6 Hz, 1H), 6.67 (dd, J=10.1, 2.2 Hz, 1H), 6.55 (dd, J=10.2, 2.9 Hz, 1H), 5.10 (s, 1H), 4.73 (s, 1H), 3.23 (d, J=8.9 Hz, 1H), 2.63~2.56 (m, 1H), 2.48 (dd, J=12.6, 3.3 Hz, 1H), 2.40~2.15 (m, 5H), 2.07 (t, J=12.4 Hz, 1H), 1.90~1.83 (m, 1H), 1.77~1.67 (m, 1H), 1.66 (d, J=13.7 Hz, 1H), 1.58~1.50 (m, 1H), 1.48~1.42 (m, 1H), 1.25~1.17 (m, 1H), 1.09 (dd, J=12.5, 3.4 Hz, 1H), 1.08~0.98 (m, 1H), 0.96 (s, 3H), 0.92 (s, 3H), 0.90 (s, 3H), 0.84 (d, J=6.8 Hz, 3H), 0.83 (d, J=6.8 Hz, 3H); 13C NMR for major isomer (126 MHz, CDCl3) δ: 153.3, 151.7, 144.9, 143.9, 143.5, 137.7, 130.0, 129.1, 128.2, 126.9, 123.8, 118.8, 116.4, 66.4, 58.5, 55.2, 51.3, 47.5, 42.3, 39.0, 37.9, 37.0, 34.1, 33.3, 32.0, 30.1, 25.9, 21.7, 21.7, 21.4, 18.6, 15.7; HRMS (ESI) calcd for C32H39N4O4 [M-H]- 543.2977, found 543.2980.
4.6 Cyclopropanation of 15a/b
According to our previously reported procedure,
[12] to a stirred solution of Et
2Zn (0.44 mL, 1.0 mol/L in hexane, 0.440 mmol, 2.5 equiv.) in dry DCM (2.0 mL) were added successively TFA (33 μL, 0.440 mmol, 2.5 equiv.) and CH
2I
2 (37 μL, 0.440 mmol, 2.5 equiv.) at 0 ℃ under N
2 atmosphere. The solution was stirred for 15 min at the same temperature. Then, the solution of
15a/b (64 mg, 0.176 mmol) in dry DCM (2.0 mL) was added dropwise. The mixture was stirred for 1 h at the same temperature and quenched with water (5 mL). The solution was extracted with DCM (5 mL×3). The combined organic layers were dried over Na
2SO
4, filtered, and concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [eluent:
V(petroleum ether)∶
V(Et
2O)=80∶1] afforded compound
16 (33 mg, 0.087 mmol, 49% yield) as a colorless oil and compound
15b (23 mg, 0.063 mmol, 36% yield) as a colorless oil.
(1'S,3aS,3a1R,4R,6aS,10aR,10bS)-5'-Isopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-3H-spiro[acephenanthrylene-4,2'-bicyclo[3.1.0]hexan]-3-one(16): $[\alpha]_{\mathrm{D}}^{20}$-108 (c 0.30, CHCl3); 1H NMR (300 MHz, CDCl3) δ: 6.87 (dd, J=10.1, 1.8 Hz, 1H), 5.96 (dd, J=10.1, 2.9 Hz, 1H), 5.20 (s, 1H), 3.13 (d, J=9.0 Hz, 1H), 2.89 (dd, J=11.4, 9.3 Hz, 1H), 2.44 (dd, J=12.6, 3.4 Hz, 1H), 2.04 (t, J=12.5 Hz, 1H), 1.85~1.80 (m, 1H), 1.76~1.53 (m, 5H), 1.48~1.34 (m, 3H), 1.22~1.17 (m, 1H), 1.06~0.88 (m, 20H), 0.33 (dd, J=7.4, 5.3 Hz, 1H); 13C NMR (75 MHz, CDCl3) δ: 200.4, 148.0, 143.1, 132.7, 129.7, 59.4, 58.5, 54.2, 53.4, 48.6, 42.3, 37.7, 37.0, 34.0, 33.9, 33.3, 32.7, 32.3, 30.4, 27.0, 25.8, 21.3, 20.3, 19.9, 18.6, 15.8, 12.1; HRMS (ESI) calcd for C27H39O [M+H]+379.2995, found 379.2987.
