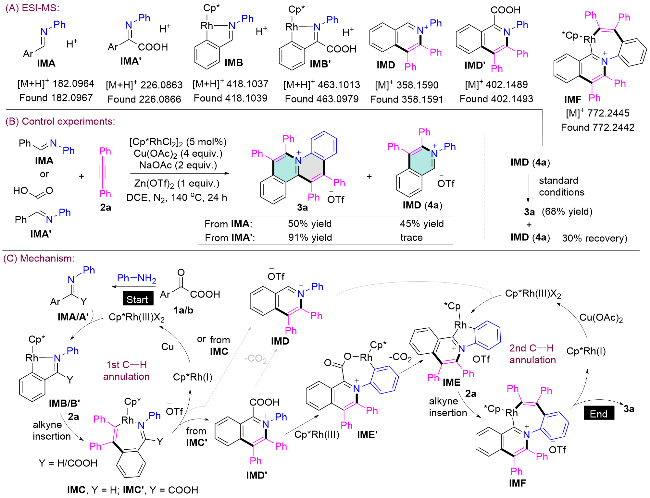
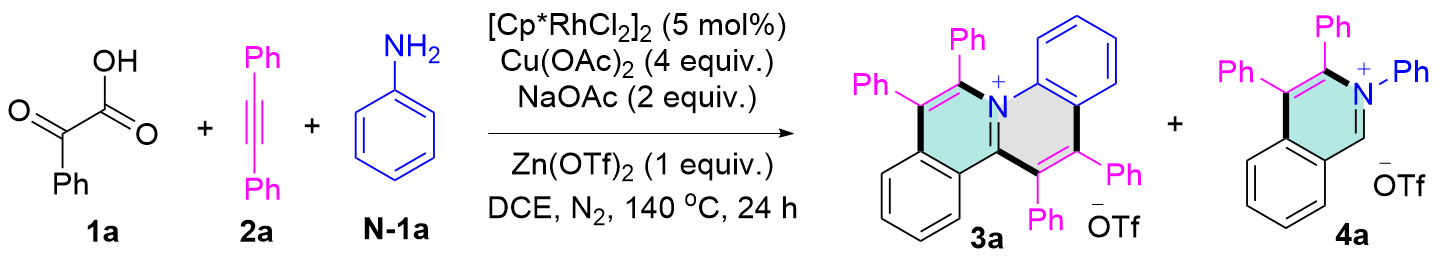向干燥的25 mL反应管(内置磁力搅拌子)中依次加入α-氧代羧酸1 (0.1 mmol)、芳基胺N-1 (0.15 mmol, 1.5 equiv.)、炔烃2 (0.2 mmol, 2.0 equiv.)、[Cp*RhCl2]2 (3.1 mg, 5 mol%)、Cu(OAc)2 (72.6 mg, 0.4 mmol, 4 equiv.)、Zn(OTf)2 (36.4 mg, 0.1 mmol, 1 equiv.)、NaOAc (16.8 mg, 0.2 mmol, 2 equiv.)以及1,2-二氯乙烷(2 mL). 将反应混合物在氮气氛围中140 ℃搅拌反应24 h. 反应完成后, 减压蒸馏去除溶剂, 残余物通过硅胶柱层析纯化[洗脱剂: V(二氯甲烷)∶V(甲醇)=100∶1→50∶1]得到目标产物3.
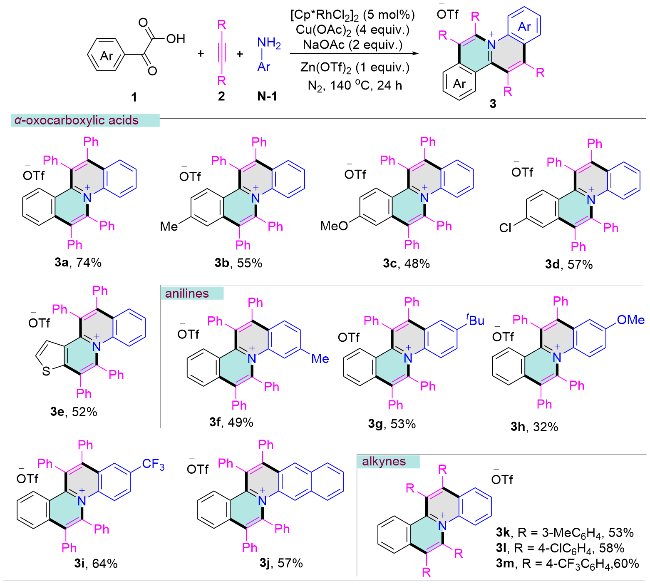
6,7,12,13-四苯基异喹啉并[2,1-
a]喹啉-5-鎓三氟甲磺酸盐(
3a): 黄棕色固体, 47 mg, 产率69%. m.p. >250 ℃ (lit.
