

Chinese Journal of Organic Chemistry >
Recent Advances of C—H Phosphonylation of (Hetero)arenes under Electrochemical Conditions
Received date: 2025-06-26
Revised date: 2025-08-08
Online published: 2025-10-10
Supported by
Nanyang Normal University(2025STP013)
Nanyang Science and Technology Major Project(24KJGG015)
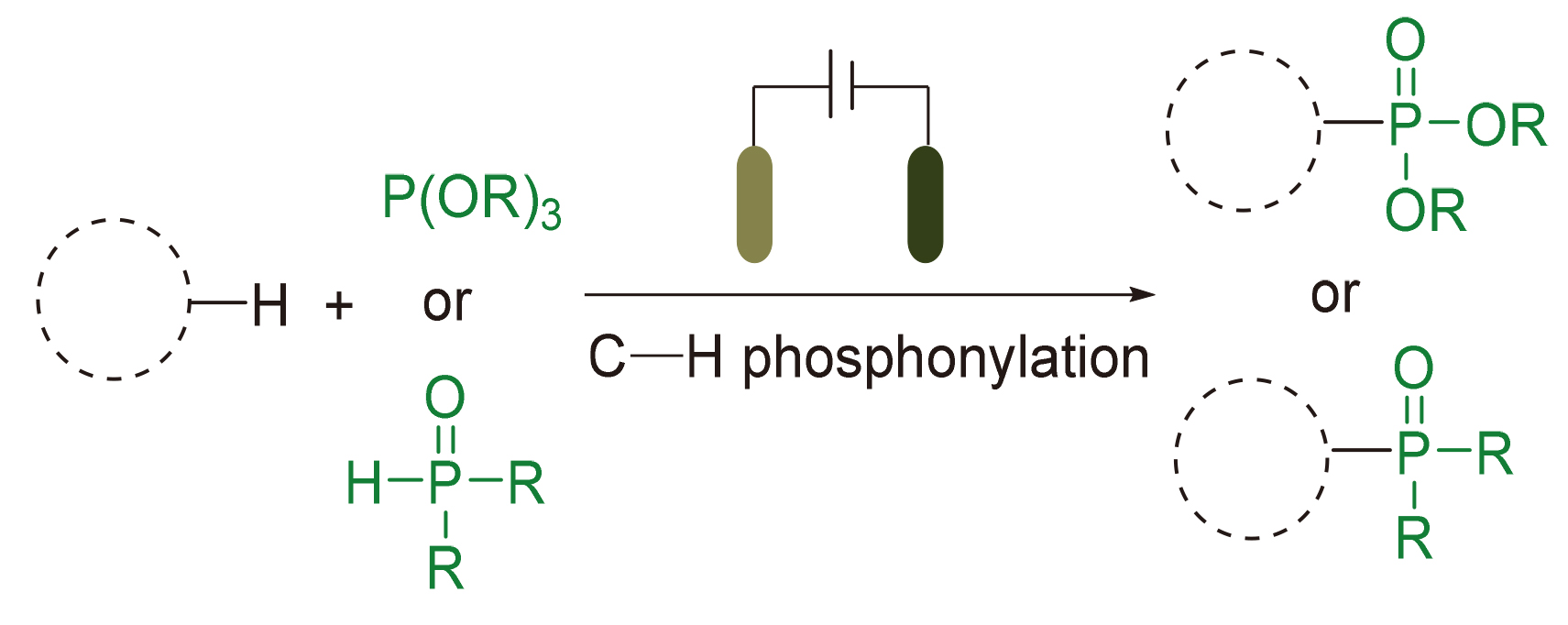
Phosphonylated (hetero)arenes, as an important class of organophosphorus compounds, exhibit broad application value in fields such as organic catalysis, medicinal chemistry, and materials science. Among various synthetic approaches, the C—H phosphonylation of (hetero)arenes stands out as one of the most efficient strategies for synthesizing these compounds due to its atom economy and step simplicity. In recent years, with the rapid development of organic electrochemistry, significant breakthroughs have been achieved in electrochemically driven C—H phosphonylation. Compared with traditional methods, the electrochemical strategy offers distinct advantages: (1) it replaces chemical oxidants with electrons, improving atom economy, and (2) it demonstrates excellent functional group compatibility under mild conditions. This review summarizes the latest advances in the electrochemical C—H phosphonylation of (hetero)arenes, with a focus on reaction mechanisms and substrate scope. For clarity, the discussed reaction systems are categorized into two major classes based on the electrolysis mode (direct electrolysis vs. indirect electrolysis).
Ruoyu Liu , Bingyu Liang , Shuyang Chen , Hongxian Sun , Yibing Sun , Yong Ye . Recent Advances of C—H Phosphonylation of (Hetero)arenes under Electrochemical Conditions[J]. Chinese Journal of Organic Chemistry, 2025 , 45(12) : 4290 -4297 . DOI: 10.6023/cjoc202506033
| [1] |
(a)
(b)
|
| [2] |
(a)
(b)
(c)
|
| [3] |
(a)
(b)
(c)
|
| [4] |
(a)
(王振华, 马聪, 方萍, 徐海超, 梅天胜, 化学学报, 2022, 80, 1115.)
(b)
(c)
(d)
(李永梅, 孙亮博, 徐坤, 曾程初, 有机化学, 2025, 45, 668.)
(e)
(朱子乐, 李鹏飞, 仇友爱, 有机化学, 2024, 44, 871.)
|
| [5] |
(a)
(b)
(c)
(d)
(周兰, 何红, 杨德巧, 侯中伟, 王磊, 有机化学, 2024, 44, 981.)
(e)
(张浩南, 徐坤, 有机化学, 2025, 45, 722.)
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
/
| 〈 |
|
〉 |