(3aR,3a1S,4S,6aS,10aR,10bR)-3'-Isopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-3H-spiro[acephenanthrylene-4,1'-cyclopentan]-2'-en-3-one (15b): $[\alpha]_{\mathrm{D}}^{20}$+179 (c 0.10, CHCl3); 1H NMR (400 MHz, CDCl3) δ: 7.02 (dd, J=10.2, 2.2 Hz, 1H), 5.88 (dd, J=10.2, 3.3 Hz, 1H), 5.08 (s, 1H), 4.78~4.75 (m, 1H), 3.14 (dd, J=11.5, 8.7 Hz, 1H), 2.83 (d, J=8.7 Hz, 1H), 2.58~2.50 (m, 1H), 2.34~2.14 (m, 5H), 2.12~2.01 (m, 2H), 1.73~1.65 (m, 3H), 1.58~1.50 (m, 1H), 1.49~1.41 (m, 1H), 1.32~1.25 (m, 1H), 1.19~1.13 (m, 1H), 1.07 (s, 3H), 0.96 (d, J=6.8 Hz, 6H), 0.92 (s, 3H), 0.91 (s, 3H); 13C NMR (75 MHz, CDCl3) δ: 200.1, 153.0, 148.7, 143.5, 130.9, 128.7, 125.0, 66.0, 55.2, 52.2, 49.7, 47.1, 41.7, 39.4, 37.1, 35.6, 34.0, 33.8, 31.8, 30.0, 25.2, 22.2, 22.0, 21.7, 21.5, 19.8; HRMS (ESI) calcd for C26H37O [M+H]+ 365.2839, found 365.2831.
4.7 Synthesis of (1'S,3aR,3a1S,4R,5'S,6aS,10aS, 10bS)-3,5'-diisopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-1H-spiro[acephenanth-rylene-4,2'-bicyclo[3.1.0]hexan]-1-one (17)
According to our previously reported procedure,
[12] to a stirred solution of
16 (42 mg, 0.115 mmol) in dry THF (2 mL) was added
i-PrLi (0.58 mL, 1.0 mol/L in hexane, 0.575 mmol, 5.0 equiv.) dropwise at -78 ℃ under N
2 atmosphere. The resulting solution was stirred for 3 h at -78 ℃ and then raised slowly to 0 ℃. The reaction mixture was quenched by the addition of water (5 mL) at 0 ℃. The mixture was extracted with EtOAc (10 mL×3). The combined organic layers were washed with brine, dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure to afford the crude product which was used without purification.
To a stirred solution of the above-obtained crude product in DCM (2 mL) was added PCC (49.6 mg, 0.230 mmol, 2.0 equiv.) at 0 ℃. The reaction mixture was stirred for 24 h at room temperature and diluted with water (5 mL). The mixture was extracted with DCM (10 mL×3). The combined organic layers were dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. Purification of the residue by column chromatography [eluent: V(petroleum ether)∶V(DCM)=5∶1] afforded compound 17 (32.0 mg, 0.076 mmol, 66% yield over 2 steps) as a white solid. m.p. 134~135 ℃; $[\alpha]_{\mathrm{D}}^{20}$+2.5 (c 0.40, CHCl3); 1H NMR (300 MHz, CDCl3) δ: 5.75 (s, 1H), 5.17 (s, 1H), 3.17 (d, J=7.6 Hz, 1H), 2.99 (dd, J=13.3, 7.7 Hz, 1H), 2.82~2.77 (m, 1H), 2.64 (sept, J=6.8 Hz, 1H), 2.37 (dd, J=12.3, 3.2 Hz, 1H), 1.99 (t, J=12.4 Hz, 1H), 1.96 (d, J=13.3 Hz, 1H), 1.75~1.68 (m, 1H), 1.66~1.57 (m, 2H), 1.46 (sept, J=6.9 Hz, 1H), 1.42~1.36 (m, 2H), 1.32~1.24 (m, 1H), 1.22~1.10 (m, 2H), 1.09 (d, J=6.8 Hz, 3H), 1.08 (d, J=6.8 Hz, 3H), 1.07 (s, 3H), 0.99 (d, J=6.8 Hz, 3H), 0.99~0.96 (m, 1H), 0.94~0.86 (m, 2H), 0.91 (d, J=6.8 Hz, 3H), 0.90 (s, 3H), 0.88 (s, 3H), 0.48~0.41 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 200.5, 167.6, 144.9, 129.5, 126.4, 60.7, 59.2, 59.1, 50.3, 47.8, 42.4, 38.4, 38.1, 34.2, 33.8, 33.2, 32.6, 32.4, 31.6, 31.2, 27.2, 25.9, 23.9, 21.9, 20.3, 20.2, 20.0, 18.7, 14.5, 13.7; HRMS (ESI) calcd for C30H45O [M+H]+ 421.3465, found 421.3457.