[13] >250 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 8.04 (d,
J=9.0 Hz, 1H), 7.85 (t,
J=9.2 Hz, 1H),), 7.77 (dd,
J=8.5, 1.0 Hz, 1H), 7.73~7.69 (m, 1H), 7.50 (t,
J=8.0 Hz, 1H), 7.42~7.32 (m, 13H), 7.29~7.26 (m, 4H), 7.23 (dd,
J=8.0, 2 Hz, 2H), 7.14~7.09 (m, 3H).
9-甲基-6,7,12,13-四苯基异喹啉并[2,1-a]喹啉-5-鎓三氟甲磺酸盐(3b): 黄棕色固体, 35 mg, 产率50%. m.p.>250 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.01 (d, J=8.5 Hz, 1H), 7.80 (d, J=8.0 Hz, 1H), 7.72 (d, J=9.0 Hz, 1H), 7.51 (s, 1H), 7.48 (t, J=7.5 Hz, 1H), 7.39~7.32 (m, 13H), 7.28~7.27 (m, 3H), 7.20 (d, J=7.0 Hz, 2H), 7.13~7.09 (m, 4H), 2.40 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 152.6, 150.1, 146.6, 141.3, 138.6, 137.0, 136.8 (2C), 136.7, 135.9 (2C), 134.4, 134.3, 134.2, 131.6, 131.2, 130.8, 130.6, 130.0, 129.7, 129.3, 129.0, 128.9, 128.8, 128.7 (2C), 128.6, 128.3, 127.9, 127.6, 126.7, 125.9, 124.2, 22.4; HRMS (ESI) calcd for C42H30N [M-OTf]+548.2373, found 548.2377.
9-甲氧基-6,7,12,13-四苯基异喹啉并[2,1-
a]喹啉-5-鎓三氟甲磺酸盐(
3c): 黄色固体, 31 mg, 产率43%. m.p. >250 ℃ (lit.
[13] >250 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 7.95 (d,
J=8.5 Hz, 1H), 7.76~7.73 (m, 2H) 7.45 (t,
J=8.5 HZ, 1H), 7.40~7.27 (m, 16H), 7.21~7.20 (m, 2H) 7.14~7.09 (m, 3H), 7.03 (d,
J=3.0 Hz, 1H), 6.86 (dd,
J=9.5 Hz,
J=2.5 Hz, 1H), 3.75 (s, 3H);
13C NMR (125 MHz, CDCl
3)
δ: 164.4, 151.8, 149.9, 141.6, 138.9, 138.7, 136.6, 136.5, 134.5, 134.2, 133.8, 132.3, 131.6, 131.2, 130.7, 130.0, 129.6, 129.5, 129.4, 129.1, 128.8, 128.7, 128.5, 128.3, 127.9, 127.3, 125.9, 124.7, 120.6, 119.3, 107.8, 56.0.
9-氯-6,7,12,13-四苯基异喹啉并[2,1-a]喹啉-5-鎓三氟甲磺酸盐(3d): 橙色固体, 36 mg, 产率50%. m.p.>250 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.02 (d, J=9.0 Hz, 1H), 7.83 (d, J=8.5 Hz, 1H), 7.77 (d, J=9.5 Hz, 1H), 7.69 (d, J=2.0 Hz, 1H), 7.50 (t, J=7.8 Hz, 1H), 7.44~7.26 (m, 13H), 7.30~7.28 (m, 4H), 7.25 (s, 1H), 7.19 (dd, J=9.5 Hz, 2.0 Hz, 1H), 7.15~7.09 (m, 3H); 13C NMR (125 MHz, CDCl3) δ: 176.1, 172.5, 161.8, 153.5, 152.8, 136.9, 136.1, 134.4, 133.7, 132.7, 131.7, 131.6, 131.3 (2C), 131.0, 130.8 (2C), 130.0, 129.7, 129.4, 129.2, 129.1, 128.9, 128.8, 128.6, 128.4, 128.3, 128.0, 127.4, 126.4, 126.0; HRMS (ESI) calcd for C41H2735ClN [M-OTf]+568.1827, found 568.1834; HRMS (ESI) calcd for C41H27- 37ClN [M-OTf]+ 570.1803, found 570.1800.
4,5,11,12-四苯基噻吩并[3',
2':
3,
4]吡啶并[1,2-
a]喹 啉-10-鎓三氟甲磺酸盐(
3e): 黄色固体, 36 mg, 产率52%. m.p. 190~192 ℃ (lit.
[13] 189~191 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 8.03 (d,
J=9.0 Hz, 1H), 7.65 (d,
J=7.5 Hz, 1H), 7.48~7.43 (m, 4H), 7.39~7.29 (m, 15H), 7.25~7.15 (m, 4H), 6.25 (d,
J=6.0 Hz, 1H);
13C NMR (125 MHz, DMSO-
d6)
δ: 153.2, 149.1, 143.8, 141.8, 136.7, 136.0, 135.9, 135.1, 134.5, 133.4, 133.2, 132.8, 132.2, 131.1, 130.9, 129.6, 129.3 (2C), 129.2, 129.1, 129.0 (2C), 128.7, 128.5, 128.4, 128.1, 127.5, 127.0, 126.7, 125.8.
3-甲基-6,7,12,13-四苯基异喹啉并[2,1-a]喹啉-5-鎓三氟甲磺酸盐(3f): 黄棕色固体, 34 mg, 产率49%. m.p. 213~215 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.84 (d, J=7.5 Hz, 2H), 7.75 (d, J=8.0 Hz, 1H), 7.72~7.68 (m, 2H), 7.40~7.31 (m, 13H), 7.28~7.26 (m, 4H), 7.22 (d, J=7.0 Hz, 2H), 7.14~7.10 (m, 3H), 2.17 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 153.1, 149.7, 141.8, 140.9, 138.4, 136.9, 136.8, 136.7, 135.2, 134.4, 134.2 (2C), 134.1, 133.5, 131.6, 131.1, 130.8, 130.0, 129.9, 129.2, 128.9, 128.7, 128.6, 128.5, 128.4 (2C), 128.2, 127.6, 127.2, 126.0, 125.9, 21.6; HRMS (ESI) calcd for C42H30N [M-OTf]+548.2373, found 548.2378.
2-叔丁基-6,7,12,13-四苯基异喹啉并[2,1-a]喹啉-5-鎓三氟甲磺酸盐(3g): 橙黄色固体, 39.3 mg, 产率52%. m.p.>250 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.96 (d, J=9.0 Hz, 1H), 7.84 (d, J=9.0 Hz, 1H),7.78~7.77 (m, 2H), 7.70 (t, J=7.5 Hz, 1H), 7.40~7.36 (m, 12H), 7.30~7.25 (m, 7H, cover the solvent), 7.16~7.11 (m, 3H), 1.20 (s, 9H); 13C NMR (125 MHz, CDCl3) δ: 153.2, 152.6, 149.5, 141.0, 138.6, 137.1, 136.8, 135.3, 135.1, 134.6, 134.4, 134.2 (2C), 131.8, 131.2, 130.8, 130.0, 129.3, 129.1, 128.8, 128.7, 128.6, 128.5, 128.4, 128.3 (2C), 127.8, 127.3, 126.2, 125.6, 123.2, 35.2, 30.8; HRMS (ESI) calcd for C45H36N [M-OTf]+ 590.2843, found 590.2849.
2-三氟甲基-6,7,12,13-四苯基异喹啉并[2,1-a]喹啉- 5-鎓三氟甲磺酸盐(3h): 黄色固体, 48 mg, 产率64%. m.p.>250 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.22 (d, J=9.0 Hz, 1H), 8.09 (s, 1H), 7.88 (d, J=8.5 Hz, 1H), 7.81 (d, J=8.0 Hz, 1H), 7.75 (t, J=7.5 Hz, 1H), 7.46~7.37 (m, 13H), 7.32~7.28 (m, 6H), 7.18~7.12 (m, 3H); 13C NMR (125 MHz, CDCl3) δ: 152.4, 141.7, 138.5, 138.2, 138.0, 136.3, 136.2, 135.1, 134.0, 133.8, 131.7, 131.6, 131.5 (2C), 130.9, 130.4, 130.0, 129.5, 129.4 (q, JC—F=32.6 Hz), 129.4, 129.1, 129.0, 128.8, 128.6 (2C), 127.8, 127.4, 126.4 (q, JC—F=4.0 Hz), 126.2, 125.3, 124.9, 123.2 (q, JC—F=273.9 Hz); HRMS (ESI) calcd for C42H27F3N [M-OTf]+ 602.2091, found 602.2098.
2-甲氧基-6,7,12,13-四苯基异喹啉并[2,1-a]喹啉-5-鎓三氟甲磺酸盐(3i): 黄色固体, 23 mg, 产率32%. m.p. 143~145 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.01 (d, J=10.0 Hz, 1H), 7.83 (d, J=8.5 Hz, 1H), 7.75 (d, J=8.5 Hz, 1H), 7.69 (t, J=7.5 Hz, 1H), 7.41~7.35 (m, 12H), 7.30~7.25 (m, 6H), 7.16~7.14 (m, 3H), 7.10 (s, 1H), 6.96 (d, J=10.0 Hz, 1H), 3.73 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 159.2, 152.3, 148.3, 140.8, 138.6, 137.2, 137.0, 135.0, 134.7 (2C), 134.3, 133.9 (2C), 131.9, 131.7, 131.2, 131.1, 130.8, 129.9, 129.8, 129.3, 129.0, 128.7, 128.6, 128.5 (2C), 127.5, 127.3, 126.3, 120.4, 107.1, 55.9; HRMS (ESI) calcd for C42H30NO [M-OTf]+ 564.2322, found 564.2331.