4.8 Synthesis of (E/Z)-1-(2,4-dinitrophenyl)-2- ((3aR,3a1S,4S,6aS,10aR,10bR)-3'-isopropyl-7,7,10a-trimethyl-3a,3a1,6,6a,7,8,9,10,10a,10b-decahydro-3H-spiro[acephenanthrylene-4,1'-cyclopentan]-2'-en-3-ylidene)hydrazine (18)
To a stirred solution of recovered 15b (18.2 mg, 0.050 mmol) in MeOH (2 mL) were added 2,4-dinitrophenyl- hydrazine 12 (49.5 mg, 0.250 mmol, 5.0 equiv.) and con. H2SO4 (9.8 mg, 0.100 mmol, 2.0 equiv.) at 0 ℃. The resulting solution was raised to 50 ℃ and stirred for 18 h at the same temperature. The reaction mixture was quenched by water (5 mL). The mixture was extracted with EtOAc (5 mL×3), and the combined organic layers were washed with brine, dried over anhydrous Na2SO4, filtered, and then concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [V(petro- leum ether)∶V(Et2O)=40∶1] afforded compound 18 as a ca. 10∶1 E/Z mixture (16.9 mg, 0.031 mmol, 62% yield) as a reddish brown solid. 1H NMR for major isomer (500 MHz, CDCl3) δ: 11.43 (s, 1H), 9.12 (d, J=2.4 Hz, 1H), 8.28 (dd, J=9.6, 2.3 Hz, 1H), 7.89 (d, J=9.6 Hz, 1H), 6.84 (dd, J=10.2, 2.3 Hz, 1H), 6.48 (dd, J=10.2, 3.1 Hz, 1H), 5.13 (s, 1H), 4.70 (s, 1H), 3.17 (d, J=8.6 Hz, 1H), 2.88 (dd, J=11.2, 9.1 Hz, 1H), 2.59~2.53 (m, 1H), 2.40~2.16 (m, 6H), 1.93 (d, J=12.1 Hz, 1H), 1.74~1.66 (m, 3H), 1.64~1.56 (m, 1H), 1.49~1.44 (m, 1H), 1.34~1.26 (m, 1H), 1.22~1.16 (m, 1H), 1.07(s, 3H), 0.93 (s, 3H), 0.92 (s, 3H), 0.82 (d, J=7.3 Hz, 3H), 0.81 (d, J=7.3 Hz, 3H); 13C NMR for major isomer (126 MHz, CDCl3) δ: 153.3, 152.1, 144.9, 144.5, 143.1, 137.7, 130.0, 129.0, 128.9, 126.5, 123.8, 116.9, 116.4, 67.2, 53.1, 50.3, 49.7, 46.4, 41.8, 39.2, 36.9, 36.5, 34.0, 33.9, 32.0, 30.0, 25.6, 22.6, 22.1, 21.7, 21.7, 19.8; HRMS (ESI) calcd for C32H39N4O4 [M-H]- 543.2977, found 543.2978.