5,6,14,15-四苯基苯并[g]异喹啉并[2,1-a]喹啉-13-鎓三氟甲磺酸盐(3j): 红色固体, 42 mg, 产率57%. m.p. 155~157 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.56 (s, 1H), 8.29 (s, 1H), 7.88~7.86 (m, 2H), 7.82 (d, J=8.5 Hz, 1H), 7.74 (t, J=6.8 HZ, 1H), 7.57~7.51 (m, 3H), 7.44~7.37 (m, 12H), 7.33 (d, J=6.5 Hz, 2H), 7.30~7.27 (m, 4H), 7.07~7.03 (m, 3H); 13C NMR (125 MHz, CDCl3) δ: 152.9, 151.5, 142.6, 138.9, 137.3, 136.6, 136.5, 135.0, 134.8, 134.2, 133.4, 132.3, 132.1, 131.9, 131.8, 131.4, 131.0, 130.3, 130.1, 129.8, 129.2, 129.0, 128.7 (2C), 128.6, 128.6, 128.5, 128.4 (2C), 128.2, 128.1, 127.2, 126.6, 126.2, 125.7; HRMS (ESI) calcd for C45H30N [M-OTf]+ 584.2373, found 584.2377.
6,7,12,13-四(间甲苯基)异喹啉并[2,1-a]喹啉-5-鎓三氟甲磺酸盐(3k): 黄色固体, 39 mg, 产率53%. m.p. 217~219 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.09 (d, J=9.0 Hz, 1H), 7.92 (d, J=8.5 Hz, 1H), 7.85 (d, J=8.5 Hz, 1H), 7.79 (d, J=8.5 Hz 1H), 7.72 (t, J=7.5 Hz, 1H), 7.52 (t, J=7.5 Hz, 1H), 7.36 (t, J=7.8 Hz, 1H), 7.31~7.28 (m, 3H), 7.19~7.07 (m, 10H), 7.01~6.94 (m, 4H), 2.32 (s, 6H), 2.23 (s, 3H), 2.11 (s, 3H); 13C NMR (125 MHz, CDCl3) δ: 153.2, 149.9, 149.2, 142.4, 141.2, 139.3, 139.1, 138.4, 137.4, 137.3, 136.9, 136.7, 135.6, 134.5 (2C, 134.4, 134.1, 131.8, 131.0, 130.2, 129.8 (2C), 129.5, 129.4, 129.2, 129.1, 129.0 (2C), 128.9, 128.5, 128.4, 128.1 (2C), 128.0, 127.8, 127.4, 126.2, 126.0, 122.1, 23.8, 21.5, 21.4, 21.2; HRMS (ESI) calcd for C45H36N [M-OTf]+590.2843, found 590.2847.
6,7,12,13-四(4-氯苯基)异喹啉并[2,1-a]喹啉-5-鎓三氟甲磺酸盐(3l): 黄色固体, 47.3 mg, 产率58%. m.p. 248~250 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.85~7.80 (m, 3H), 7.75 (d, J=4 Hz, 2H), 7.55 (t, J=8.0 Hz, 1H), 7.46~7.44 (m, 3H), 7.38~7.27 (m, 13H), 7.11 (d, J=7.5 Hz, 2H); 13C NMR (125 MHz, CDCl3) δ: 150.7, 136.9, 136.7, 136.5, 135.8, 135.6, 135.5, 135.4, 135.2, 134.9, 134.8, 134.2, 133.4, 133.1, 132.8, 132.6, 132.4, 131.6, 130.1, 129.9, 129.7, 129.3, 129.2, 129.0, 128.7, 128.1, 128.0 (2C), 127.2, 126.2, 125.7. HRMS (ESI) calcd for C41H2435Cl4N 672.0628, found 672.0633; HRMS (ESI) calcd for C41H2437Cl4N [M-OTf]+ 680.0532, found 680.0539.
6,7,12,13-四(4-三氟甲基苯基)异喹啉并[2,1-a]喹啉- 5-鎓三氟甲磺酸盐(3m): 黄棕色固体, 57 mg, 产率60%. m.p. 167~169 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.81~7.72 (m, 7H), 7.65~7.57 (m, 11H), 7.53 (d, J=8.0 Hz, 2H), 7.39~7.32 (m, 4H); 13C NMR (125 MHz, CDCl3) δ: 152.2, 150.5, 141.8 (2C), 140.4, 139.4, 137.8, 137.7, 136.8, 136.2, 135.2, 135.1, 133.9, 132.4, 132.3, 131.4, 131.2 (q, JC—F=32.5 Hz), 130.6, 130.1, 129.9, 129.7, 129.0, 127.9, 127.1, 126.3 (q, JC—F=3.8 Hz), 126.0, 125.8, 125.7 (3C), 125.5 (q, JC—F=3.4 Hz), 124.5, 123.7 (q, JC—F=270.8 Hz), 120.2; HRMS (ESI) calcd for C45H24- F12N [M-OTf]+ 806.1712, found 806.1721.
2,3,4-三苯基异喹啉-2-鎓三氟甲磺酸盐(
4a): 深灰色固体, 33 mg, 产率56%. m.p. 188~190 ℃ (lit.
[13] 188~190 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 9.95 (s, 1H), 8.69 (d,
J=5.5 Hz, 1H), 8.08 (t,
J=7.5 Hz, 1H), 7.99 (t,
J=7.5 Hz, 1H), 7.77 (d,
J=6.0 Hz, 1H), 7.53 (br s, 2H), 7.37~7.33 (m, 6H), 7.26 (br s, 2H), 7.07~7.03 (m, 5H).
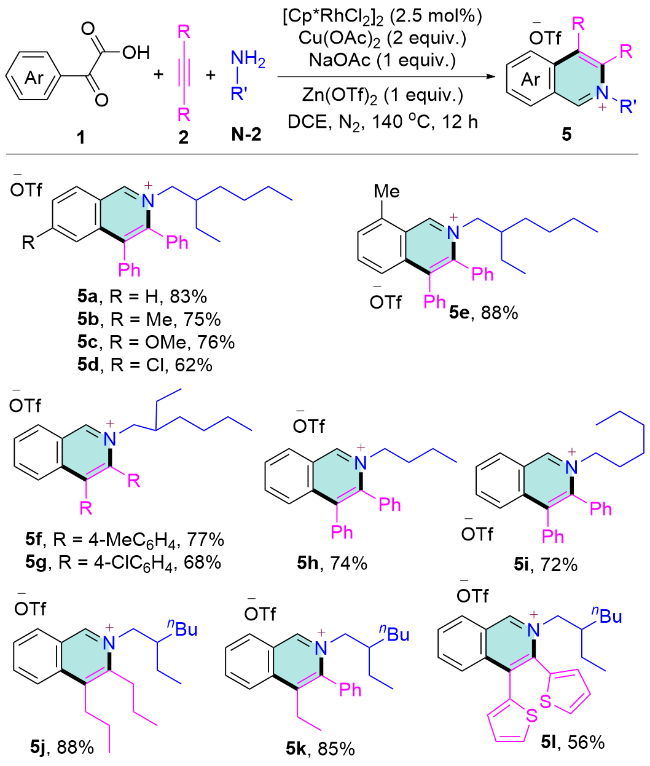
向干燥的25 mL反应管(内置磁力搅拌子)中依次加入α-氧代羧酸1 (0.1 mmol)、烷基胺N-2 (0.15 mmol, 1.5 equiv.)、炔烃2 (0.1 mmol, 1.0 equiv.)、[Cp*RhCl2]2 (1.6 mg, 2.5 mol%)、Cu(OAc)2 (36.3 mg, 0.2 mmol, 2 equiv.)、Zn(OTf)2 (36.4 mg, 0.1 mmol, 1 equiv.)、NaOAc (8.4 mg, 0.1 mmol, 1 equiv.)以及1,2-二氯乙烷(2 mL). 将反应混合物在氮气氛围下140 ℃搅拌反应12 h. 反应完成后, 减压蒸馏去除溶剂, 残余物通过硅胶柱层析纯化[洗脱剂: V(二氯甲烷)∶V(甲醇)=100∶1→80∶1]得到目标产物5.
2-(2-乙基己基)-3,4-二苯基异喹啉-2-鎓三氟甲磺酸盐(5a): 浅灰色固体, 45 mg, 产率83%. m.p. 144~146 ℃; 1H NMR (500 MHz, CDCl3) δ: 10.22 (s, 1H), 8.77 (d, J=8.5 Hz, 1H), 8.03 (t, J=7.5 Hz, 1H), 7.98 (t, J=7.5 Hz, 1H), 7.65 (d, J=8.5 Hz, 1H), 7.38~7.22 (m, 8H), 7.14~7.08 (m, 2H), 4.71~4.64 (m, 2H), 1.62~1.55 (m, 1H), 1.32~1.23 (m, 2H), 1.19~1.03 (m, 4H), 0.91~0.85 (m, 2H), 0.74 (t, J=7.5 Hz, 3H), 0.64 (t, J=7.5 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 151.9, 144.0, 139.7, 137.9, 137.5, 133.2, 132.0, 131.4, 130.9, 130.8, 130.3, 130.2, 128.9, 128.7, 128.6, 127.1, 126.4, 63.8, 40.4, 29.8, 28.1, 23.2, 22.7, 14.0, 10.4; HRMS (ESI) calcd for C29H32N [M-OTf]+ 394.2530, found 394.2533.