4.9 Synthesis of (1S,1a'R,2a'R,2a1'S,5a'R,5b'R, 9a'S,10a'S)-3-isopropyl-5b',9',9'-trimethyl-1',1a',2a',2a1',5a',5b',6',7',8',9',9a',10'-dodecahydro-3'H-spiro-[cyclopentane-1,2'-cyclopropa[f]acephenanthrylen]-2-en-3'-one (19)
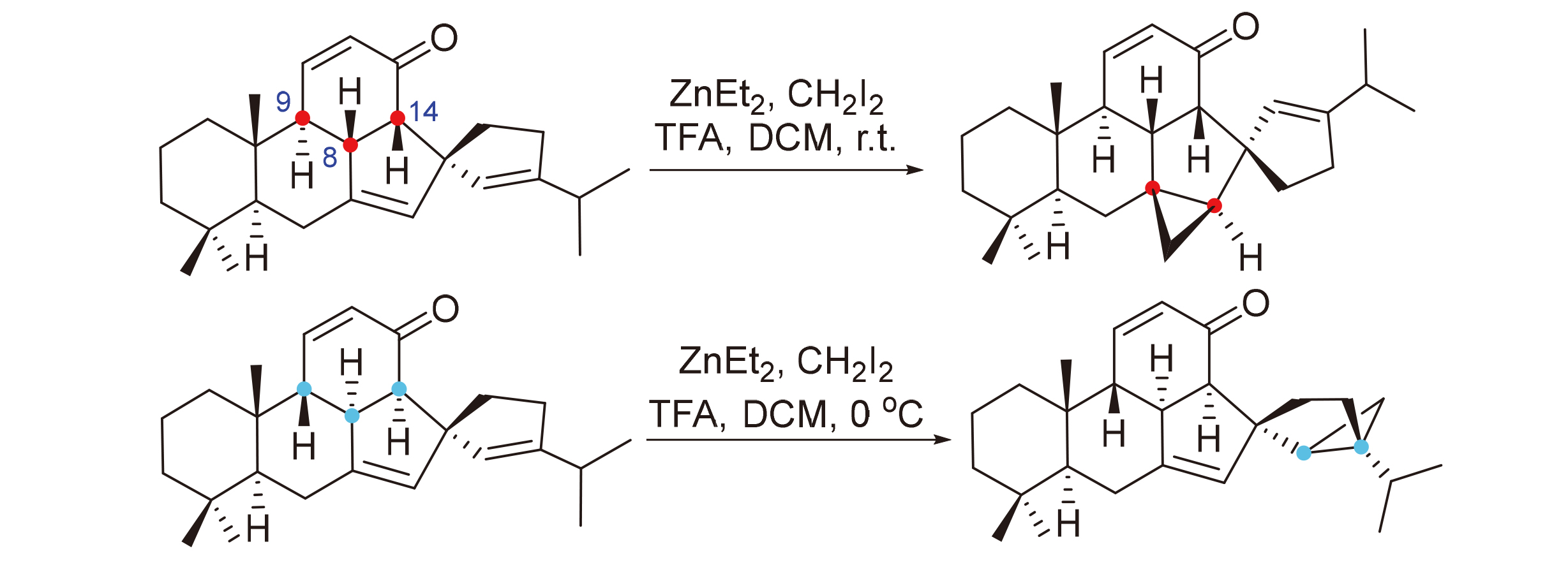
To a stirred solution of Et2Zn (0.288 mL, 1.0 mol/L in hexane, 0.288 mmol, 8.0 equiv.) in dry DCM (1 mL) were added successively TFA (21 μL, 0.288 mmol, 8.0 equiv.) and CH2I2 (48 μL, 0.576 mmol, 16.0 equiv.) at 0 ℃ under N2 atmosphere. The resulting solution was stirred for 15 min at the same temperature. Then, the solution of 15b (13.0 mg, 0.036 mmol) in dry DCM (1 mL) was added dropwise. The resulting solution was stirred for 4 h at room temperature and quenched with water (10 ml). The solution was extracted with DCM (5 mL×3), and the combined organic layers were dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [eluent: V(petroleum ether)∶V(Et2O)=80∶1] afforded compound 19 (11.0 mg, 0.029 mmol, 81% yield) as a white solid. 1H NMR (300 MHz, CDCl3) δ: 6.94 (dd, J=10.3, 1.7 Hz, 1H), 5.88 (dd, J=10.3, 3.1 Hz, 1H), 5.03 (s, 1H), 2.45 (dd, J=11.7, 7.6 Hz, 1H), 2.35~2.30 (m, 1H), 2.28~2.21 (m, 2H), 2.19~2.13 (m, 3H), 2.08~2.00 (m,1H), 1.76~1.68 (m, 2H), 1.61~1.55 (m, 2H), 1.51~1.43 (m, 3H), 1.25~1.21 (m, 2H), 1.17 (s, 3H), 1.11 (dd, J=8.0, 3.8 Hz, 1H), 0.95 (d, J=6.9 Hz, 3H), 0.94 (d, J=6.9 Hz, 3H), 0.91 (s, 3H), 0.86 (s, 3H), 0.68~0.64 (m, 1H), 0.40 (dd, J=8.0, 5.3 Hz, 1H); 13C NMR (101 MHz, CDCl3) δ: 200.1, 153.6, 150.2, 130.8, 126.2, 60.4, 53.8, 50.2, 47.9, 42.7, 41.0, 36.9, 36.7, 34.7, 33.6, 33.5, 33.0, 31.7, 31.3, 30.0, 26.1, 22.7, 22.3, 21.7, 21.5, 19.8, 16.7; HRMS (APCI) calcd for C27H39O [M+H]+ 379.2995, found 379.2989.