2-(2-乙基己基)-6-甲基-3,4-二苯基异喹啉-2-鎓三氟甲磺酸盐(5b): 浅灰色固体, 42 mg, 产率75%. m.p. 186~188 ℃; 1H NMR (500 MHz, CDCl3) δ: 10.08 (s, 1H), 8.64 (d, J=8.5 Hz, 1H), 7.78 (d, J=8.0 Hz, 1H) 7.37~7.29 (m, 7H), 7.25~7.20 (m, 2H), 7.12~7.05 (m, 2H ), 4.65~4.58 (m, 2H), 1.60~1.54 (m, 1H), 1.30~1.22 (m, 2H),1.18~1.01 (m, 4H), 0.90~0.85 (m, 2H), 0.73 (t, J=7.5 Hz, 3H), 0.63 (t, J=7.5 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 151.1, 149.9, 144.0, 138.7, 138.1, 133.6, 133.4, 131.7, 130.9, 130.8, 130.2, 128.8 (2C), 128.7, 128.6, 125.5, 125.3, 63.5, 40.3, 29.8, 28.1, 23.3, 23.2, 22.7, 13.9, 10.4; HRMS (ESI) calcd for C30H34N [M-OTf]+408.2686, found 408.2688.
2-(2-乙基己基)-6-甲氧基-3,4-二苯基异喹啉-2-鎓三氟甲磺酸盐(5c): 浅灰色固体, 44 mg, 产率76%. m.p. 131~133 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.97 (s, 1H), 8.65 (d, J=9.0 Hz, 1H), 7.53 (dd, J=9.0, 2.0 Hz, 1H), 7.36~7.30 (m, 6H), 7.26~7.19 (m, 2H), 7.13~7.06 (m, 2H ), 6.78 (d, J=2.5 Hz, 1H), 4.58~4.50 (m, 2H), 3.81 (s, 3H), 1.57~1.51 (m, 1H), 1.28~1.21 (m, 2H), 1.17~1.01 (m, 4H), 0.89~0.84 (m, 2H), 0.74 (t, J=7.3 Hz, 3H), 0.62 (t, J=7.5 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 166.9, 150.0, 144.2, 140.8, 137.4, 134.1, 133.6, 131.0, 130.8, 130.7, 130.1 (2C), 130.0, 128.8, 123.9, 122.7, 104.9, 63.0, 56.2, 40.3, 29.9, 28.1, 23.2, 22.7, 14.0, 10.4; HRMS (ESI) calcd for C30H34NO [M-OTf]+ 424.2635, found 424.2640.
2-(2-乙基己基)-6-氯-3,4-二苯基异喹啉-2-鎓三氟甲磺酸盐(5d): 浅灰色固体, 36 mg, 产率62%. m.p. 178~180 ℃; 1H NMR (500 MHz, CDCl3) δ: 10.19 (s, 1H), 8.75 (d, J=9.0 Hz, 1H), 7.88 (dd, J=9.0, 2.0 Hz, 1H), 7.59 (s, 1H), 7.38~7.23 (m, 8H), 7.14~7.07 (m, 2H), 4.67~4.59 (m, 2H), 1.61~1.56 (m, 1H), 1.29~1.23 (m, 2H), 1.18~1.01 (m, 4H), 0.90~0.84 (m, 2H), 0.74 (t, J=7.5 Hz, 3H), 0.63 (t, J=7.5 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 151.7, 145.2, 145.0, 138.8, 138.7, 133.5, 132.6, 130.8, 130.7, 130.4, 130.1, 129.1, 128.9 (2C), 128.8, 125.5, 125.4, 64.0, 40.3, 29.8, 28.0, 23.2, 22.7, 13.9, 10.3; HRMS (ESI) calcd for C29H3135ClN 428.2140, found 428.2142; HRMS (ESI) calcd for C29H3137ClN [M-OTf]+430.2110, found 430.2117.
2-(2-乙基己基)-8-甲基-3,4-二苯基异喹啉-2-鎓三氟甲磺酸盐(5e): 浅灰色固体, 49 mg, 产率88%. m.p. 211~213 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.93 (s, 1H), 7.87 (t, J=7.5 Hz, 1H), 7.74 (d, J=7.0 Hz, 1H), 7.47 (d, J=8.5 Hz, 1H), 7.36~7.24 (m, 8H), 7.12~7.07 (m, 2H), 4.75 (d, J=7.5 Hz, 2H), 3.06 (s, 3H), 1.56~1.50 (m, 1H), 1.29~1.21 (m, 2H), 1.19~1.03 (m, 4H), 0.91~0.85 (m, 2H), 0.74 (t, J=7.5 Hz, 3H), 0.64 (t, J=7.5 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 148.7, 144.0, 140.6, 139.9, 138.7, 137.2, 133.6, 131.8, 130.9 (2C) 130.2, 128.8, 128.7, 128.6, 128.5, 126.5, 124.6, 64.0, 40.2, 29.8, 28.0, 23.2, 22.7, 19.1, 14.0, 10.3; HRMS (ESI) calcd for C30H34N [M-OTf]+ 408.2686, found 408.2689.
2-(2-乙基己基)-3,4-二对甲苯基异喹啉-2-鎓三氟甲磺酸盐(5f): 浅灰色固体, 44 mg, 产率77%). m.p. 140~142 ℃; 1H NMR (500 MHz, CDCl3) δ: 10.20 (s, 1H), 8.75 (d, J=8.0 Hz, 1H), 8.00 (t, J=7.5 Hz, 1H), 7.95 (t, J=7.5 Hz, 1H), 7.65 (d, J=8.5 Hz, 1H), 7.18~7.06 (m, 6H), 7.01~6.93 (m, 2H), 4.69~4.60 (m, 2H), 2.34 (s, 6H), 1.59~1.54 (m, 1H), 1.30~1.22 (m, 2H), 1.18~1.00 (m, 4H), 0.88~0.82 (m, 2H), 0.74 (t, J=7.5 Hz, 3H), 0.65 (t, J=7.5 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 151.7, 144.2, 140.5, 138.7, 138.1, 137.3, 131.9, 131.2, 130.7, 130.6, 130.0, 129.5, 129.4, 129.3, 127.8, 127.0, 126.4, 63.7, 40.3, 29.9, 28.1, 23.2, 22.7, 21.5, 21.4, 14.0, 10.4; HRMS (ESI) calcd for C31H36N [M-OTf]+ 422.2843, found 422.2840.
3,4-双(4-氯苯基)-2-(2-乙基己基)异喹啉-2-鎓三氟甲磺酸盐(5g): 浅灰色固体, 42 mg, 产率68%. m.p. 135~137 ℃; 1H NMR (500 MHz, CDCl3) δ: 10.05 (s, 1H), 8.70 (d, J=8.5 Hz, 1H), 8.05 (t, J=7.0 Hz, 1H), 7.99 (t, J=7.5 Hz, 1H), 7.62 (d, J=8.5 Hz, 1H), 7.41~7.24 (m, 6H), 7.13~7.06 (m, 2H), 4.68~4.59 (m, 2H), 1.59~1.54 (m, 1H), 1.30~1.25 (m, 2H), 1.21~1.05 (m, 4H), 0.93~0.87 (m, 2H), 0.78 (t, J=7.5 Hz, 3H), 0.69 (t, J=7.5 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 152.2, 143.0, 138.7, 137.7, 137.0, 135.4, 132.4, 132.3, 131.9, 131.5, 131.5, 129.5, 129.3, 129.2, 129.0, 127.1, 126.2, 64.0, 40.4, 29.9, 28.2, 23.3, 22.7, 14.0, 10.4; HRMS (ESI) calcd for C29H3035Cl2N [M-OTf]+ 462.1750, found 462.1756; HRMS (ESI) calcd for C29H3037Cl2N [M-OTf]+ 466.1695, found 466.1702.
2-丁基-3,4-二苯基异喹啉-2-鎓三氟甲磺酸盐(
5h): 浅灰色固体, 36 mg, 产率74%. m.p.>250 ℃ (lit.
[12] >250 ℃);
1H NMR (500 MHz, CDCl
3)
δ: 10.37 (s, 1H), 8.76 (d,
J=8.5 Hz, 1H), 8.01 (t,
J=7.5 Hz, 1H), 7.96 (t,
J=7.5 Hz, 1H), 7.64 (d,
J=8.0 Hz, 1H), 7.38~7.27 (m, 8H), 7.12~7.10 (m, 2H), 4.64 (d,
J=8.0 Hz, 2H), 1.89~1.82 (m, 2H), 0.91~0.83 (m, 2H), 0.74 (t,
J=7.2 Hz, 3H).