4.10 Synthesis of (1R,1a'S,2a'S,2a1'R,5a'S,5b'R, 9a'S,10a'R)-3-isopropyl-5b',9',9'-trimethyl-1',1a',2a',2a1',5a',5b',6',7',8',9',9a',10'-dodecahydro-3'H-spiro-[cyclopentane-1,2'-cyclopropa[f]acephenanthrylen]-2-en-3'-one (20)
To a stirred solution of Et2Zn (0.328 mL, 1.0 mol/L in hexane, 0.328 mmol, 8.0 equiv.) in dry DCM (1 mL) were added successively TFA (24 μL, 0.328 mmol, 8.0 equiv.) and CH2I2 (55 μL, 0.656 mmol, 16.0 equiv.) at 0 ℃ under N2 atmosphere. The resulting solution was stirred for 15 min at the same temperature. Then, the solution of 8a (15.0 mg, 0.041 mmol) in dry DCM (1 mL) was added dropwise. The resulting solution was stirred for 4 h at room temperature, quenched with water (10 mL) and then extracted with DCM (5 mL×3). The combined organic layers were dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. Purification of the residue by column chromatography on silica gel [eluent: V(petroleum ether)∶V(Et2O)=80∶1] afforded compound 20 (12.6 mg, 0.033 mmol, 81% yield) as a white solid. $[\alpha]_{\mathrm{D}}^{20}$-72 (c 0.50, CHCl3); 1H NMR (300 MHz, CDCl3) δ: 7.01 (dd, J=10.2, 2.1 Hz, 1H), 5.86 (dd, J=10.2, 3.0 Hz, 1H), 5.16 (s, 1H), 2.40~2.33 (m, 2H), 2.31~2.19 (m, 2H), 2.15~2.05 (m, 3H), 1.96 (t, J=13.3 Hz, 1H), 1.93~1.87 (m, 1H), 1.77~1.60 (m, 1H), 1.55~1.51 (m, 1H), 1.48~1.42 (m, 1H), 1.30~1.22 (m, 2H), 1.20~1.12 (m, 2H), 1.10~1.03 (m, 2H), 0.97 (d, J=6.8 Hz, 6H), 0.92 (s, 3H), 0.85 (s, 3H), 0.83 (s, 3H), 0.58 (dd, J=5.2, 3.2 Hz, 1H), 0.25 (dd, J=7.9, 5.2 Hz, 1H); 13C NMR (75 MHz, CDCl3) δ: 200.5, 154.1, 150.3, 131.6, 126.3, 59.6, 55.3, 53.8, 51.7, 45.0, 42.4, 38.1, 38.0, 35.7, 35.2, 33.3, 33.1, 30.9, 30.2, 29.8, 28.0, 21.8, 21.7, 21.2, 18.8, 15.4, 12.5; HRMS (APCI) calcd for C27H39O [M+H]+ 379.2995, found 379.2989.
Supporting Information Copies of
1H and
13C NMR, and other 2D NMR spectra for the compounds
E-7-D,
8-D,
8a,
10,
11,
13,
15b, and
16~
20. The Supporting Information is available free of charge via the Internet at
http:// sioc-journal.cn/.