2-己基-3,4-二苯基异喹啉-2-鎓三氟甲磺酸盐(5i): 浅灰色固体, 37 mg, 产率72%. m.p. 124~126 ℃; 1H NMR (500 MHz, CDCl3) δ: 10.51 (s, 1H), 8.80 (d, J=8.0 Hz, 1H), 8.02 (t, J=7.5 Hz, 1H) 7.97 (t, J=7.5 Hz, 1H), 7.65 (d, J=8.5 Hz, 1H), 7.39~7.36 (m, 3H), 7.33~7.32 (m, 3H), 7.27~7.26 (m, 2H), 7.13~7.11 (m, 2H), 4.67 (d, J=8.0 Hz, 2H), 1.90~1.84 (m, 2H), 1.26~1.20 (m, 2H), 1.18~1.12 (m, 2H), 1.11~1.06 (m, 2H), 0.78 (t, J=7.2 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 151.3, 143.9, 139.6, 137.8, 137.4, 133.2, 131.9, 131.3, 130.7, 130.4 (2C), 130.1, 129.0, 128.9, 128.6, 127.7, 126.3, 59.8, 31.9, 30.8, 25.8, 22.3, 13.9; HRMS (ESI) calcd for C27H28N [M-OTf]+ 366.2217, found 366.2221.
2-(2-乙基己基)-3,4-二丙基异喹啉-2-鎓三氟甲磺酸盐(5j): 棕色油状物, 42 mg, 产率88%. 1H NMR (500 MHz, CDCl3) δ: 10.01 (s, 1H), 8.64~8.61 (m, 1H), 8.14~8.10 (m, 2H), 7.89~7.86 (m, 1H), 4.73 (d, J=8.0 Hz, 2H), 3.14~3.05 (m, 4H), 1.90~1.85 (m, 1H), 1.77~1.71 (m, 4H), 1.56~1.49 (m, 2H), 1.31~1.26 (m, 6H), 1.17 (t, J=7.2 Hz, 6H), 0.93 (t, J=7.5 Hz, 3H), 0.83 (t, J=7.0 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 151.3, 144.1, 137.4, 137.3, 132.5, 130.5, 126.5, 123.5 (2C), 62.7, 41.5, 31.0, 30.8, 30.0, 28.5, 24.3, 23.6, 23.5, 22.9, 14.6, 14.5, 14.0, 10.7; HRMS (ESI) calcd for C23H36N [M-OTf]+ 326.2843, found 326.2844.
3-乙基-2-(2-乙基己基)-4-苯基异喹啉-2-鎓三氟甲磺酸盐(5k): 白色固体, 40 mg, 产率85%. m.p. 139~140 ℃; 1H NMR (500 MHz, CDCl3) δ: 10.08 (m, 1H), 8.75~8.72 (m, 1H), 8.24~8.17 (m, 2H), 8.00~7.96 (m, 1H), 7.66~7.63 (m, 3H), 7.42~7.41 (m, 2H), 4.50 (d, J=6.0 Hz, 2H), 2.87 (q, J=7.5 Hz, 2H), 1.63~1.54 (m, 1H), 1.31~1.26 (m, 2H), 1.18 (t, J=7.5 Hz, 3H), 1.14~0.97 (m, 4H), 0.90~0.81 (m, 2H), 0.74 (t, J=7.3 Hz, 3H), 0.62 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 143.4,137.5, 136.8, 132.9, 131.1, 131.0, 130.8, 130.1 (2C), 129.7 (2C), 127.2, 123.9, 63.7, 40.4, 29.8, 28.1, 23.2, 23.1, 22.7, 15.0, 14.0, 10.4; HRMS (ESI) calcd for C25H32N [M-OTf]+ 346.2530, found 346.2533.
2-(2-乙基己基)-3,4-二(噻吩-2-基)异喹啉-2-鎓三氟甲磺酸盐(5l): 棕色油状物, 31.2 mg, 产率56%. 1H NMR (500 MHz, CDCl3) δ: 10.31 (s, 1H), 8.81(d, J=8.0 Hz, 1H), 8.11 (t, J=7.5 Hz, 1H), 8.02 (t, J=7.5 Hz, 1H), 7.92 (d, J=8.5 Hz, 1H), 7.55 (dd, J=5.0 Hz, 1.0 Hz, 1H), 7.47 (dd, J=5.0 Hz, 1.0 Hz, 1H), 7.22 (d, J=3.0 Hz, 1H), 7.10~7.08 (m, 1H), 7.07~7.05 (m, 1H), 7.03~7.02 (m, 1H), 4.82~4.73 (m, 2H), 1.78~1.71 (m, 2H), 1.37~1.29 (m, 2H), 1.17~1.08 (m, 3H), 1.03~0.97 (m, 2H), 0.78 (t, J=7.8 Hz, 3H), 0.73 (t, J=7.3 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 153.2, 138.6, 138.2, 138.1, 135.8, 133.7, 132.5, 132.3, 132.1, 131.4, 130.7, 129.7, 129.1, 127.6, 127.4, 127.2, 126.5, 64.4, 40.8, 29.9, 28.2, 23.3, 22.8, 14.0, 10.4; HRMS (ESI) calcd for C25H28NS2 [M-OTf]+406.1658, found 406.1661.